Final report for FNE22-033
Project Information
This project seeks to define the concentration of hormone needed to induce rooting on hybrid chestnut suckers using a common stool bed layering method to improve the profitability to the farmer. This study involved searching past technical papers and talking to European nurseries and farmers to understand best practices for European chestnut trees and see how they can best be applied to our hybrid cold hardy chestnuts that are worthy of cloning.
Gleaned from the technical papers on when to apply hormone and potential ranges of Indole-3- butyric acid (IBA-K) an experiment was devised to test out the ranges that would give the best rooting results. It was found that applying 4000 ppm of IBA-K hormone treatment 60 days before the first frost at the location of the farm was the best time as shoot growth typically slows at this time. Using this method 44% of the suckers dug up were rooted and had the best developed roots.
By layering known good chestnut trees chestnut orchards will see an improvement in nut productions and only have to plant half of the trees that is normally done when planting seedlings. When planting seedlings twice as many are planted and in years 8 through 10 half of the unproductive seedlings are culled. The cost of the seedling, fertilizer, weeding, spraying, and maintenance are halved for the first 8 to ten years by planting on half of the trees.
This information is available freely from the websites of Tompkins County CCE, NY, Z's Nutty Ridge and New York Trees Crops Alliance Cooperative. The brochure was distributed at two educator conferences, the national joint conference of Chestnut Growers of America and the National Nut Growers of America and hosted by The Chestnut Restoration Project at SUNY ESF. Also, with many one-on-one discussions at booths, a field tour and field day. The information was shared with relevant permaculture, agroforestry and nut tree groups on Facebook and newsletters to over 2000 subscribers.
This project seeks to define the concentration of hormone needed to induce rooting on hybrid chestnut suckers using a common stool bed layering method to improve the profitability to the farmer. The rooted suckers are identical copies of the mother trees and are called clones. Clones are asexually reproduced copies of a superior tree providing genetically identical trees and are often called a “cultivar”.
The objective for this proposal are:
- Improve orchard profitability via cloning superior trees.
- Develop the proper hormone “recipe” and illustrated instructions to enable any grower can clone via stool bed layering their superior trees.
- New cultivar generation as superior trees are cloned via stool bed layering they can become named “cultivars” as copies can be made to be shared throughout the Northeast US.
- Increased permaculture as cultivars will encourage more trees to be planted as their quality is known reducing risk to the farmer.
This project will result in new knowledge that will allow growers of chestnut trees to readily improve productivity, by cloning their own trees, replanting, and sharing genetics for the benefit of the whole region. Layering is a tool to help increase orchard yield per acre for every crop year using superior trees. A superior tree we define as an annually high producing hardy tree with good disease resistance. Currently chestnut propagation regionally is either through grafting or seedlings and both are far from ideal. Layering to date has been met with limited results and needs improving for our regional genetics to propagate superior trees and improve sustainability for the whole region.
Chestnut grafts have very high graft failure rate8 and are expensive. Graft failure has been reported to be as high as 100% after five years by the late Dr. Fulbright at Michigan State University. Grafting has been the only option regionally to get the genetics of superior trees. As many small and not so small orchards up to 12 acres7 are being planted with seedlings and are often double planted. As the double planted seedling trees crowed each other, half of the least productive trees are removed in years 5 through 10. It is from these more productive seedlings trees the superior trees can be layered. As grafted trees do not have roots from the original mother trees (a.k.a. “ortet”), their suckers are worthless to layer. One way to have the original genetics of grafted trees on their own roots is to tissue culture9 them and it is a highly skilled effort requiring expensive laboratory equipment to do so reliably. The only other method is to root cuttings of juvenile branches and this is a somewhat complex method with high failure rate10 if not done with very controlled atmosphere and is outside most grower’s skill set and will be a propagation method to be studied in the future. It is the newly planted seedling orchards that we will find superior trees to clone by the average grower without a high skill set or expensive equipment via layering. Layered clones have the maturity of the mother tree and will start producing nuts by the second or third year.
Most young chestnut trees will send up many suckers that could be cloned. As the tree matures and is finally identified as a tree worth being cloned it produces only a few to no suckers. More mature trees can be encouraged to grow suckers with a good pruning and if necessary, by coppicing (a.k.a. cutting down) the tree to be cloned. Once cloned the young trees are then a very good source of suckers for layering. Should the mother tree be coppiced one of the suckers can be allowed to grow and we have witnessed them growing six to eight feet per year for the first couple of years as the roots have a lot of energy to send upwards.
Layering requires that the mother tree sends up suckers at the base of the tree that then can be encouraged to grow their own roots with the application of the correct amount of an auxin hormone and the one that is often used is Indole-3-butyric acid (IBA) diluted in water, powder or oil that best works for the tree’s genetics. Often secondary ingredients are included such as 1-Naphthaleneacetic acid (NAA), vitamins and other adjuncts by companies that make rooting compounds. It is primary to first understand what hormone concentration and what is used to dilute it, (water, alcohol or oil) to promote rooting before the secondary ingredients are tried to further rooting percentages. We seek to find the basic ingredients needed to promote rooting for as many trees as possible in this study. We are not trying to find the ideal treatment for a given tree. The idealized treatments can be found at a much later time once we have a few clones to work with.
In year one we intend to find the basic ingredients needed to root suckers for a good percentage of trees. Once a narrow range of IBA hormone is found then secondary treatment can be researched to see if further improvements in rooting can be made. The second year will include NAA hormone and vitamins known to help other woody species root. The result of this effort will allow growers of chestnut trees to clone their own trees to replant and share their genetics for the benefit of the whole region.
Dawn and Jeff Zarnowski, owners of Z’s Nutty Ridge - NY’s first commercial hazelnut orchard and nut tree nursery, have been growing nut trees since 1992. After moving to a hilltop in Cortland NY, where the hill sides were being reforested post agricultural abandonment, they began to search for trees that would benefit both nature and humans. In their search they identified hazelnuts and chestnuts , both native to the landscape, as ideal and have since propagated the orchard with hazelnut cultivars using layering and are now ramping tissue culture. (Z’s Nutty Ridge nursery LLC was incorporated in 2011).
Both Jeff and Dawn are members of The American Chestnut Foundation (NYTACF) and the Northern Nut Growers Association (NYNGA). In addition, Dawn is a board member of the NYNGA, and Jeff is a director of the recently formed NYTCA Cooperative. Jeff also works part-time for SUNY Environmental Science and Forestry as their Chestnut Green House and Field Manager. Together, they published a book titled, “How to Grow Hazelnuts and Chestnuts”.
Z’s Nutty Ridge currently has one full time and one part-time employees, and a third employee will join the farm in 2024. The farm is 115 acres with two orchards that have deer exclusion fence on 15 acres with 12 acres planted with nut trees and room to expand. It is a fully equipped tree nursery, and they sell trees online and through word-of-mouth. There are currently three greenhouses.
Z’s Nutty Ridge is in a unique position to study layering chestnuts. Their skills, knowledge, and experience, including their networked resources, Technical Advisor, and Key Cooperators should allow for maximum return.
Cooperators
- - Technical Advisor (Educator)
- - Technical Advisor (Researcher)
- - Technical Advisor (Researcher)
Research
As we do have a varied gene pool due to the seedling genetics, there will variability of rooting for a given treatment. Ideally, we would have clones to work with to find the best hormone treatment for that cultivar, that will produce the highest percentage of rooting and the greatest number of roots. As explained previously we need to find the best basic hormone treatment needed to establish clones first. Our goal is to find the treatment that will provide some clones for a good portion of the trees layered. Therefore, we need a minimum of two years for the project and time to disseminate the resulting information. The first year we test a wide range of hormone treatments and collect the resulting data. The following year we re-layer the same trees with a narrow range of hormone concentrations based on the previous findings with adjuncts that may further increase rooting percentage.
Stool bed layering involves:
- Eliminate previous years suckers from the base of the trees by end of March/April. Selecting only trees that have more than six suckers removed.
- Mound chestnut with compatible mulch around the base of the mother tree a minimum of six inches. The mulch allows for etiolation of the sucker shoots where the hormone will be applied.
- Early June through to September apply hormone dissolved in water, alcohol or oil to etiolated suckers and tie off below the hormone. The etiolated stems are scarified prior to applying hormone. New in 2023', monitor shoot growth to slow prior to applying hormone treatments.
- The following late fall/early spring pull away mulch and cut sucker from the mother tree and gather data that includes number of suckers that have roots, the number of primary and secondary roots and length of each root.
Study Year One
Our research will involve one variable for the first year and that will be the concentration of Indole Butyric Acid (IBA) using Vaseline using the above method of stool bed layering and another secondary variable of when to layer.
The number of suckers that sprout from the base of the mother tree can range from zero to over twenty. Only those trees with at least six suckers at the time of layering will be used. As we want as much data as possible to encompass as much genetic variability as possible so we will want to layer as many trees as is practicable. We know a portion of the trees we mulch will not generate the minimum number of suckers desired. Since we know from the number of suckers cut off in March how likely we are to get six or more suckers and will select mother trees accordingly. Therefore, we will initially put compost on no less than 50 trees. This will most likely generate more than 40 trees with more than 6 suckers and definitely more than 20 trees with six or more suckers. This is based on past experience of removing suckers from our chestnut trees. Six suckers will allow for the negative control, and the following concentrations of Indole Butyric Acid (IBA): 1000, 2000, 4000, 6000, and 8000 parts per million(ppm). If there are additional suckers per tree we can then add in more concentrations of IBA or repetition of the above concentrations. Final decisions on the extra sucker treatment, will be based on the number of extra suckers available and what is found from relevant paper and network research on layering chestnuts.
