Final report for FNE17-877
Project Information
In this project littleneck clams were added as a secondary crop species to a floating oyster farm on an aquaculture lease in subtidal waters (seafloor always submerged). Seed clams (1mm) were grown in a surface-water nursery alongside oysters until they were large enough to be deployed in rigid shellfish bags in the sediments below the oyster farm. Clams were checked weekly for signs of depredation and fouling, while shell lengths and the total volume of clams in each treatment were measured monthly. High mechanical/sifting losses (60%) were observed during the nursery portion of the experiment (June 21-Aug 2) but the clams that were retained grew on average 6 times in length in this time period. After deployment into grow-out equipment, clams grew to an average length of 11mm by the end of November with no statistical difference between surface and sediment treatments. Individuals as large as 17mm seen in each treatment.
The economic analysis planned for the study was affected by this as well seed size and retention as well. Even clams that showed exceptional growth were still too small to make judgments of economic viability (without a very large degree of conjecture). I believe that many of the problems experienced during the nursery phase could be solved by purchasing larger seed and/or utilizing a smaller and more rigid mesh for nursery equipment. Even with a considerable drop-off in growth rates, it is reasonable to expect clams to reach market size (1.5in – 38mm) by a year to a year-and-a-half at this site.
Project updates were posted on the farm website and social media, with over 550 individuals reached. Links to the final report will be shared in these venues. Methods and equipment schematics have been shared on FarmHack, an agriculture tool and technique wiki community. Additionally, a full account of the project will be presented at the SARE Our Farms, Our Future conference in April 2018.

Introduction:
Unlike land based agriculture, subtidal aquaculture offers the opportunity for farmers to expand their operations and diversify their crops by utilizing the vertical space of the water column. Most seafarms are monocultures that only use the portion of the water column that provides their specific crop optimal growth. Adding a species with different environmental needs would allow a farmer to expand in-place and reap the benefits of additional products and increased operational resilience to disease and pests (without needing to re-enter the arduous lease permitting process). Eastern oysters and littleneck clams are a good pairing for both husbandry and market. Small, intensive oyster farms (which make up a large proportion of aquaculture leases in the region) use floating equipment to grow oysters in the top few feet of the water column where strong currents deliver the most suspended plankton. Littleneck clams optimally grow in soft sediments and could be placed on longlines on the seafloor below an existing oyster farms. In this arrangement, the oysters and clams would not compete for food or space. The species are not closely related and could offer a backup income if disease or pests decimate one crop Littleneck clams are an attractive secondary crop for oyster farmers. They are unique in that they are most valuable at a small size. Both species are sold in the per-piece half-shell (raw) market, which offers a considerably better price than the per-pound commodity market (where other northeastern clams and mussels are sold). Additionally, farm grown shellfish often offers a premium over wild harvested shellfish on the half-shell market. The contained, subtidal (always submerged) method of growing littleneck clams tested by this project offers considerable environmental and cultural benefits over the traditional intertidal methods used on the east coast. In intertidal culture, clams are planted in netted plots or bags on exposed mudflats. This practice can be disruptive to wildlife (including sensitive shorebirds) directly excluding them from critical feeding areas or driving them away with increased activity. In many places these farming practices are prohibited or limited by law. The intertidal zone has traditionally been used as a commons for wild-harvest of clams and marine worms. There is strong resistance to removing any portion of the fishing commons for private use.
Intertidal clam culture also finds conflict with shore-front property owners and those engaged in outdoor recreation for aesthetic reasons. Growing clams below an existing oyster farm would avoid these conflicts. Where subtidal culture of littleneck clams is practiced, seed clams are sown loose on the ocean floor and are harvested by dredging or dragging (at great detriment to other bottom dwelling species and long-term habitat structure). This method is not compatible with floating oyster culture, as dragging is impractical in the footprint of the ropes and anchors that make up a floating farm. The contained subtidal method tested in this project does not rely on any bottom dragging or dredging. Clams are contained in rigid-plastic shellfish bags and are brought to the surface with ropes.
