Final report for FNE19-940
Project Information
Decreasing access, uncertainty, and concern about wild harvest seafood sustainability is leading to an increasing interest in aquaculture. While seaweed aquaculture is a promising new area for growth in the US, production is limited to a small number of farms that cultivate just one or two species of kelp. The red seaweed nori (Porphyra spp.) is the most valuable seaweed in the world, cultivated on a large scale in Asia. We proposed to develop a new approach to cultivation in Maine waters for a native species of nori, Porphyra umbilicalis, utilizing an innovative three dimensional vertical growing system. Nursery and farm cultivation techniques were explored for this new potential crop. Initial success with seed production led to biofouling issues in the nursery, resulting in smaller than anticipated seed. Cultivation of smaller seed on the farm was inhibited by a high degree of diatom fouling, which smothered the young seed. A small section of seed that escaped the heavy biofouling grew into healthy, clean plants, almost ready to harvest by February. Our work was included in a number of presentations, workshops, and tours as a promising new crop under development.
This project seeks to develop reliable nursery, cultivation, and harvesting protocols for production of nori using our innovative three-dimensional vertical cultivation system.
Objective 1: To develop nursery protocols for nori that will produce viable seed. The measurable benefit will be the new aquaculture crops this new seed will enable farmers to grow.
-What affects spore viability of wild harvested and nursery reared seed stock, and how can it be improved?
-How can freezing be utilized to store spore solution and seeded lines?
Objective 2: To grow out seeded growing systems on open water farm to determine optimal cultivation parameters. This will inform seaweed farmers of seasonality, culture period, and techniques for grow out.
What are the optimal growing seasons and environmental conditions?
-What are the optimal husbandry techniques for each crop?
-How to optimize quality and yield, avoiding issues of biofouling?
Objective 3: To develop and trial nori growing system harvest and processing systems. Seaweed farmers will be able to harvest and process their own crops, or sell to other buyers.
-How will the vertical growing systems be managed at sea for tending and harvest?
-Can we optimize yields through continuous harvest, what are the options for post harvest processing?
The potential for growth in new ocean crops is immense in the cold temperate waters of Maine and New England. Maine is a resource dependent state, with a marine fisheries and farming heritage that is changing with a changing world. A once diverse wild-caught seafood industry has declined by almost 70% in the last 20 years, creating a dominant lobster fishing industry that is dangerously dependent on a species that is sensitive to ocean warming, disease, and stress (Steneck et al., 2011). Would-be fishermen are barred from entering closed or limited entry fisheries, and the loss of many viable fisheries like shrimp and groundfish have reduced opportunity for working in seafood. Potential new land farmers in coastal Maine face the challenges of land affordability, lack of infrastructure and financing, and poor soils (Ilseman, 2014). Maine’s marine shellfish and seaweed aquaculture industries, however, offer opportunities that are lacking in traditional fishing and farming. Aquaculture is still an open opportunity; anyone can apply for a lease to farm in Maine waters and acquire a farm. Seaweed, especially, has great potential for become a new crop for Maine , with a host of benefits. Seaweed aquaculture is a sustainable and environmentally responsible form of food production, requiring no fresh water, fertilizers, pesticides, herbicides, or arable land, and very little fossil fuel, and can be USDA organically certified in Maine through the Maine Organic Farmers and Gardeners Association (MOFGA). A new seaweed aquaculture industry in Maine could contribute to Maine’s working waterfront and marine and agricultural heritage by providing new opportunities for diversification in the traditional fishing and farming rural communities, especially in rural downeast coastal areas, while producing healthy sustainable seafood and agricultural products. Seaweed crops have a winter growing season from fall to spring, opposite the busy lobster and tourist season, making it an attractive option for economic diversification for lobster fisherman, shellfish farmers, or others interested in sea farming.
While the opportunity is great for the development of a seaweed aquaculture industry in Maine, the technology for commercial scale production of economically valuable crops does not yet exist. Maine was the first state to commercially cultivate sugar kelp in 2010, but the value of this crop is very low, showing a need for diversification. We propose to develop new cultivation technology for a higher value native red seaweed crop in Maine in order to diversify the seaweed aquaculture industry and increase economic value.