We will ask that the grower provide any genetic background information on the mother tree and what the health, disease history, age and productivity of the trees are. We will also soil sample and do a leaf analysis of the mother trees as the nuts are half filled in the summer, to see if there is any correlation between tree health, age and the amount of suckers at the base.
Ideally the trees layered will have many suckers to allow for a control and multiple hormone treatments. The control will be a layered sucker that is treated the same as all other suckers but will not have any hormone applied just Vaseline. Each sucker will have a different hormone concentration. The number of suckers on a given tree will determine the number of treatments given. What concentrations are applied to trees with fewer suckers will be determined as we finalize the experimental design based on any previous work we can gather during the first three months of the research.
All data will be analyzed using ANOVA by Dakota Matthews/ Patricia Fernandes/Jeff Zarnowski. It is often at the end of the first round of measurements that another relevant data point is discovered and any findings to this end that are found will be reviewed for year two.
A report with all data collected will included in our project update.
Study Year Two
After reviewing the data from 2022’, we wanted to bracket the previously determined best IBA hormone concentration to further improve rooting overall. The control will be the same as in year one. We will redo the range of IBA hormone of year 1 and in addition, we will be able to add an additional hormone Naphthaleneacetic acid (NAA) and limited additions to further increase rooting percentages with the extra suckers. Except for the planned changes, all the exact same steps done in year one will be repeated on the same trees to minimize any unforeseen variables. This plan also allows for less suckers per tree on the following year as the number of suckers usually declines with the age of the mother tree. The number of suckers layered will be increased to cover a wider treatment of hormones. Those additional suckers will have the hormone concentrations repeated with NAA or vitamin additions. The additions will be decided based on research in year one and included in the report for year 1.
At the end of year two the rooting percentages per treatment, the number of primary and secondary roots and root length will be remeasured as was done in year one. All data will be analyzed using ANOVA by Dakota Matthews/Patricia Fernandes/Jeff Zarnowski unless there is an obvious choice for hormone treatment.
A report with all data collected will be included in our project update.
Summary of 2023 results
Based on the results from 2022 and lessons learned, we modified our approach to layer chestnuts in 2023’, with good results and a clear success in finding a hormone treatment that will yield rooted suckers for most chestnut genetics. The changes for 2023' efforts were two-fold, one change was to monitor sucker growth and the second change was to repeat first year hormone treatments and include adjuncts as well.
Monitoring growth is the result of having been armed with knowing that layering chestnuts too early causes the suckers to choke then die as happened in 2021’ and then finding a reference by Takao Liiv that growth of the sucker must be monitored for proper timing of when to layer. We did periodic measurements of sucker growth. The purpose of monitoring sucker growth is to hold off layering until sucker growth slows and yet layer the suckers with a minimum of one months growing season left and ideally two months growing season to increase root yield and quality.
A repeat of the first years hormone treatments done in 2022’ and adding additional treatments with adjuncts were needed to make up for the terrible growing season, due the lack of rain developing near zero rooted suckers. In order to accomplish this added effort, we needed to expand the number of suckers layered to 300. The expansion of suckers layered was required as we had essentially lost a years worth of layering effort in 2022’ and had to combine two years worth of layering in 2023’ Efforts in 2022’ were not totally lost as the farmer and technical staff team combined efforts yielded knowledge for very good results in 2023’. Technical reference papers and European practitioner research combined with previous years layering efforts solved the unknown problem of when to layer, as the most important item to successfully layer chestnuts.
The research found when to layer, and which hormone applied produced: the most rooted suckers, the highest quality of roots and generated the most rooted suckers across a wide array of genetics. That hormone treatment is 4000ppm IBA-K dissolved in water and blended with Vaseline.
Hormone Treatments Applied
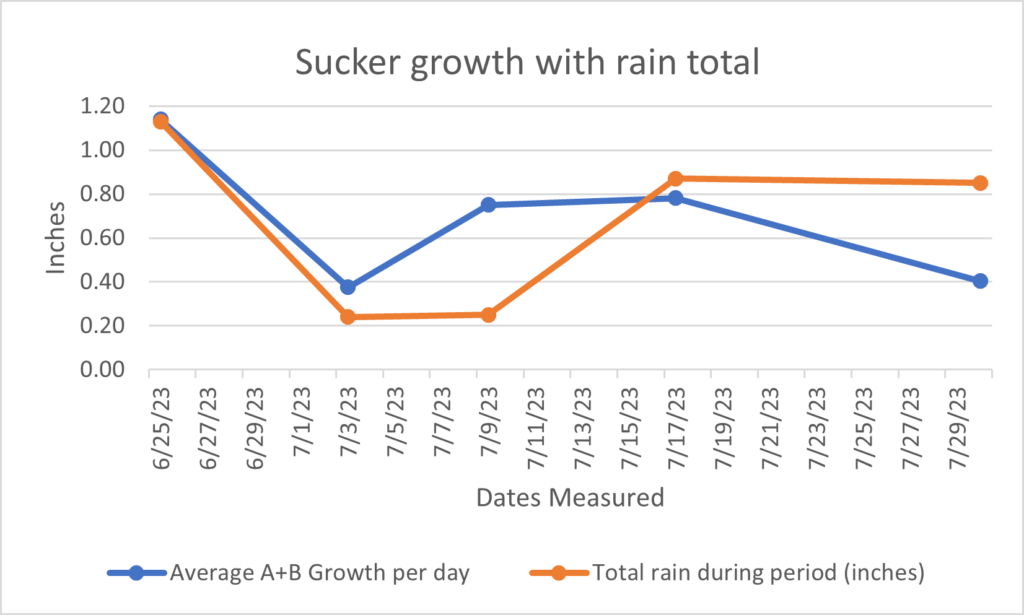
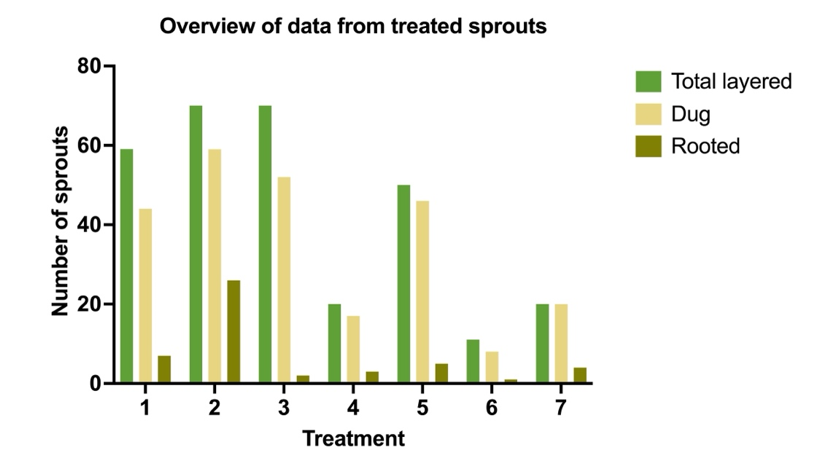
Having monitored the growth of shoots at Z's Nutty Ridge it appeared that shoot growth was slowing at the beginning of August. Chestnut suckers were layered from July 22nd through August 11th, 2023'. Various treatments of hormones were tried including: 4000ppm IBA, 4000ppm IBA-K, 4000ppm IBA-K + 4000ppm NAA, 8000ppm IBA, Cinnamon, Cinnamon with honey, and Pedro Urquijo formula containing, approximately 4000 IBA + 4000ppm NAA, 2,4-D, honey, glycerin and baby powder as shown in Table #4. The clear winner of hormone formulations was 4000ppm IBA-K dissolved in water and then well blended into Vaseline. IBA-K is a water solvable form of IBA. The 4000ppm IBA-K hormone in Vaseline yielded 44.07% rooted suckers with 73% of the suckers having root quality being "okay" or "well" rooted for the highest quality of roots as well. This treatment was tested on a total of 10 different trees, with 70 suckers treated and had 59 suckers survive and 26 suckers rooting. More information on the Pedro Urquijo formula and how we were led to it is further described by outreach from Technical Advisor Patricia Fernandez below. Hormone treatments were tracked by the color of ribbon tied around the treated trees.
When to Layer Chestnut Suckers
Layering procedure was the same as 2022' but sucker growth was monitored. A tall stake was made and had inch markings on it so the tallest two suckers of a tree could be measured periodically. The tallest suckers were chosen as they were the first to appear and were easiest to identify from week to week. Suckers emerge from the base of the tree continuously over the spring and summer months. Some suckers sprout after layering.