This project was designed to test the viability of growing clams in contained-culture in the sediments below a suspended-culture oyster farm.
Original project objectives included:
1) Measure the growth rates of clams under three different stocking densities
2) Note degree of mortality under different treatments
3) Record fouling and pest presence
4) Determine the market value of clean, cultured littlenecks through interviews with area shellfish buyers
5) Estimate projected value of crop (if best stocking densities were applied across the entire farm)
6) Perform a cost/benefit analysis to determine if this method could supply a viable supplemental farm income given equipment costs and labor
7) Determine actual value of crop (if market size within the study period)
Cooperators
- - Technical Advisor
Research
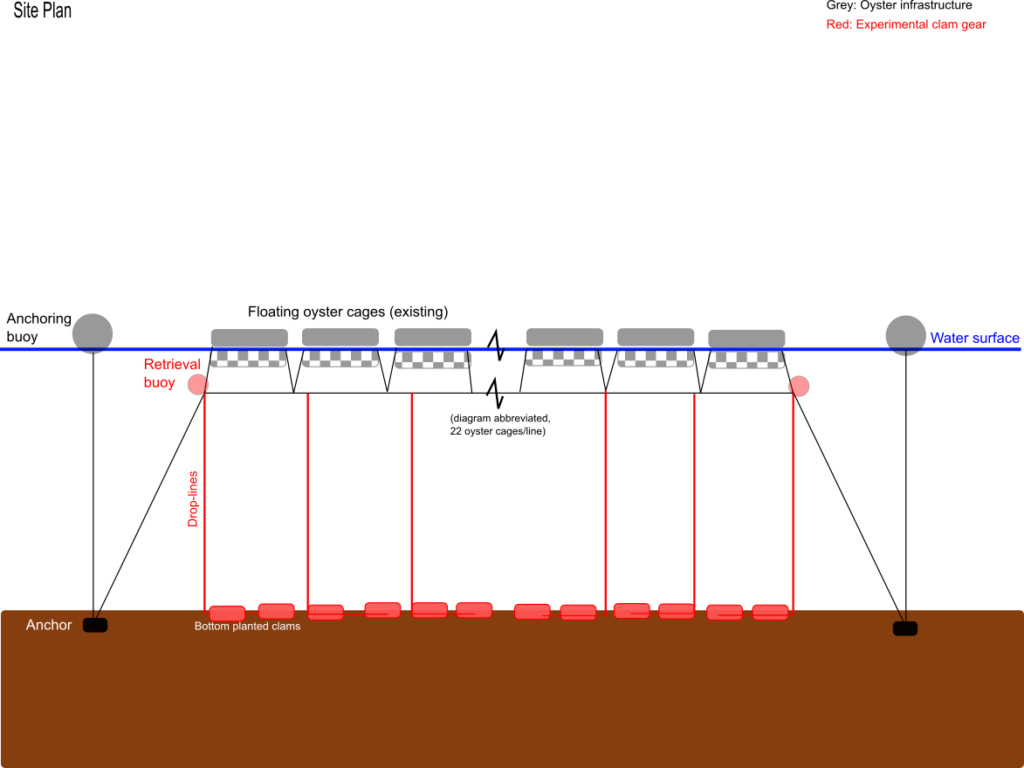
Nursery and bottom-planting gear were built in the spring of 2017
Nursery gear consisted of 0.7mm soft mesh scallop bags held open with sheets of HDPE screen. These bags were deployed in 4 tier lantern nets built out of 18mm mesh oyster bags, suspended under mooring buoys. Later a portion of these soft nursery bags were moved into floating oyster cages (as farm space allowed).





Clam seed (1mm) arrived in mid June. The seed was split into the 0.7mm soft mesh bags, was sifted on-board in a seawater filled tank, and was deployed in lantern nets (later moved to oyster cages). Seed lost through the mesh was grown for several weeks in the on-board tank until it was large enough to be retained by the 0.7mm the mesh. Soft mesh bags were swapped out weekly to avoid excessive algal fouling.