The red seaweeds known as nori (Porphyra spp.) are highly nutritious, highly valued, relatively rare sea vegetables from the North Atlantic, making them species of interest for aquaculture. While there is increasing interest from farmers and buyers for the high value nori, there is no commercial source of seed, and no growing system that will yield a commercially viable harvest. We propose to develop reliable nursery systems for production of high quality seed, new cultivation systems for increased biomass production in Maine waters, and harvesting and processing technologies to increase efficiency and value of the new crops.
Cultivated extensively in Japan, Korea, and China, nori is the most valuable seaweed in the world. “Nori” is a general term for a hundred and fifty plus species of a red foliose sheet like seaweed (Porphyra sensu lato, Bangiales, Rhodophyta) found around the globe. Large scale nori cultivation takes place on extensive fixed or floating nets that require regular drying. The typical Asian-style nori farm would be difficult to manage in Maine, with extreme winter weather and massive floating mats of rockweed (Ascophyllum nodosum) that would tangle in the nets. A submerged vertical growing system strategically set, tended, and harvested, has the potential to produce a viable crop without the regular drying and surface gear, opening up the potential for scaling of the technology
in the United States.
Porphyra umbilicalis is a unique species of nori on the Maine coast in that it directly regrows blades from neutral spores produced by mature blades, bypassing the normal diploid conchocelis phase present in other nori species (Blouin et al., 2007). A high intertidal seaweed, P. umbilicalis is regularly exposed to freezing, freshwater, and desiccation. P. umbilicalis has been successfully seeded on lines and nets in the nursery, but commercial production has not been realized due to a lack of growing technology that will work in Maine waters. Wildemania amplissima, a larger, subtidal nori species, also has potential for cultivation in New England, but requires the development of mass conchocelis cultures in the nursery for spore production (He & Yarish, 2006; Carmona et al., 2006). This is a secondary target candidate for aquaculture.
Nori plants are much smaller in size than the large brown kelp species that are currently cultivated in Maine. While kelp plants can reach 10-15 feet and biomass at 10-20 lbs per foot at harvest, nori plants are only 6-10”, and yield much lower biomass at harvest. For this reason, the typical kelp horizontal longline system is not appropriate. To increase surface area for cultivation while reducing fouling at the surface, we propose to trial a vertical three-dimensional growing system. Our new proprietary design utilizes linkable “Growing Modules” (GM) with three-dimensional structured systems that offer increased growing area while withstanding extreme ocean environments. The systems efficiently optimize farm space and gear, and may be integrated as a potential short season crop for diversified year round sea farm production. Each module is constructed with lobster trap wire for strength, lightness, and durability in the marine environment. The system is designed to be easily integrated with other shellfish, seaweed, or fish farming systems, can be easily adopted by traditional fishermen, and utilizes readily available traditional marine materials. The new design has substantial benefits over existing seaweed farm system designs, permitting denser growth and higher yields than long line systems, with reduced fouling than is common on net systems, and no tangling as is common in dropper lines. This 3-D growing system is reusable, uses readily available materials, is easily deployed and harvested, can be adapted to a wider range of growing areas, and is easily integrated into existing aquaculture and fishery operations. This new technology is especially interesting to the growing oyster industry as a means to diversify their farming operations in the colder months, and to farmers that are limited to more protected, shallower areas not suitable for kelp farming. The seed required for the growing modules would be produced in the Springtide nursery utilizing similar kelp seed production systems. Reliable seed is the foundation for all successful seaweed crops; so native nori seed production will be optimized in the nursery to provide healthy, timely, dependable, organic seed for this new industry. The establishment of nursery seeding facilities and relevant culture systems are crucial to the development of nori cultivation in the United States. The technology developed for nori in Maine can be adapted to other small, high value seaweed species and areas. New high value seaweed crops will be a source of sustainable versatile biomass with significant positive economic and environmental impacts, especially in rural coastal communities.
CITATION LIST
Blouin, N. B., X. G. Fei, J. Peng, C. Yarish, and S. H. Brawley. 2007. Seeding nets with neutral spores of the red alga Porphyra umbilicalis (L.) Kützing for use in integrated multi- trophic aquaculture (IMTA). Aquaculture 270, 77-
Carmona, R., Kraemer, G.P., Yarish, C. 2006. Exploring Northeast American and Asian species of Porphyra for use in an integrated finfish-algal aquaculture system. Aquaculture, 252: 54-65.
Green, L and C Neefus. 2014. The effects of short- and long-term freezing on Porphyra umbilicalis Kützing (Bangiales, Rhodophyta) blade viability. Journal of Experimental Marine Biology and Ecology 461, 499-503.