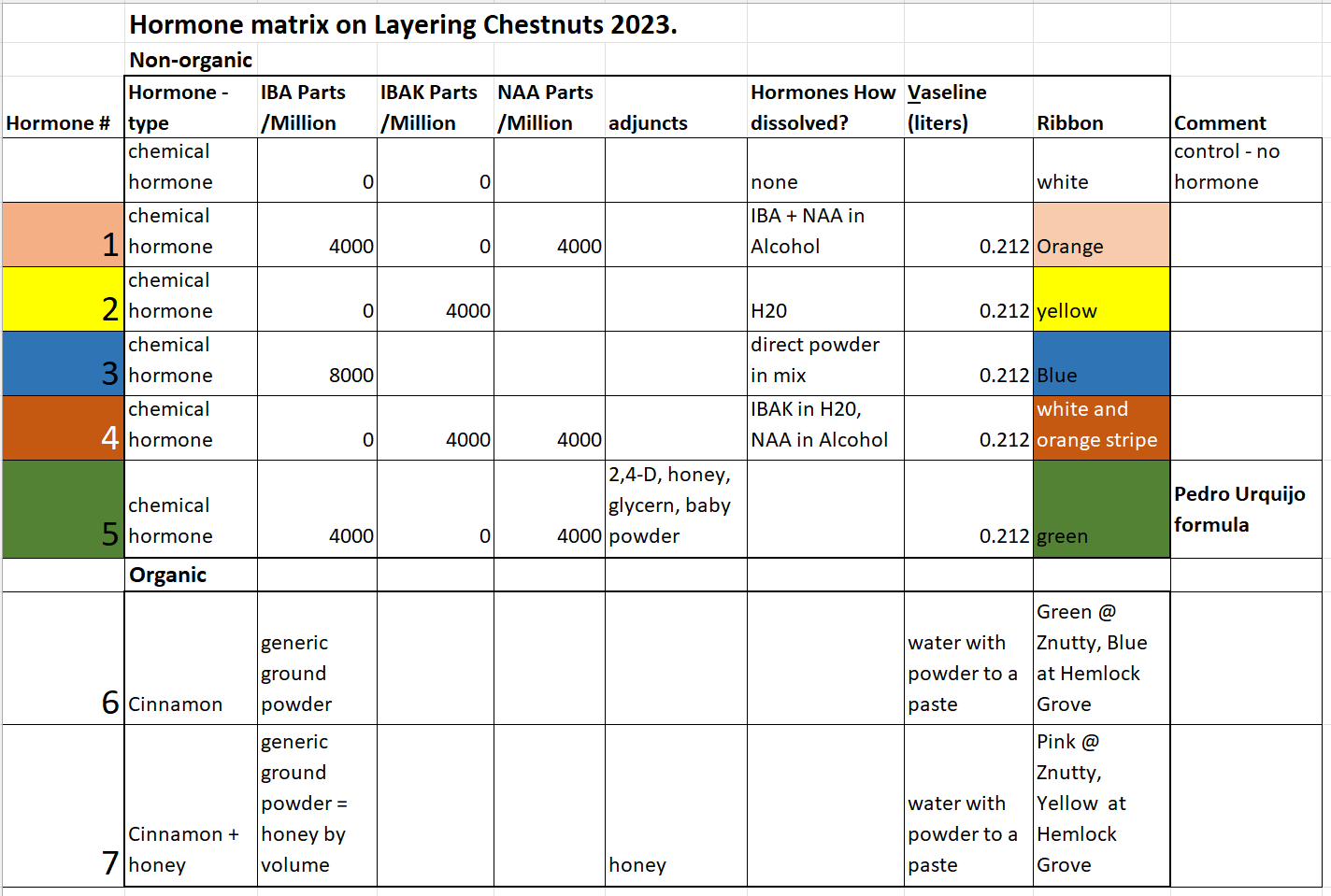
Layering Chestnut trees in 2023 was initially looking to be a disaster with a hard 24 degree Fahrenheit frost on May 18th. Spring came early and all fruit and nut trees budded out early and became susceptible to a hard frost. The May 18th frost burnt all or most all leaves and flowers on all fruit and nut trees in the surrounding area except for hazelnuts. The sucker or two that wasn't layered per tree in 2022' was burnt and most didn't regrow. New suckers took over delaying the start of the seasons growth by no less than two weeks even with an early start. Weather events only further strengthen the need to monitor the sucker growth.

We all know rain has a direct relationship on plant growth. To help understand the relationship between sucker growth and rain we charted total rain and average growth per day between measurements. We needed to understand was sucker growth due to recent rains or a natural slowing of growth. The average growth of suckers per day somewhat matches rain for the period until the last measurement on July 30th. We then decided we needed to start layering chestnuts in earnest. In reviewing the data as early as July 17 an increase in rain brought about little increase of growth and it might have been possible to layer even as early as July 17th. We know that layering too soon causes death to the suckers and we expect some suckers to die via choking prior to prior to stimulated root growth and the earlier we layer the more suckers will die. Even with the majority of suckers were layered on August 6th and 9th, we still had 18% or 56 suckers dead or missing when dug. From previous years efforts we learned that suckers that die often fall over and the small ones go unnoticed in the thick leaf mulch. We do have some suckers that die from rodent pressure when bark is stripped from the stem.
Timing for when to layer needs to be simplified for the average grower. Having a grower monitor suckers growth and rain fall in the busy season of the year may prove to be too much. The average first date of frost in Cortland, New York is October 1st according to Cornell Cooperative Extension. According to Takao Li, of Hyogo Agricultural University, we should monitor shoot growth until growth slows and still have at least one months growth and ideally two months of growth left in the season. This means if we layer two months before October 1st we need to layer on or around August 1st. At Z's Nutty Farm Location we have approximately 2030 base 50 Growing Degree Days (GDD) per year and if we layer automatically on August 1st we still have on average 806 GDD or 39.7% of the growing season left.
Layering Results
All hormone treatments and results identify one hormone treatment that stands above all for number of roots and quality of roots. From Table # 5 we see that hormone treatment # 2(yellow ribbon) was superior by far with 37% of the shoots layered rooted with the highest grade for the root quality of 8.77. Meaning root quality was graded by a weighted system. The system for grading was weighted by root quality with the lowest quality root being rated a "1" where there were only one or two roots less than 8 inches. A root with a weighted grading of "2" had 3 to 6 roots totaling less than 16 inches and a grade "3" had 6 to 12 shoots with many side shoots. A grade of "4" had greater than 12 shoots and too many side roots to count. Each rooted layer was graded by quality and had the quality multiplier applied to all the rooted layers and averaged per treatment.
Each hormone treatment had a color and a number assigned to it. To understand what the hormone treatment number was we refer to Table # 4. On Table #4 we see hormone treatment number 2 was a yellow ribbon on the layers and was 4000 IBA-K dissolved in water and then blended into Vaseline. The complex Pedro Urquijo formula with tree different hormones with adjuncts including glycine and honey didn't do well with 10% of suckers rooted and a poor root quality rating of 1.2. The second best rooting percentage was for the organic treatment using cinnamon and honey in equal parts and diluted with enough water to make a paste. The tree is now called Uni-Graft as Technical Advisor Brian Caldwell believes that this tree allows most scion wood to be grafted without graft failure, unlike most rootstock for chestnuts. The rooting results were 4 trees out of 7 rooted with a root quality weighted grade of 4.0. This tree is the same tree that had 7 suckers root out of 20 using just cinnamon in 2022'. It appears that the Uni-Graft is one of the easier trees to root suckers as the control rooted. As a reminder the control per tree was a sucker that had a nylon zip tie put on it without hormone as a statistical control. The control sucker on Uni-graft tree had only one root that was about 4 inches long without any side shoots. No other control produced roots and the Uni-graft is the lone exception.