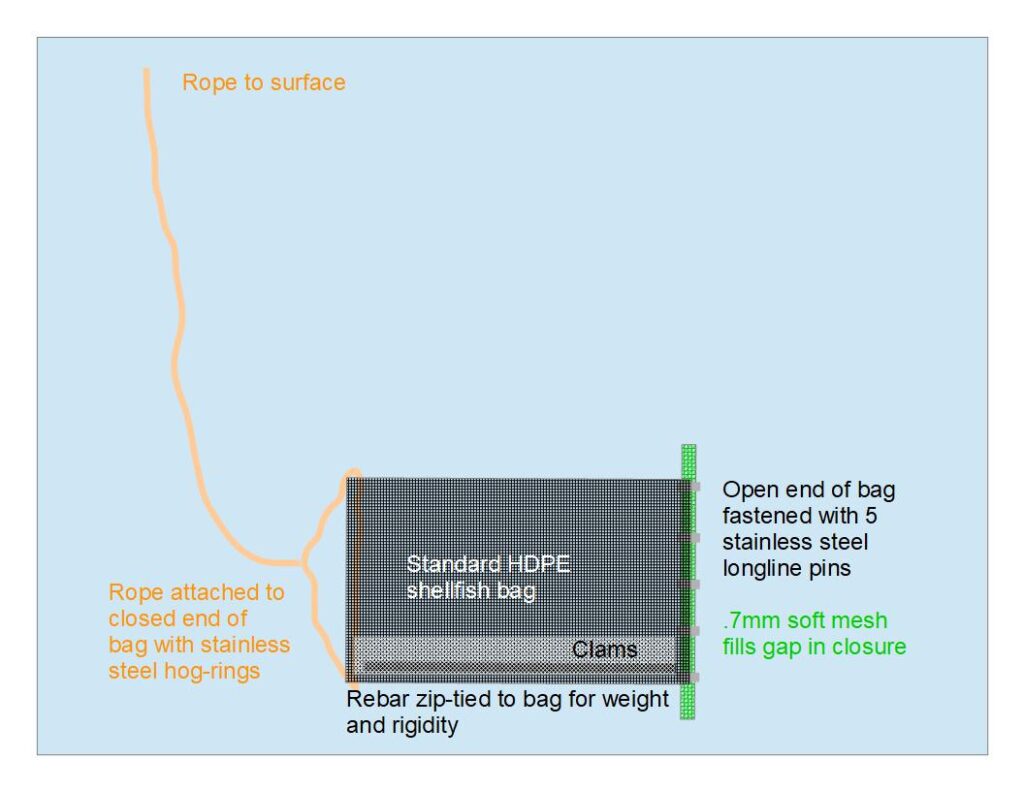
Seed was sieved again in August. Individuals that were large enough to be retained by the ridged mesh 4mm bottom-planting bags were set aside. These individuals were split into three different groups. Two of the groups were deployed in bottom-planting bags. These consisted of commercially available ridged-mesh oyster bags, strengthened with stainless steel hog-rings and weighed down with rebar. These bags were lowered with ropes to the soft substrate below the farm in two different locations-one at the northern end of the farm, the other at the southern end. The remaining group of larger clams was retained in surface gear similar to the nursery equipment. This treatment was included to gauge the optimal time to switch from nursery equipment to bottom-planting.


------------------------------------------------------------------------------------------
This sieving/planting phase began a month later than originally scheduled because clams had not reached an appropriately large size in July. There were no nursery-growth benchmarks to compare to in comparable conditions, so this may amount to a poor timing estimate during the project-planning phase. An unusually strong red-tide event that slowed oyster growth on the farm in June and July may have affected the clams' growth as well.
Bottom planted bags were hauled to the surface weekly to check for fouling and to prevent them from sinking too deep into the sediments. Surface/nursery bags were rotated between sun and shade on a weekly schedule to avoid buildup of algae.
Length, volume, fouling, predator presence, and mortality measurements were taken on a monthly schedule until the end of November. The width and depth shell measurements that had initially been planned were abandoned (unlike the oysters used in prior experiments the clams dimensions were almost universally proportional).