He, P and C Yarish. 2006. Developmental regulation of mass cultures of free-living conchocelis for commercial net seeding of Porphyra leucosticta from Northeast America. Aquaculture, 257, 373-381.
Ilseman, KA. 2014. Maine Farmers’ Perspectives: Barriers and Optimism to Providing Food to
Feed Maine People, A Qualitative Study Using Focus Groups. Electronic Theses and Dissertations.
Steneck, RS et al, 2011. Creation of a Gilded Trap by the High Economic Value of the Maine
Lobster Fishery. Conservation Biology. 25:5. 904-912.
Redmond developed kelp nursery systems that led to the initiation of kelp aquaculture in the US in 2010. From 2012-2016, she worked as the nation’s first seaweed aquaculture Sea Grant extension specialist, spurring the development and expansion of seaweed farming. While at Sea Grant, Redmond managed a seaweed research nursery at the UMaine Center for Cooperative Aquaculture Research, developed new crops and techniques, education programs, hosted the “Seaweed Scene” farming conference, and co founded the Maine Seaweed Festival. Her work resulted in new farms, new farmers, new seaweed products, and new crops, including skinny kelp and winged kelp, and research on the cultivation of native nori, dulse, gracilaria, and horsetail kelp. Redmond left her public position to move the R&D of seaweed aquaculture into commercialization, and founded Springtide Seaweed, an organically certified 25acre seaweed nursery and farm, and co-founded the Maine Seaweed Exchange. Andrea “Trey” Angera, Jr. is managing director (COO) of Springtide, and director of the Maine Seaweed Exchange. He directs processing operations and product development, coordinates events, provides third party consulting services to the seaweed
aquaculture industry, and creates and conducts educational programs for the company. His extensive experience in business startups and operations, and background in the organic meat and seafood industries are invaluable for the development of the new seaweed aquaculture industry. Springtide Seaweed produces and sells certified organic kelp seed, as well as farmed fresh and dried seaweed powders, flakes, and value added products. They also offer a wide range of seaweed aquaculture educational and consulting services.
Cooperators
- - Technical Advisor (Educator)
Research
Seed production
Seed cultures were produced for nori (Porphyra umbilicalis) by collecting reproductive plants in April, July, October, and November from the wild. Plants were cleansed via fresh water rinsing, filtered seawater rinse, physical scraping and wiping of the blades, and desiccating overnight in damp paper towels. Submerging blades in clean filtered seawater stimulated release of spores, and seaweed string wound around 2” diameter pvc pipes inside of 20 gallon tanks were inoculated in the nursery by pouring spore solution over string, covering in UV treated, filtered seawater, and leaving with gentle aeration to attach at 10 C, in cool white fluorescent lighting (~5-10 µmol photons m2 s-1, 12 h: 12h (L:D) for 12 hours, then adding stronger aeration, increasing light 50-60 µmol and changing cultures every 5 days with f/2 media until seed germinated and developed into small blades.
Spore viability : At release, spores were settled onto glass slides and cultivated in the culture tanks described above. Spores were tested for viability by determining percent germination per 100 random spores 5 days after release under the microscope.
Nursery reared seed stock and viability
Porphyra umbilicalis spores were settled on glass plates and cultured to 1mm at 10C, light at 12:12 LD, 30-50 µmol photons m2 s-1 (Blouin et al., 2007), when they were scraped off and tumble cultured inside an environmental chamber in order to develop nursery reared seedstock for spore production. Seeded lines of P. umbilicalis were frozen for one month and viability compared to non-frozen lines.
Culture season
Seed was produced in April, July, October, and November, and seed placed out on the farm in October, December, and January on coated wire cages. Plant growth and biofouling was monitored through the season until February.
Seed Production
Culture string was seeded in the nursery from wild collected reproductive plants in April, July, October and November (see table). For all collections, release and germination rates of spores was high, and seeded spools were thickly seeded with developing blades within 2 weeks of seeding. All spools developed normal, clean, juvenile blades for the first four weeks, then developed increasingly damaging biofouling.