Data Analysis
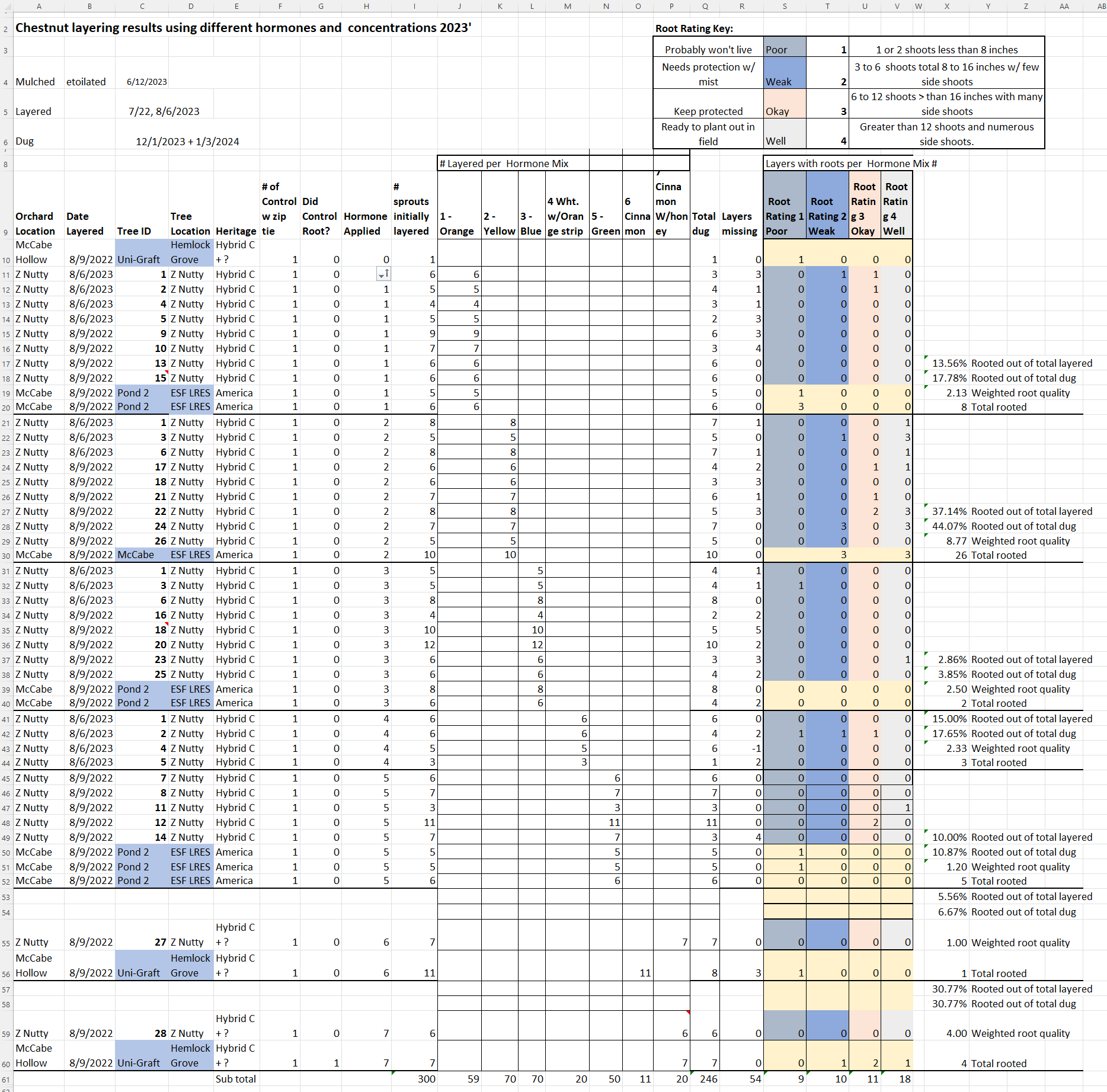
A summary table of chestnut layering results by hormone treatment is shown in table # 6. One can see that Treatment # 2 using 4000ppm IBA-K was by a large margin the best treatment to produce rooted suckers. With the assistance of Patrícia Fernandes, Ph.D. and Dakota Mathews using ANOVA analysis, using information from Table # 5, the following data was ascertained.
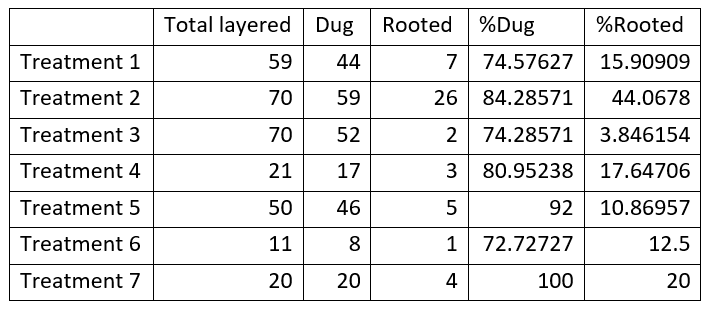
A plot using the summary table #6 above illustrates how treatment #2 rooted more suckers than any other treatment.
If plot #2 is simplified further to better illustrate the rooting percentages by hormone treatment we have plot #3.
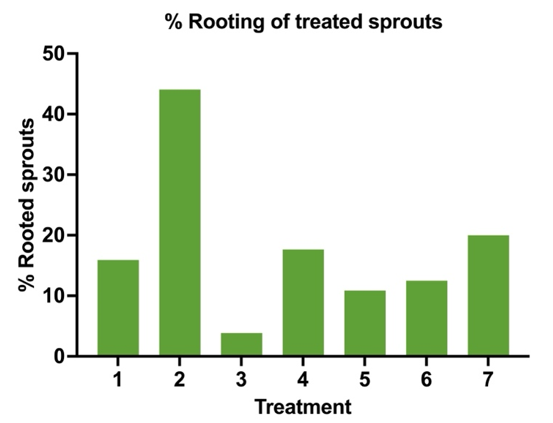
Now that we know Treatment # 2 using IBA-K in Vaseline is more than 2.5 times better rooting than the next best treatment, we now need to understand the rooting quality of the treatments. Rooting quality will indicate how well the rooting suckers (a.k.a. sprouts) will survive being cut away from the mother tree.
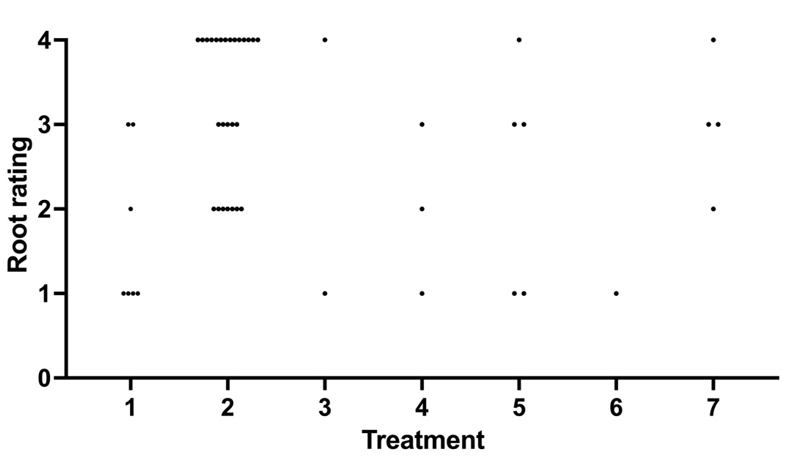
As previously described root quality was given a weighted score based on the total length of roots and number of roots. Plot # 4 illustrates the quality of each sucker for a given treatment. Each sucker was given a point by root grade by treatment with Treatment # 2 also have the highest quality of roots. Treatment # 2 had a weighted root quality grade of 8.77 and more than twice the next highest quality grade from Treatment # 7.
One of the objectives of the study is to find the best hormone treatment that allows rooting across the most genetics possible. As orchard type chestnut trees have varied hybridized genetics from America, Europe, China, Korea and Japan. Treatments 1,2,3,4,5 are the only groups with more than 3 different layered trees and, therefore, the only ones that can be compared statistically. The dug and rooting percentages were calculated for each tree and considered one sample. Dug and rooted sprouts calculations were performed taking into account the following # of samples: Treatments # 1, # 2 and # 3 have 10 samples, Treatment # 4 has 4 samples and Treatment # 5 has 8 samples.
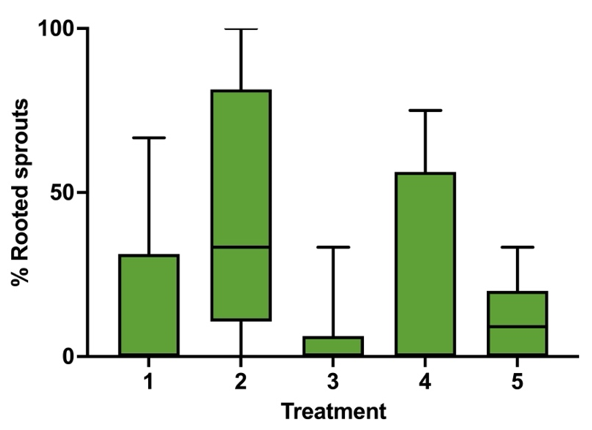
In plot # 5 The Box plot summarizing rooting data. with whiskers show minimum to maximum and Line shows the median. A Kruskal-Wallis test (Non-parametric ANOVA analysis). The treatments were not statistically different. P value=0.1040

An example of rooting quality for treatment #2 that was identified in the field by a yellow ribbon is shown in Picture #9.
European outreach by Patricia Fernandez Technical Advisor
Search for what hormones and application techniques used by practitioners in Europe found that Vaseline is the most used to dilute the hormone and the below reference that led us to the Pedro Urquijo formula as posted in a book titled, "CHESTNUT FARMING GUIDE FOR NUT PRODUCTION”, J. Fernández-López et al. (2014). Guía de cultivo do castiñeiro para a produción de castaña. Xunta de Galicia. Consellería do Medio Rural e do Mar. ISBN: 978-84-453-5160-4. This book and referenced hormone formula were found by Patricia Fernandez after discussions with the books editor Josefa Fernández López.
Josefa Fernández López stated, "con estelink podes descargar o libro "Guía de cultivo do castiñeiro para a produción de castaña" Na páxina 47 está escrita a descrición do método de "layering".Eiquí chamámoslle aporcado e vos amontoa. Na páxina 58 hai dúas fórmulas para a preparación das auxinas. A primeira era a que se empregaba en Lourizán. O punto 2 ten un erro pois a cantidade de alcohol etílico ten que ser inferior ó escrito. Probablemente 100 ml de alcohol sexan suficientes. Si pos 1 litro entón o alcohol non seda evaporado. Si precisas algo máis eiquí estou. Saúdos e sorte para o produtor americano. Fina”.