Nursery Phase-
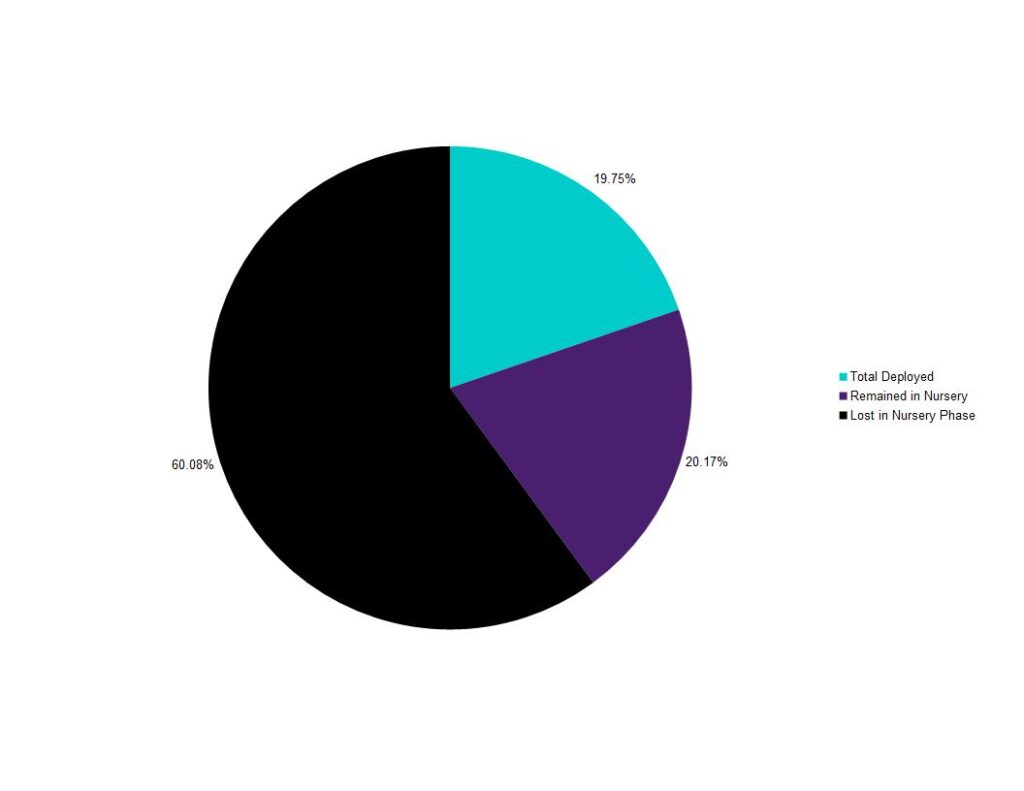
The vast majority of the loss observed in this experiment occurred during the nursery-phase (see chart). Just over 60% of the 1mm clam seed was lost between June 21 and Aug. 25 during the nursery portion of the experiment. Though significant losses were anticipated and planned for, this figure was higher than expected. The sand-like 1mm seed was exceptionally difficult to work with. Clams that were retained on 1mm mesh in the hatchery before sale sifted through .7mm mesh when deployed in the field- even with pre-deployment sieving. There was no notable mortality in the nursery.

Nursery Growth:
Clams grew at a very high rate during the nursery-phase. Shell length grew over 6x (on average) in the time between June 21 and Aug. 8, despite highly unusual red-tide conditions that stunted oyster growth during a portion the same period.
Nursery Fouling and Predation:
The .7mm mesh nursery bags picked up a small amount of algal fouling. This was easily mitigated by rotating sun-facing bags into the shade and switching fouled bags with fresh bags on a weekly basis. Bags were never fouled enough to inhibit water flow and feeding.
Predation was not an issue in the contained system of the nursery. No predators were observed within the nursery bags.