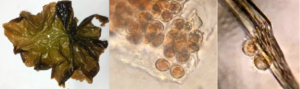


Nursery cultivation of seed stock suffered from high loads of algal and protist contamination, resulting in a decreasing quality of seed. Initial seeding, germination, and development was healthy and clean, but seed culture health began to quickly diminish after about one month in the nursery. A single celled and filament-forming green algal growth appeared in small patches upon the seed string, and quickly spread to all spools and the inside walls of the tanks. Despite frequent spraying of the seed spools and cleaning of the tanks, the algae continued to spread. Juvenile blades were prey to ciliate attack, and exhibited dead patches on the blades. Severe and persistent contamination is the major issue in successful production of seed for seaweed farms, as was exhibited by this trial. The second seeding in October did not suffer from the same heavy contamination load as in the first trial, but seed was in the nursery for less time and placed out on the farm at a smaller size.


Spore viability : At release, spores were settled onto glass slides and cultivated in the culture tanks described above. Spores were tested for viability by determining percent germination per 100 random spores 10 days after release under the microscope. Spore viability was very high for all seedings, with a slightly higher percentage in October. July germination was at 95%, and October germination was at 97%. Spores were released readily from wild harvested plants and were available in high densities.
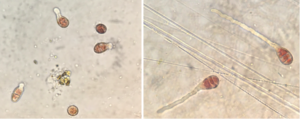
Nursery reared seed stock and viability
Porphyra umbilicalis spores were settled on glass plates in July and cultured to 1mm at 10C, light at 12:12 LD, 30-50 µmol photons m2 s-1 (Blouin et al., 2007), when they were scraped off and tumble cultured inside an environmental chamber. These plants were cultured in the nursery to several cm in length, but blades were completely covered in brown algal contamination and experienced blade disintegration. These plants had to be discarded and a new culture is being developed. Wild harvested blades are also being cultivated in the nursery for the establishment of clean seed stock. Wild harvested blades were trimmed, cleansed, and are being tumble cultured to determine efficacy of utilizing wild collected for seedstock cultures.

Cultivation
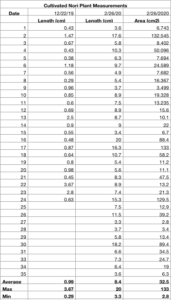
Seeded lines were placed out on the farm for open water cultivation through the fall and winter. Seed was wound around two coated lobster wire frames, one cylindrical and one rectangular, and hung from a buoy approximately 1 foot below the surface of the water. The farm experienced a heavy diatom set in October, and continued to suffer from the same fouling through Feb., which coated seed lines in a heavy film of ropy diatom growth that was difficult to remove through spraying. Fouling diatoms are probably Berkeleya rutilans, a colonial tube forming diatom that can form filamentous-like structures by developing a mucilaginous sheath. This growth is a nuisance type of biofouling not easily removed and can persist throughout the growing season. Heavy growth of this algal contaminant can smother smaller seed. Seed frames were sprayed every two weeks and checked for growth, but most of the seed lines did not develop any blades due to the smothering nature of the diatom contamination. An extra seed spool that had been hung on the farm did develop blades on a portion of line that had become unraveled and was not subject to the same fouling as the seed on the frames. This line was wound around a rope and positioned between two surface buoys to track growth of the blades. When discovered in December, blades were an average length of 1 cm (min .3cm, max 3.67cm). On Feb. 26th, average blade length of 8.4cm (min 3.3cm, max 20cm). While the line was covered in a diatom film, after spraying, plants were clean and healthy, not carrying any biofouling on the blades themselves.




Intertidal trial
In order to test an intertidal environment, seeded line was placed out between two intertidal pilings so that plants experienced regular tidal cycles. After one week, small seed was still visible on the line, but after two weeks, the string was completely clean. This is probably due to chafing from nearby Ascophyllum, which is almost impossible to keep out of shoreline areas. Any type of surface or intertidal cultivation will always struggle with the large mats of Ascophyllum (rockweed) which float through Maine coastal waters.

Temperature and Light
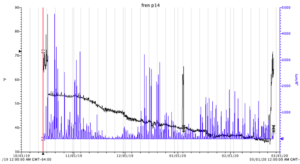
Temperature and light was measured continuously with Onset Hobo pendant temp/relative light data meter. The temperature remained around 53 F through October and November, when it started to decline steadily through February, to a low of 33 F at the end of February. Low light levels indicate a degree of diatom fouling on the meter itself, and a normal decrease of light through the winter months.