Translation: "With this link you can download the book "Chestnut Cultivation Guide for the production of chestnut" On page 47 is written the description of the method "Layering." Here we call you apostrophe, and I'll pile you up. On page 58 there is Two formulas for the preparation of auxins. The first was the one that was He used it in Louie. Point 2 has an error because the amount of alcohol Ethyl has to be lower than written. Probably 100 ml of alcohol Be enough. If you post 1 liter then the non-silk alcohol evaporates. If you need anything else here I am. Cheers and luck to the producer American. Fine"
Chapter: Josefa Fernández López, Ma Eugenia Miranda Fontaiña, Santiago Ramos Rodríguez, Adrián López Villamor, Immaculada López Muñoz (2014). Técnicas de produción de plantas. In: Guía de cultivo do castiñeiro para a produción de castaña. Xunta de Galicia. Consellería do Medio Rural e do Mar. J. Fernández-López. pp: 43-75. Translation summary of pages 46 to 49 below
Remove leaves in the first 20 cm of each shoot
- Place a wire on the base of each shoot but make sure to leave some shoots unwired, these will ensure the stump’s nutrient uptake. Using a manual stapler simplifies this task.
- Apply rooting auxins in the leafless area after doing two to three wounds vertical to the cambium, in the shoots’ base. The auxins are applied in Vaseline (4 mg/g IBA or combined 4 mg/g IBA with 4 mg/g NAA) or following a more complex formula developed by Pedro Urquijo (in detail below).
- Layer the area treated with auxins with soil.
The roots develop in the following months and the shoots are ready by the end of Fall, when they lose their leaves. There is a great variation in root development between genotypes.
Pedro Urquijo formula
List of ingredients:
- NAA: 60 g
- IBA: 70 g
- 2,4-D: 4 g
- Baby powder (it translates from talco ventilado): 7 Kg
- Honey: 5 kg
- Glycerin 30° neutral and very pure: 340 cc
- Water: 3.060 cc
- Methyl alcohol: 100 cc
- Ethyl alcohol: 4 liters
These ingredients should be enough for 100,000 shoots.
- Dilute 2,4 D in the methyl alcohol.
- Add NAA and IBA to 4 liters of ethyl alcohol and mix well (make sure to dilute it properly).
- Mix the two solutions from the previous steps and add the baby powder. Stir for 10 minutes in order to achieve maximum homogeneity.
- Pour the paste on a soft and polished surface (about 2-3 m2) in a thin layer (not more than 5 mm thick) to dry quickly (not more than 48h).
- Once the paste is dry, turn it into powder by crushing it with a mortar. It can be preserved in this state for a long time without losing viability.
- To obtain the final paste, mix the powder from the previous step with 5kg of honey and 3400 cc of water and glycerin 10% (3060 cc water and 340 cc glycerin). Once the mixture is done, use a blender to prevent clump formation.
- The final paste can be packed in compressible tubes, ready to use.
- Once the paste is packed, store it in the fridge until its used.
2022 Layering Efforts
In 2022' the first year of research, trees were layered on June 25th, July 20th, July 27th and September 3rd 2022, at Hemlock Grove, SUNY ESF, Z’s Nutty Ridge and Finger Lakes Nut Farm respectively. In total, for all four farms, 24 trees were layered consisting of 232 suckers. Only one tree generated rooted suckers in 2022. We believe the lack of rooting is due to dry weather. On the tree that did root, it didn’t receive any of the IBA treatments as it was at Hemlock Grove, an organic farm. The organic treatments were cinnamon paste and seaweed paste being substituted for Indole 3-Butyric Acid (IBA) dissolved in Vaseline. This tree rooted 10 suckers out of a total of 45 suckers layered. Of the 20 suckers that were treated with cinnamon paste, seven had produced roots for a 35% rooting. Although totally unexpected, 35% rooting we would consider a decent first year success if that were the average for all trees.
IBA concentrations studied in 2022' were 1000ppm, 2000ppm, 4000ppm, and 8000ppm and one hormone addition that was 4000ppm + 4000ppm 1-Naphthaleneacetic acid (NAA). NAA is often used in conjunction with rooting hormones on hardwood propagation and was suggested in references[i],[ii]. The NAA addition was a result of having more suckers to layer than what we had originally estimated. Our method and hormone concentration brackets were backed by technical papers and current practices on European (Castanea sativa) X Japanese (Castanea crenata) trees in Europe and was selected to be a good range to find what hormone concentration to use on our cold hardy regional genetics. Majority of our regional genetics are mainly Chinese hybrids that are crossed with European, Japanese and American chestnuts for nut quality, hardiness and disease resistance.
Of course, we are at the mercy of nature and the summer of 2022' was the driest since 2016’. At Z’s Nutty Ridge we have two trees that are six years old planted near the layered trees drop their leaves due to drought. We also had whole branches on hazelnut trees turn brown not 100 feet away from the layered trees. We noticed many of the smaller caliber suckers were dead when dug and wondered if the drought had been an influence. One would believe that a very dry summer would have the least impact on suckers from a coppiced tree, where the water needs would have been reduced for the root systems, depending on the number of suckers and their growth. In technical reference material the shorter suckers often root with a higher percentage[iii] lending to our belief the severe dry spell we had this summer hindered our ability to generate any rooted suckers except for one tree. The one tree was at Hemlock Grove farm and had little moisture problems being at the base of a hill and very near the home of Brian Caldwell.
The lack of rooted suckers doesn’t allow us to compare statistics of rooting percentages and quality between treatments for the year.
Since we didn’t have any suckers root with IBA treatments, our timing of when we layered didn’t provide any information. A review from one technical paper detailed below[iv] illuminated when to layer. Going forward we now know to monitor sucker growth, to know when layer and to water the suckers during dry periods.
Background of Previous data collected:
This study was initiated by chestnuts and hazelnuts being planted throughout the Northeast. Hazelnuts for the most part will root their suckers if given hormones to enhance root development and is routine, well known procedure. Hazelnut suckers can be layered as early as late June and as late as the first week in September. Applying similar techniques to chestnuts in 2021’ gave very little results and the search was on to gather previous and current practical knowledge on the subject and is the purpose of this study.
Starting in the spring of 2021’ chestnut trees were mulched at Zs Nutty Ridge and at State University of New York at Environmental Science and Forestry (SUNY ESF) to produce etiolated shoots for layering. The experiment involved layering three trees at Z’s Nutty Ridge using the same layering method as hazelnuts using the same hormone made from 2000ppm of IBA dissolved in ethyl alcohol and diluted to proper concentration with water on July 7. There were five trees at SUNY ESF layered using 4000ppm, 5000ppm or 8000ppm IBA dissolved in ethyl alcohol and diluted to proper concentration with Vaseline (Petroleum jelly) on August 5th. Of the 29 suckers layered at Z’s Nutty Ridge all had died and were snapping at the nylon zip tie placement. It appeared that the suckers had quickly outgrown the zip ties and choked. Of the 31 suckers layered at SUNY ESF, four suckers did have some roots and three survived being potted. The suckers that produced roots were three trees from the 5000ppm and one tree from the 4000ppm concentration.
Conclusions from the 2021 efforts at this point were few. Water diluted hormones generated zero roots when layered during the first week of July. It appeared we layered too soon at Z’s Nutty Ridge, as some suckers can grow over four feet from an uncoppiced tree and a coppiced tree can have suckers grow 7 feet or more. We don’t believe or suspect that the hormone concentration was too high as this is considered on the weaker side for woody perennial layering. The SUNY ESF trees had 13% rooting using double or more concentration of IBA than what was done at Z’s Nutty Ridge and none of the medium or larger suckers were dead. This information will be used in conjunction 2022’ to help understand our efforts to layer chestnut trees. Also, the differences in the above treatments and timing could be due to the genetic variances between Chinese hybrids (Castanea mollissima x hybrid) versus pure American chestnut (Castanea dentata) and are part of the current efforts to understand.
Efforts layering trees in 2022’
The goal is to find a hormone concentration that will work for most regionally adapted, healthy productive chestnut hybrids.
The main effort of this study is to layer chestnut trees involved with one main variable and that was the concentration of IBA. A secondary one was bracketing the timing of the layering as was learned from 2021 effort during the spring of 2022. The common method for layering chestnuts in Europe was found to be documented as a result of this grant, from the technical paper searches and direct contact with European practitioners.
Generating suckers is fairly simple, duplicating what is done for hazelnuts, coppice the desired tree (if mature) early spring and mulch the stump to encourage etiolated stems. As discussed during the proposal, adjustments will be made as items are discovered. Timing of layering was staged from lessons learned from the initial layering attempt the previous year (2021). All suckers were layered except a few stems that would continue to feed the root system for next years follow on layering experiments.
The steps of layering suckers are as follows: 1. Tie off the base with a wire or similar constrictive device like nylon zip ties or “hog rings”, 2. Wound the stem above the “Tie” and apply rooting hormone, 3. Back fill the etiolated stems to encourage roots to form, 4. Cut suckers after plants have senesced for the season and before sprouting the following spring. Although we have found that suckers dug in the spring have heavier caliper roots for hazelnuts, this isn’t part of the study. However, we will have comparable data for hazelnuts once they are dug in the spring of 2023’.