Growout Phase-
Growth:
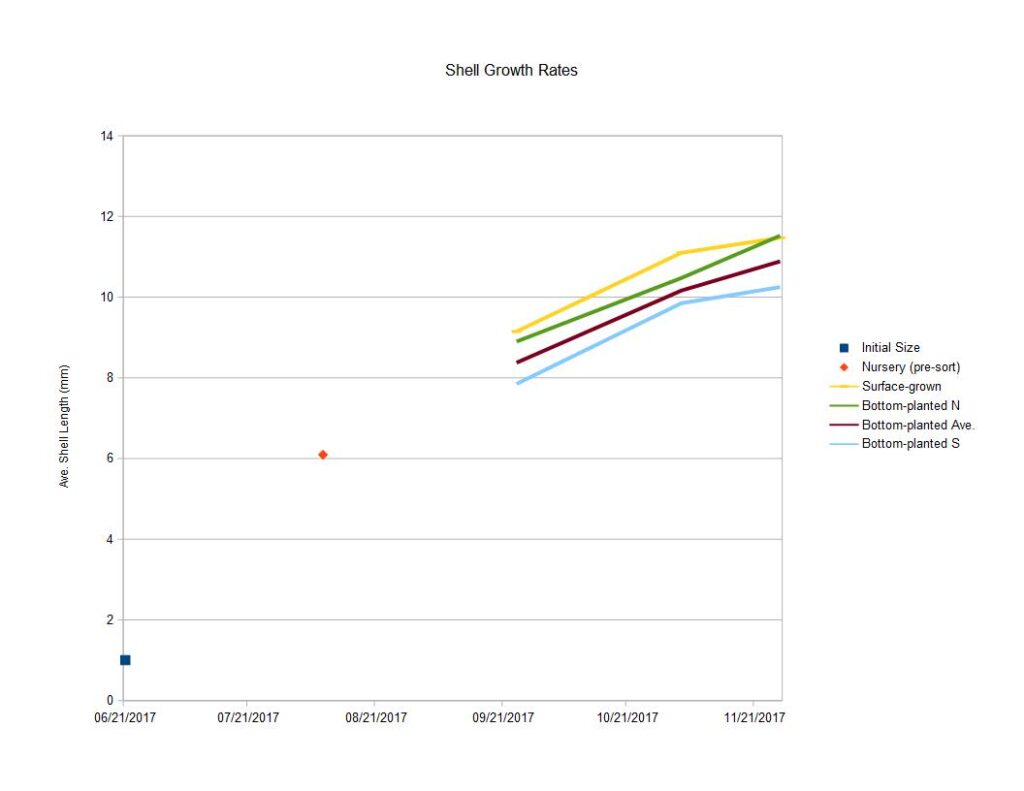
Shell length grew in both surface and sediment treatments by significant margins, but differences between treatments fell within standard error. The high losses in the nursery-phase reduced the number of replicates in the study, contributing to large standard error figures. The trends seen in this measurement suggest clams grown on the surface grew the fastest until late October, while sediment-grown clams grew faster in the final month of the experiment. On average, the surface-treatment clams reached a length of 11.5mm by the end of November and sediment-grown clams reached a length of 10.9mm. In both treatments, individuals were observed measuring as long as 17mm.