Nursery raised juvenile blades suffered from various degrees of contamination from epiphytes, endophytes, and marine fungi. A red endo-epiphyte belonging to Acrochaetium was observed growing from the base of juvenile blades, as well as a filamentous brown (probably Ecotcarpus), and the clear hyphae of marine fungal organisms. The persistent and destructive green algae present in cultures could be either Acrosiphonia or Spongomorpha, the two being nearly indistinguishable, and present as single cells and filaments. Algal, bacterial, fungal, and protozoa contamination of seed can impair the health, growth rates, and survivability of the juvenile plants, and can infect and compete with the cultivated plants on the farm, and should be avoided at all costs. The high infection rates in the wild blades and contamination of nursery seed emphasize the need for clean nursery reared seedstock for the production of spores. Wild blades are difficult to clean, and it is apparent that contaminating organisms are being passed from parents to juveniles.
Nursery reared juvenile blades were consistently successfully cultivated to a 0.5-1mm size, but then would succumb to fouling and poor health. Porphyra umbilicalis has a high intertidal distribution in the wild, and survives in a constant cycle of emersion and submersion. Continual submersion in the nursery could be contributing to poor health and lack of growth after the 1mm seed size is reached, coupled to the increasing levels of biocontamination. Wild plants, when cultivated in a constantly submerged environment, will all begin to exhibit poor health and eventual disintegration, unless subjected to a daily drying period. Commercial nori production relies on an elaborate net system that allows plants regular desiccation to mimic natural environment. Other local subtidal nori species might make better candidates for aquaculture. Larger seed may be more successful on the farm, as evidenced by the growth of plants on a small section of line that contained seed at 1-3 mm in December, developing clean and normal blades 3-20cm by February.
Every effort to domesticate a new seaweed species from the wild reveals a wealth of information about strategies, opportunities, and issues that will inform continual progress toward cultivation. Further development of a local nori species for culture will need to involve reducing biocontamination risk in the nursery, strategies for overcoming heavy diatom fouling on the farm, increased seed size, and investigations into new species that will be more amendable to continual submersion.
Education & outreach activities and participation summary
Participation summary:
Redmond, S. 5/16/19. "Seaweed Farming in Maine". Schoodic Institute at Acadia National Park. Winter Harbor, ME. Invited Speaker. 20ppl.
Redmond, S. 6/19/19. "Farming the Future: The Promise of Ocean Seaweed". Shaw Institute, Blue Hill, ME. Invited Speaker. 60ppl.
Redmond, S. 7/17/19. "Farming Seaweed in Frenchman's Bay". Sullivan-Sorrento Historical Society. Invited Speaker. 20ppl.
Redmond, S, and A. Angera. 8/10/19. "Seaweed: Food, Feed, and Fertilizer." Northeast Organic Farming Association Summer Conference, Hampshire College, Amherst, MA. Invited Speaker. 40pp.
Redmond, S. 10/25/19. Korean Broadcasting System. Farm Tour for documentary. https://www.youtube.com/watch?v=qU5KOgkbgK0&t=827s. Korean national audience.
Redmond, S. 12/11/19. "Seaweed in New England". New England Vegetable and Fruit Conference. Manchester, NH. Invited Speaker. 80ppl.
Redmond, S and A. Angera. 1/11/20. Seaweed: Food, Feed, and Fertilizer. Northeast Organic Farming Association Winter Conference, Worcester State University, Worcester, MA. Invited Speaker. 40ppl.
Redmond, S and A Angera. 1/18/20. Seaweed Farming Workshop. Maine Seaweed Exchange, Gouldsboro, ME. Organizers. 30ppl.
Learning Outcomes
This project showcased issues with seed production and farm growth for a local species of seaweed, and will allow for continued focused research on key bottlenecks, including nursery production, biofouling issues, and alternative nori species for cultivation.
Project Outcomes
Any effort towards the cultivation of a wild seaweed species will yield invaluable information and experience, and this project was no exception. It highlighted the challenges with domesticating a high intertidal nori species in Maine waters, as well as underlined the need for advanced nursery systems for the production of healthy seed. This project will inform further research and development into the cultivation of valuable local nori strains for food and medicine.
The challenges associated with nursery cultivation are many, and will require more sophisticated systems for control of biofouling organisms. Though you may succeed one year in producing seed, another entire set of unforseen issues will arise the next. Because seaweed cultivation is such groundbreaking work in the US, it is difficult to determine issues without gaining valuable experience through experimental trials. These experiences strongly suggest a more focused investment in nursery technology and techniques for reliable seed production.