Items gleaned from search for technical papers and practitioners of chestnut agriculture in Europe by Dakota Matthews with input from Patrícia Fernandes PhD, yielded useful information to use in consideration for layering, even if it wasn’t well quantified, that includes:
- Layering process,
- How to mix IBA with Vaseline,
- Eliminate tall suckers from layering,
- Other hormone additions,
- When to layer.
We will discuss each item individually with a new perspective having seen the results from this year’s efforts.
-
Layering process was the most feared item going into the study and having references reinforce using the same technique for hazelnuts as described above was a welcomed relief.
- Mixing dissolved IBA with Vaseline(petroleum jelly) confirmation was another item needed to add confidence to the original presentation that was used to initiate chestnut layering efforts.
- Eliminate tall suckers from layering as they have a lower rooting percentage or won’t root, was discussed in Brian Caldwell’s master thesis and in relevant Japanese paper written by, Dr. Takao Li, Of Hyogo Agricultural University, Accepted July 5, 1959 , “Propagation of chestnut by layer.” Caldwell’s thesis had zero shoots root over 30cm in one year and had the same layered trees have 50% of the shoots root that were over 41cm the second year. Dr. Takao Li had less then 2% of the shoots produce roots above 41cm. As Brian Caldwell did have 50% of the suckers root over 41cm in one year out of two while, Dr. Li had zero suckers root on one cultivar this wasn’t enough of a study to stop layering taller suckers. Brian Caldwell thought that the suckers in year two had the wounding and hormone application higher from the native soil line by 3 to4 cm may have assisted rooting. When layering we generally put the “Tie” 2 to 4 cm above the native soil line to facilitate cutting the sucker from the mother tree. Also, this study wasn’t to maximize percentage of rooting as we are trying to find the best hormone concentration to have viable rooted suckers on the most trees with a diverse background, so all suckers were rooted.
- Hormone and other adjuncts are secondary to the selected IBA hormone concentration that include:
- 2, 4, 5-trichlorophenoxy-proplonic acid,(2,4,5-Tp)
- 1-Naphthaleneacetic acid (NAA)
- 2,4-Dichlorophenoxyacetic acid (2,4-D)
- Honey,
- Cinnamon
The hormone and adjuncts were found in technical papers, and External Technical References with one exception. Brian Caldwell suggested using cinnamon powder to enhance root growth, so we tried it along with seaweed. Seaweed paste did have two layers root out of 20 layers but the two suckers were among tightly spaced suckers that had cinnamon on them. Listed in the Appendix: External Technical Reference section one hormone recipe contains, IBA, NAA, 2,4-D and honey for European hybrid layering. Also, a honey with cinnamon combination may be of use for organic growers, yet to be proven for chestnuts. Quick search on the internet shows honey, cinnamon and in combination being used to root plants of all types.
- When to Layer suckers is a common concern. As we have now learned, a calendar date proves not to be near as important as how the shoots growth is progressing. As good example learned from layering on July 7, 2021 was too soon as suckers choked at Z’s Nutty Ridge but layering two weeks earlier on June 25, 2022 at Hemlock grove farm generated roots. According to Dr. Li the best time to layer is when the shoots slow down their growth but have two months’ time to root. Li’s study found that there was a 61% increase for trees that were easy to root and a 47% increase in rooting for difficult to root trees when allowing 2 months over 1 month time. One may conclude longer is better but we also know too early the shoots will choke and die. Here in upstate New York that would be layering suckers between beginning of June and before the last week of July for there to be two months before the possibility of frost for a normal year. Going forward at Z’s Nutty Ridge we will monitor sucker growth weekly on a sample number of trees and when the suckers slow down growth between June and July we will then layer the suckers. This form of monitoring suckers maybe be applicable for all types of trees that are layered.
Having layered 32 trees over a two-year period we see that sucker growth can vary wildly. Growth depends on how healthy and the age of the coppiced tree was, along with how many suckers are on the root crown. The tree at Hemlock Grove farm had 45 suckers on it and we were able to layer very early in the season, while the average tree layered in 2022’ had 13.8 suckers. Leason learned is to monitor sucker growth and watch for a slowing and hopefully with two months of the growing season left to maximize rooting.
Layered Chestnut Information 2022
A spread sheet detailing the tree location, heritage, timing and IBA concentration used for the first year study is shown in Table #1. We believed that we should be able to layer at least 20 trees that had six or more suckers in the grant proposal and a total of 24 trees were layered with a total of 232 suckers.
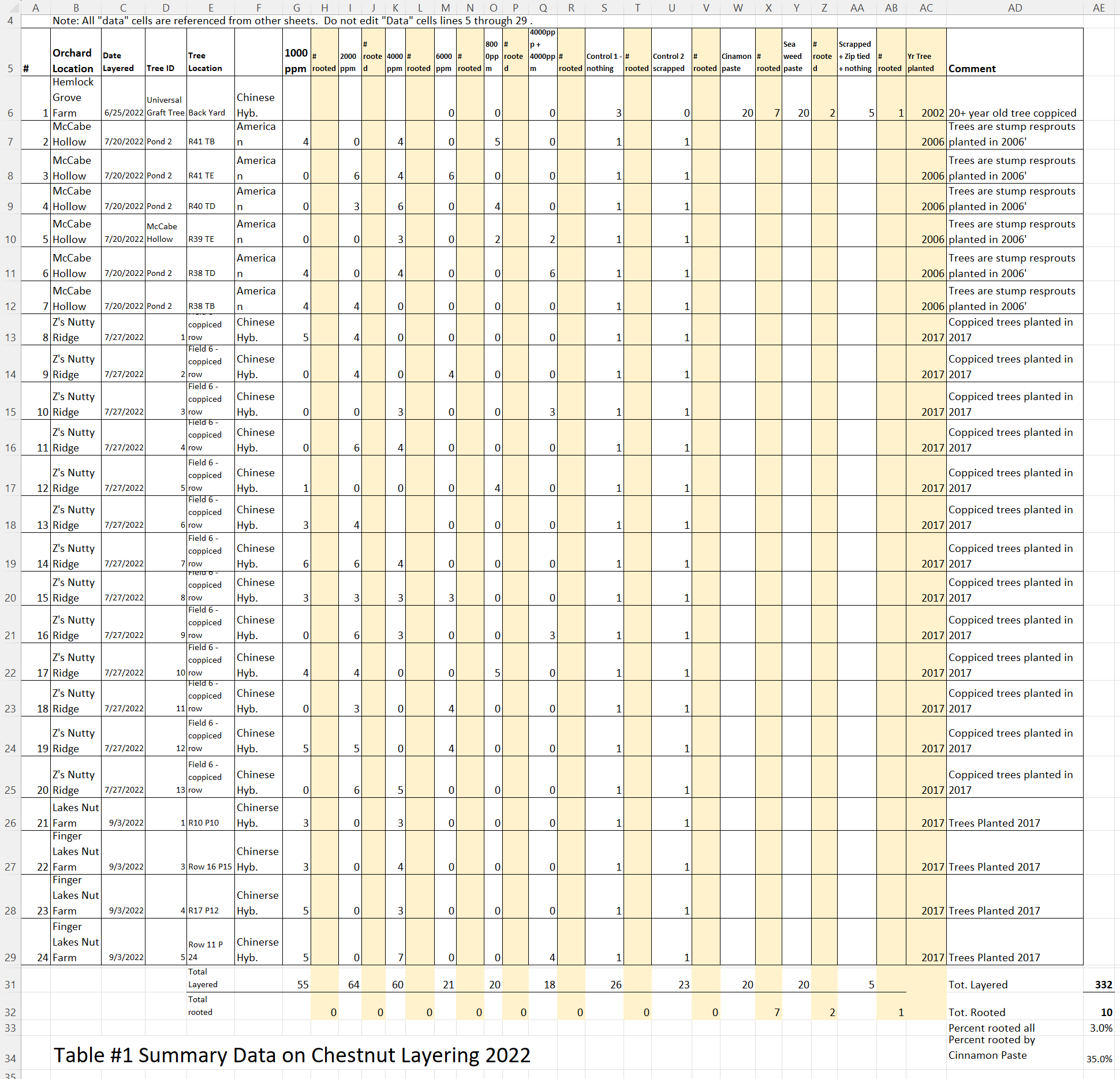
Soil Analysis
Table #2 shows the soil data from the four farm locations and locations within the farm close to layered trees. As there weren’t any rooted layers this year, it is difficult to see if soil has an impact on layering percentage. Chestnuts need a pH of 6.5 or less for good health and growth. Oddly, the only tree that layered in this year’s study was at Hemlock Grove and it had a pH of 6.73.