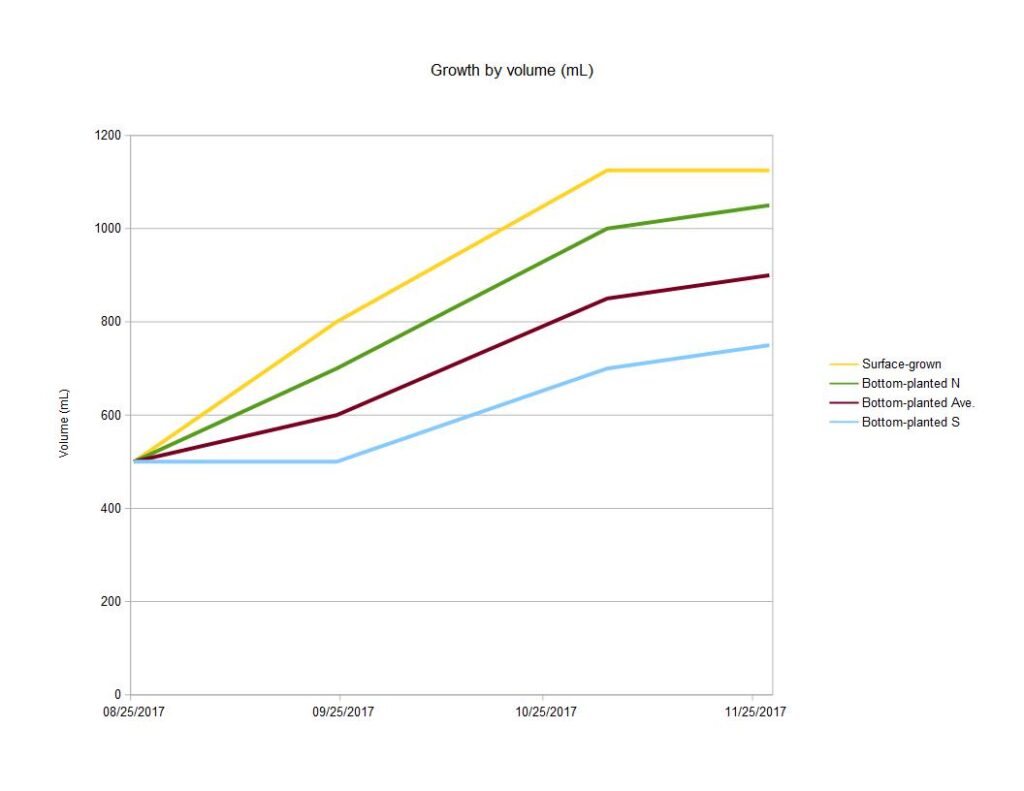
The total volume of clams in each treatment mirrored the trends seen in shell length measurements. Clams deployed on the surface grew faster than sediment-grown clams until late October, when the trend reversed. This could be a seasonal/environmental effect (surface waters cool faster in the fall) or a product clam age/growth stage (larger clams grow faster in the mud). Additional measurements in subsequent seasons could clarify this question.
Measurements on graphs below include
1.surface-grown clam growth (yellow),
2. bottom planted clam growth-Northern deployment (green),
3. bottom planted clam growth-Southern deployment (light blue),
4.average growth of bottom-planted clams - average of Northern and Southern deployments (red)
Fouling/Colonization:
Surface-grown clams picked up a very small amount of blue-mussel fouling in November (6 individuals/bag). For all practical purposes this is negligible. Sediment-grown clams were free of fouling organisms. Fouling across the farm was exceptionally light in 2017 compared to other seasons. These results may reflect that trend.
Other non-fouling organisms colonized the clam-growing equipment. Marine worms were found in all treatments in small numbers. Bags with sediment-grown clams picked up small numbers of juvenile razor clams and soft-shell clams (both commercially valuable species). One sediment bag also contained mud snails. Rock crabs were also observed in the bottom-planted bags (see next section)

Predation/Mortality:
Rock crabs were observed in small numbers in the sediment-planted clam bags (0-2 individuals in each weekly bag-check). It is unclear whether they entered through the bag-flaps as adults or settled in as juveniles and grew in-place. Despite the presence of this predator, mortality was very low- less than 1% at the final measurement for all treatments.
This project demonstrated that littleneck clams can be grown in the same footprint as
eastern oysters, adding an additional crop to an existing farmwithout sacrificing lease-space. Both the floating nursery and sinking grow-out bags were technically viable methods for growing hardshell clams and were compatible with existing oyster farm practices. Growth rates were excellent in all treatments, with rates as high as 17 fold shell growth seen in some individuals. Fouling, depredation, and other mortality was negligible during the grow-out phase. The labor involved in tending the project was very reasonable, with measurements and observations taking the vast majority of the field-time. The amount of maintenance needed for commercial clam-culture would be comparatively minimal without the added labor necessary for this experiment.
There were significant difficulties related to the small seed available for the project. The vast majority of loss seen in this project was due to the difficulty handling such tiny seed clams and the lack of commercially available equipment for growing them in the field at this stage. The clams that remained after the nursery phase were too small and too few to run the planned density tests as designed. This shortcoming was due to both the novelty of the farming technique and the lack of existing growth-studies on which to base the project timeline. The economic analysis planned for the study was affected by this as well seed size and retention as well. Even clams that showed exceptional growth were still too small to make judgments of economic viability (without a very large degree of conjecture). I believe that many of the problems experienced during the nursery phase could be solved by purchasing larger seed and/or utilizing a smaller and more rigid mesh for nursery equipment.
Commercial potential:
Local wholesale buyers placed the price of farmed local littleneck clams at between 18 and 33 cents each. Restaurants surveyed valued clams at 50 cents per piece.
Even with a considerable drop-off in growth rates, it is reasonable to expect clams to reach market size (1.5in - 38mm) by a year to a year-and-a-half at this site. Only a third of the growth seen in 2017 would need to be observed in 2018 to achieve this. If negligible mortality rates continued, the 14550 clams in the growout-phase of this experiment could have a wholesale market value ranging from $2619-$4802 and a direct-to-customer value of $7275. These numbers could more than double if the additional 14850 clams retained in the nursery could reach market size by the end of the 2018 season.
With relatively low seed costs (.006 cents per 1mm clam), durable equipment, and minimal labor (outside of experimental measurements), littleneck clams could make a very attractive second-crop on small subtidal oyster farms, particularly if seed losses could be reduced.
Education & outreach activities and participation summary
Participation summary:
Outreach for this experiment has consisted of project updates distributed through the farm website and social media. Over 550 users have viewed project-related posts on the webpage in the last year without formal promotion (does not include social media views). A stand-alone page to display 2017's final results is set to go-live upon the approval of my final report and will be promoted through widely read farmer-focused industry list-serves. Tool and technique information has been uploaded to FarmHack (a farm tool design and modification wiki), and will include links to the approved final report on SARE's server.
In addition, I've informally explained the work in-depth to 4 neighboring farmers and 7 shellfish dealers/buyers. This project will also be presented during poster sessions in SARE's Our Farms, Our Future conference in April 2018.
Learning Outcomes
To date, 15 farmers have heard a first-hand report of this techniques feasibility.
One farmer has experimented in a similar manner with wild seed stock.
Project Outcomes
I will continue growing clams as a supplemental/backup crop, though I plan on adjusting my nursery practices or cutting out the nursery phase entirely by buying (newly available) larger seed. The high growth rates seen in this experiment show the method is technically viable, and presents an opportunity to tap into the unaddressed market of locally farmed clams.
Loss during the nursery phase was the biggest inefficiency in this experiment. An improvement in nursery methods could cut into the only substantial loss seen over the course of the project. Use of a smaller and/or more rigid mesh could keep small seed from sifting through nursery gear during bad weather/high seas. The same result could also be achieved with existing equipment through the purchase of larger clam seed (available locally in 2018- but not during the course of the experiment).
The loss of seed and its small starting size forced me to cut out the density experiments I had originally planned and reduced the number of replicates in my experiment. Without determining an optimum density, I was unable to calculate the farm carrying-capacity or perform the farm-scale economic analysis of the technique. Further study of optimal planting density would be very useful in determining the economic viability of this technique.
Despite nursery losses and the narrowing of the experiment, I was able to observe that it was possible was able to grow clams at a very high rate with minimal fouling and minimal loss once clams were planted in final grow-out equipment.
I will continue to grow clams in this manner on my own farm in the future.
Aside from experimental measurements and documentation, labor for this method was relatively low. Clam growth was very fast and depredation was negligible. If the levels of labor and grow-out loss can remain low, this method of growing clams could present an easy way to diversify existing oyster farms.
Demand for locally-farmed clams is strong and wild stocks of other species of clams continue to decline. I hope to tap into the new and growing market for Maine grown littleneck clams, providing my farm with a more diverse offering of products and higher a total yield. I think other small scale oyster farmer's in the region could easy adopt and benefit from these techniques, as well as wild-harvest clammers transitioning to aquaculture.