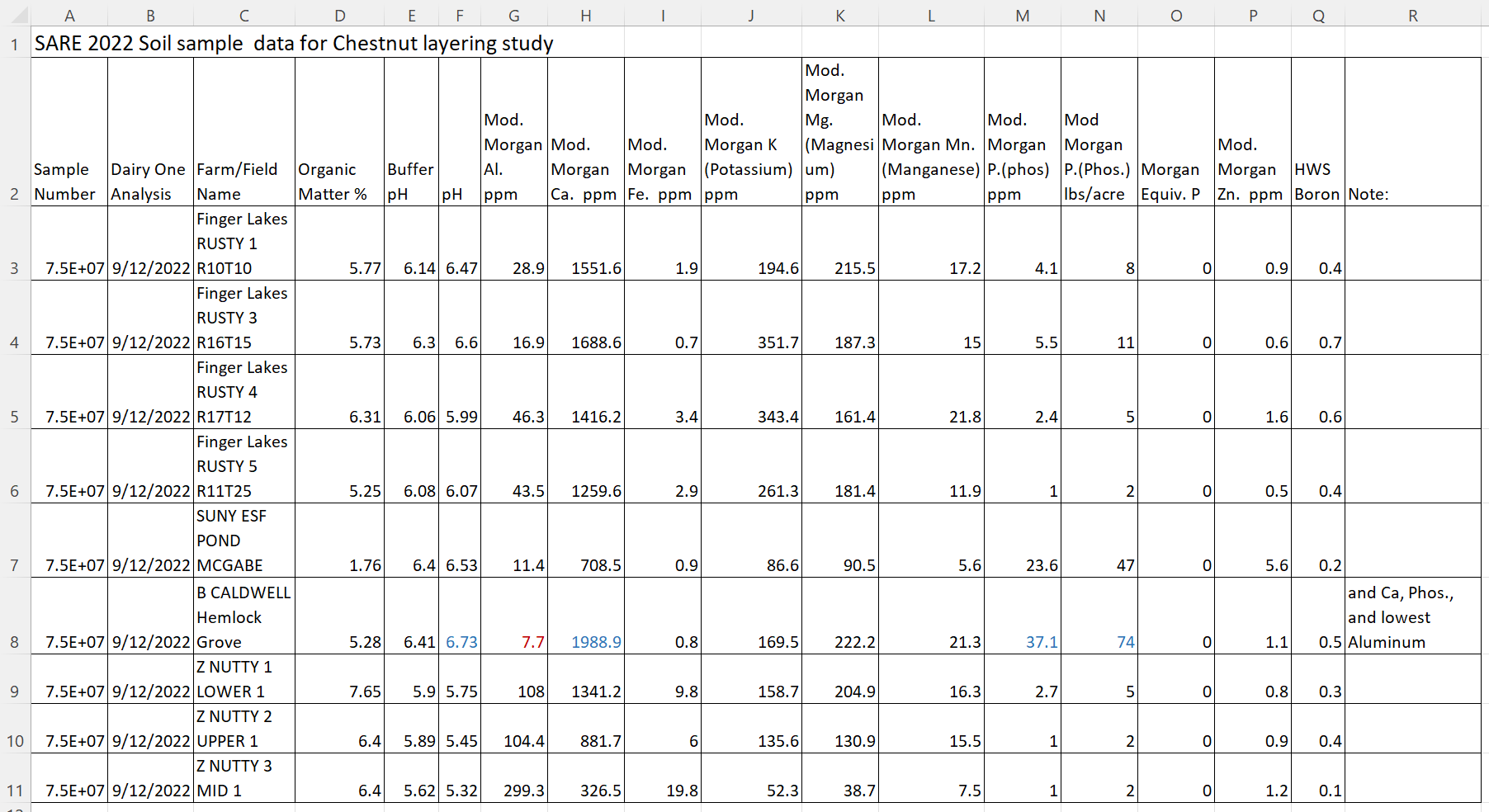
Table #2 Soil Analysis
Leaf Analysis
Table #3 shows the leaf analysis for the four farm locations where the layers were located. Chestnuts need above 50 ppm of Boron for good nut size. Hemlock Grove soil and leaf analysis with be taken this coming year.
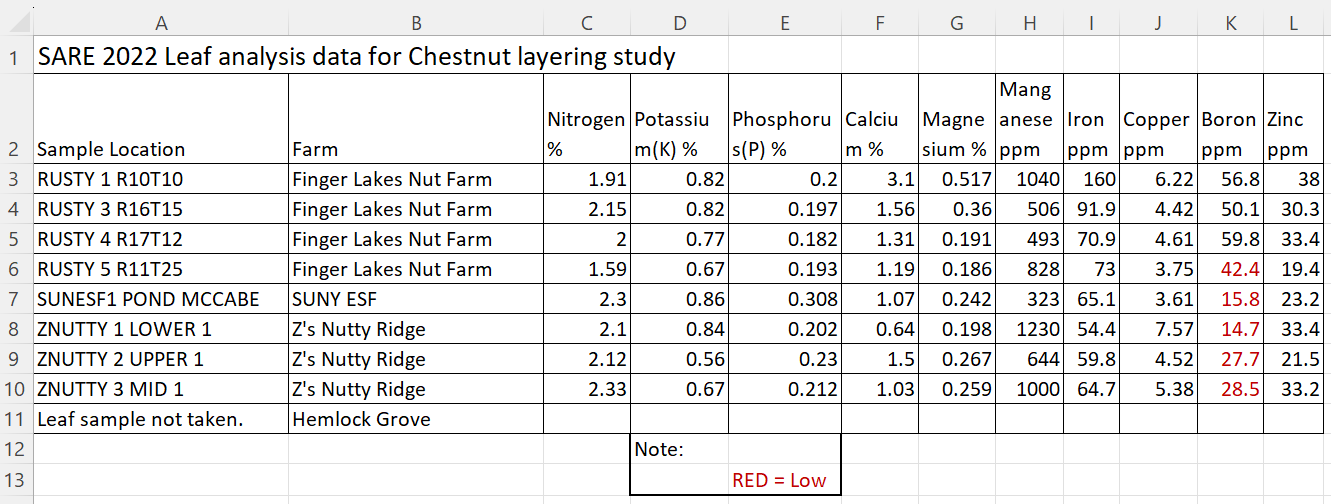
The bottom third of the leaves are stripped from the suckers and is normally taken when nuts are filling on the tree to understand if there is any nutrient deficiencies hindering production. As the tree layered at Hemlock Grove was early In the season the samples weren’t taken, arrangements will be made for 2023’.
Field pictures of layering effort.

The McCabe Hollow plot is at State University of New York Environmental Science and Forestry (SUNY ESF) and notice how in Table #1 that five of the six trees are identical clones, named Pond2 generated from tissue culture. The trees are part of The Chestnut Restoration Project headed by Dr. Powell and Dr. Newhouse. Having 5 clones to work from will allow for high repetition of treatments and allow for various treatments to be studied at the same time. We were able to layer 62 suckers from five cloned trees.


Individual layered stems had colored ribbon tied to stem to keep track of treatment used. Controls were also color coded.


The number 12 in Picture # 5 represents the tree number in Table #1 for Z’s Nutty Ridge. Notice the different color ribbons for the different treatments used on the suckers.

Rooted suckers were potted and put into cold storage for the winter they will be monitored in the spring for survival and growth.
Technical paper search turned up a very relevant paper written by, Takao Li, Of Hyogo Agricultural University, Japan, July 5, 1959 , “Propagation of chestnut by layer.” We appreciate this paper as it is a practical guide on timing, concentration of hormones and Chinese chestnuts trees were part of the study.
A good portion of the translated salient points were given in the summary.
In continuation of the preceding research, layering experiment has been conducted on Japanese and Chinese chestnut trees making use of growth promoting substances in lanolin paste.
- Varieties which were especially hard to root with IBA alone, rooted better with a mixture of IBA and 2, 4, 5-Tp, and with 2, 4, 5-Tp alone. Also by foliar application of 2, 4, 5-Tp to seedlings in layering bed, vigorous rooting was obtained.
- Rooting ability of cultural variety was greater after shoots have stopped growing than during their elongating period.
- The longer the length of shoot of cultural variety, the lower the percentages of rooting.
- Layers which were grafted on vigorous stock required a longer duration in rooting than those grafted on less vigorous stock.
- Concerning the rooting percentages and rooting time, considerable differences were observed among juvenile seedlings and two types of varieties of which one was easy and the other hard to root. Higher rooting percentages were accompanied with longer rooting time.
[i] Josefa Fernández López , 2014 “CHESTNUT FARMING GUIDE FOR CHESTNUT PRODUCTION”, Board of Wales, Ministry of Rural and Marine Affairs, Santiago de Compostel.
[ii] Vielba, J.M.; Vidal, N.; José, M.C.S.; Rico, S.; Sánchez, C. Recent Advances in Adventitious Root Formation in Chestnut. Plants 2020, 9, 1543. https://doi.org/10.3390/plants9111543
[iii] Brian Caldwell, 1986, MS Thesis: “Improved Propagation Methods for Chestnut” Cornell University.
[iv] Takao Li, Hyogo Agricultural University, July 5, 1959 , “Propagation of chestnut by layer.”
Conclusions 2023
This effort has established a chestnut layering method that will give most hybrid chestnuts grown the ability to be cloned. The mother tree (a.k.a. ortet) cloning can be done without any special equipment, tools or expertise using readily available low cost items. The cost per tree can be less than the cost of one seedling chestnut tree and without the risks associated with seedling trees.
Initially the efforts for 2023 were to further refine the best hormone concentrations and then add adjuncts to see if they would further enhance rooting. However, the original plan did not appreciate the need to understand how important it was to find the best time to layer and how devastating an extra dry summer would complicate efforts. Therefore, extra effort to expand the number of suckers layered with a total of seven different hormone treatments on 37 trees. Some trees had many suckers, so we are able to have up to four different hormone treatment per tree with a total of 47 separate hormone treatments on 300 suckers in total. The initial proposal had a minimum of 120 suckers layered each year.
As a result the typical back yard or orchard grower will be able to clone superior trees to share and replant. Providing superior clones to increase the average yield per tree and orchard. Chestnut clones will increase the regional food supply using sustainable permaculture methods and in the native region of chestnuts.
Typically seedling trees are used to establish a chestnut orchard. The seedling orchard is double planted and the inferior trees are removed as the trees start to crowd each other. Also, there is a severe shortage of good nursery stock to establish nut orchards throughout the colder native regions of chestnut and hazelnuts. With the ability to clone regionally adapted productive trees, a portion of the demand can now be satisfied by the grower for either replanting in the unproductive locations or expanding their orchard. The clones will be a known productive tree without the risk of a seedling tree. Orchard expansion can then be using clones on single spacing of 20' x 20' for 108 trees per acre.
Cost will only be mulch, wire or nylon zip ties and some time to mulch, possibly monitor sucker growth, apply hormone and cut the clones from the mother tree. Assuming a lower yield of 36%, and if 300 suckers are layered, a grower should be able to yield 108 trees for an acre of orchard expansion. Item cost is approximately $25 for hormone and nylon zip ties. Another $30 dollars for tar paper if they do not have other items that can be substituted. Orchards have plenty of much that is normally used around trees anyway. The remaining cost is labor and shouldn't exceed 20 hours to layer 300 suckers and then cut the suckers from the mother tree. If we assume $20 an hour for labor that is a total of $455 for 100 trees for a cost of $4.55 per tree. Much cheaper than a seedling tree with many unknowns that you have to double plant take care of and then remove half of them over a ten year period.
Armed with chestnut layering requirements next steps are for outreach. Outreach will be an illustrated brochure, an educators brochure and video presentation will be disseminated to nut-based organizations, extension agents, and farmers. Also, in person outreach seminars will be offered. One outreach effort is already scheduled at a national joint conference by the Chestnut Growers Association and the Northern Nut Growers Association to be held in Syracuse NY, hosted by State University of New York at Environmental Science and Forestry (SUNY ESF). The joint conference has scheduled farm tours of Z's Nutty Ridge LLC and Finger Lakes Nut Farm, where on farm education/demonstration on layering chestnuts will be offered.
Conclusions 2022
With one year of the two-year SARE study behind us we have come a long way in understanding when to layer. Having gone through a total of two seasons of layering chestnut trees it is evident that timing of when to layer has proven to be very important. A weekly sample of sucker growth to verify growth has slowed prior to layering. Also, conscious effort to have more than one month and hopefully two months of growing season is available after layering trees. The root crown cannot be stressed due to lack of rain. The moisture of soil and mulch will be monitored and corrected if needed. Rain data will be recorded as we now have a weather station at Z’s Nutty Ridge.
Although we weren't planning to add secondary hormone treatment in year one, we had so many suckers we did include one secondary hormone NAA. Also, being forced to change rooting treatments for organic farming it did allow for more of what year two of the study was for.
Also, since we have so many suckers to layer we will redo year one efforts and at the same time be able to include year two efforts. Year two efforts are finding what additional hormones and adjuncts may improve rooting.
Education & outreach activities and participation summary
Participation summary:
Three educational tools were created. Two brochures were created, one for the farmer and one for the educator, where the educator brochure included extra background information to assist explaining any questions a farmer may have. Also, for the joint CGA and NNGA at SUNY ESF a poster on, “Layering Chestnuts” was accepted.
In addition to the one-on-one brochure handout and discussions for the New York Chapter of the National Nut Growers Association and three days of handouts during the National CGA and NNGA meeting described above, brochures were brought to two different educator conferences. The first one was on March 14th at New York State Conservation District Employees’ Association, conference at the 2024 Water Quality Symposium Class-agroforestry track, where an announcement was made about the Layering Chestnut brochure, where over twenty educators received the brochure with some time to discuss one-on-one with many who received them. The second educator conference was on March 18th, by the Tompkins County Agriculture and Farmland Protection Board, during the 2024 Tompkins County Agricultural Summit, Ithaca, NY. Brochures were brought to the conference and given out to over 20 educators. Although many brochures were given out the effectiveness was lessoned due to the time constraints didn’t allow for any discussions. Hence, the reason for only 24 estimated agricultural educators were reached.


We did not yet have any Journal articles or live on-farm demonstrations or online training classes. We did have two field days and two poster presentations. We did a 4-H field tour at Z’s Nutty Ridge with approximately 24 members and their parents, on May 10th, 2024. Budding growers were shown how we layer, graft and tissue culture hazelnut trees and we can now layer chestnuts with SARE’s assistance. The tour ended with planting a tree and 4-H members were given a tree to plant if they wanted.
The second field day was July 24th, 2024, as Z’s Nutty Ridge was selected to be a field tour for the joint NNGA and CGA national conference. The Layering Chestnuts poster was presented, brochures were handed out with many one-on-one and small group discussions. There were approximately 80 attendees comprising researchers, educators, farmers and would-be farmers.
There were two poster sessions at the joint NNGA and CGA national conference on July 21 through the 24th, 2024, where Layering Chestnuts poster was posted for two days and the sessions allowed dedicated time during the conference to the posters. NNGA conference was attended by approximately 250+ people that had many educators, researchers, growers and soon to be growers from across the nation, with much face time to discuss how to layer chestnuts.

The Layering Chestnuts brochure was distributed through newsletters, web sites and Facebook groups. The Layering Chestnuts brochure was linked in two Z’s Nutty Ridge newsletters that go out to over 2000 subscribers. The Layering Chestnuts brochure was posted to: Tomkins County, NY Cornell Cooperative Extension agroforestry page, New York Tree Crops Alliance Cooperative, and Z’s Nutty Ridge web pages.
The Layering Chestnuts brochure was posted on relevant nut grower, agroforestry and permaculture Facebook groups and shared widely. A request by NNGA to turn the Layering Chestnuts into an article for possible inclusion into their publication called, "The Nutshell".
Learning Outcomes
Farmers were generally unaware that it is possible to clone their own chestnut trees. Farmers and would be farmers learned that they can clone their best trees, eliminating seedling purchases of unknown quality and have nut production sooner, while saving money. This new information enlightened the farmers that they can be part of orchard improvement for the Northeast and colder regions of the continent.
Project Outcomes
Many tens of thousands of chestnut seedlings have been planted throughout the Northeast with the need to find regionally adapted chestnut trees for breeding and cloning. Now knowing that we can clone our own trees will greatly improve production in existing orchards and allow for this genetic improvement in new orchards. By cloning, farmers won’t have to double plant seedlings of unknown quality, saving time and money of planting, protection, fertilizer, pruning, and weeding for a 7 to 10 years of years before removal of 50% of the trees planted. Saving the tree cost, planting and maintenance and subtracting the time minor expense to layer trees has to be over $300 in total cost per tree planted and then removed. The time, energy and money saved by planting known quality cloned trees, allows for the human capital to double the acreage of agroforestry, assuming the land is available.
Because of this SARE grant we can comfortably state, “By planting cloned chestnut trees at final spacing through stool bed layering, saves almost 50% of the cost for 8 to 10 years of seedlings that are double planted and then have to remove half of the trees.”
Cloning is a year in advance planning to obtain results, and this may sound like a long time but planting trees is a life long endeavor. It will be a few years to clone enough quality trees before whole orchards are planted using clonal stock, but the orchard improvement can now begin.
This newly gained knowledge is assisting with a grant the has been awarded to Tompkins County CCE, " Facilitating Regional Hazelnut and Chestnut Industries through Identification of Improved Genetic Materials and Management practices", and now is seeking further grants to continue this important effort. Also, this newly gained knowledge is assisting to help propagate American chestnut trees to repopulate and restore the forest.
Permaculture and agroforestry practices are expanding faster than quality stock can be grown. Having experience layering hazelnuts and finding out that doing identical practice to chestnuts resulted in death of the chestnut suckers was bewildering. Northeast SARE grant propelled the effort to fruition by making it a priority effort through financial assistance and a fixed timeline. Having the experienced scientists from, The American Chestnut Research & Restoration Project at SUNY ESF being able to do paper searches along with multilingual conversations with European farmers and nurseries narrowed the hormone treatments and timing needed to be successful. We had to overcome two weather events the almost ruined our experiment. Because of the importance of the subject and SAREs assistance we redoubled our effort in 2023 to have very satisfactory results. In future studies, we will minimize the ability of mother nature to interfere by irrigating as we will still need to refine for trees we plan to clone for multiple years.
All researchers and growers who seek genetic improvement or cost savings should and need to use this information for chestnut propagation.