Final Report for FS13-271
Project Information
Cover crop treatments (CCs) were planted two weeks after In-vitro plantain plantlets were planted in the field. After 3 months CCs were soil incorporated and replanted. Soil samples were collected to determine SMB, RSOM, and DHA. The major pitfalls on this stage were above normal rainfall (Table 1), poor germination of Crotalaria seeds; and rodent attack to sorghum seeds. This research shows an increase in all these soil health indicators, demonstrating benefits on soil health attributes by using CCs.
Introduction
In Puerto Rico (PR), plantain (Musa paradisiaca AAB) production is the main crop industry. It is an intensive high input whose current agronomic affect soil quality (SQ), being recalcitrant soil organic matter (RSOM) and soil biota (SB) reductions some of the principal aspects. The RSOM is the part of soil organic matter (SOM) that resists microbial decomposition. During the investigation it also was decided to consider the measure of labile soil organic matter (LSOM). The LSOM pools can be considered as fine indicators of SQ that influence soil function in specific ways, and much more sensitive to changes in soil management practice (Haynes, 2005). Labile compounds are carbohydrates and proteins; and recalcitrant compounds are mainly lignin, but also suberins, resins, fats and waxes (Rovira, 2002).
The goal of this project was to improve the SQ by increasing RSOM and SB through the adaptation and implementation of CC to plantain production systems in PR. The cover crops used were Canavalia ensiformis (Jack Bean), Crotalaria juncea (sunnhemp), and Sorghum bicolor (sorghum). In Gurabo, we experience two main problems of poor germination of sunnhemp and sorghum seeds. The sunnhemp is a legume with elevated production of lignin. Due to the high lignin cellulose, and hemicellulose content in the stem, residue will be slow to break down (Sheahan, 2012). Lignin biodegradation is hindered under anaerobic conditions, a common result of waterlogging (Rovira, 2002). With the increase of rain in Gurabo during April, May, and September to December 2016 (Table 1) the incorporation and replanting of the CCs were affected. The soil on the farm is compounded by Caguabo, Juncos and Mabi (Web Soil Survey, USDA). Caguabo is described as well-drained soil but Juncos and Mabi go from moderate to poorly drained soil. These soils combined with the increase of rain in the moment of incorporation and replanting the area affected the degradation of sunnhemp and germination of this and also of sorghum. The most successful CC was the Jack bean with a rapid germination and adaption to sun and shadow conditions that provide the plantain plantation.
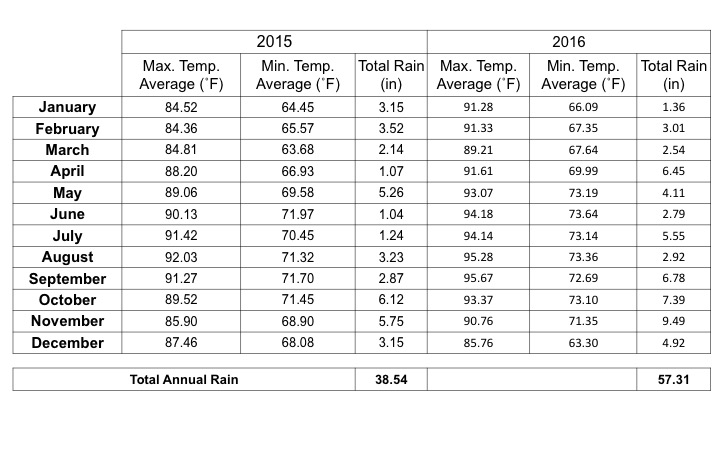 Table 1. Summary of maximum and minimum temperature and total precipitation reported in Gurabo, Puerto Rico during 2015 and 2016.
Table 1. Summary of maximum and minimum temperature and total precipitation reported in Gurabo, Puerto Rico during 2015 and 2016.
The objective of this project is the evaluation of cover crop intercropping in a plantain plantation to conserve and promote a healthy soil environment for plantain production. The effectiveness of the cover crop integration to the plantain system is evaluated using the following soil quality indicators: 1) Recalcitrant Soil Organic Matter (RSOM), 2) soil microbial biomass (SMB) and 3) Dehydrogenase activity (DHA).
Cooperators
Research
The study was conducted at our rented land at the University of Puerto Rico Agricultural Experiment Station (UPR-AES) at Gurabo, PR. The RSOM and DHA analyses were conducted at the Chemistry and Agroenvironmental Sciences Laboratory (UPR-AES) at Río Piedras, PR. The SMB was conducted at the Universidad del Turabo School of Natural Sciences and Technology Laboratories (UTSNST).
Six CC treatments were evaluated in 80 feet2 plots [{1}Jack Bean (JB) (Canavalia ensiformis), {2}Sunnhemp (Crotalaria juncea), {3}sorghum (Sorghum bicolor), {4}JB+Sunnhemp, {5}JB+sorghum, {6}JB+Sunnhemp+sorghum]. Four replicates were established per CC treatment, arranged in a completely random experimental design, including control plots (bare soil).
Soil Sampling
CCs were mowed with a Sicklebar and incorporated to the soil at flowering stage (3 months after planting). Soil samples were obtained for each treatment replica (28 samples) using a soil probe after two weeks of CC’s soil incorporation. Samples were processed in the UTSNST and in the UPR-AES. Soil sampling was made two times during 2016, April and September.
RSOM
Two methods were used for the lecture of organic carbon. For total organic carbon the methods used were those described by Walkley and Black (1934) for May analysis and by Nelson and Sommers (1996) during September analysis. Removable organic carbon was analyzed using the method of hot water described by Wang et al. 2011 and the Tekmar TOC analyzer. Two different methods were used for better results in a more complete oxidation of the organic carbon that the Walkley and Black method does not provides. However, this change made impossible the comparison of the results through time except for the changes of each treatment in comparison with the control.
DHA
Dehydronase activity was determined by the Casida et al. (1977) method. This method consists in the addition of an aqueous solution of TTC and incubated for 24 hours. At the pass of the 24 hours, the soil is filtered and read with a wavelength of 485nm. Dehydronase is one of the principal soil enzymes involved on the oxidation of organic matter and a good indicator of soil quality.
SMB
Microbial counts were determined by most probable number method on the heterotrophic culture media tryptic soy agar (TSA). One gram of soil was sequentially diluted on 9 ml of saline solution (0.85% NaCl w/v). TSA plates were divided in eight sections. An aliquot of 10 ul for each dilution tube was cultivated on separate section. Thus, a plate had eight representations of the original sample. Plates were incubated at room temperature for 24 hours. Appearance of colonies were registered as positive count. The collection of positive counts was interpreted with reference table to assign microbial density.
RSOM analyses
Recalcitrant soil organic carbon (RSOC) and the labile organic carbon (LOC) estimation took significant longer time than expected due to technical difficulties during extraction and analysis of the samples collected in May and September 2016.
Data of the LOC content for each treatment is shown in Figure 1. Comparisons of both periods (May and September 2016) demonstrate an increase in LOC for all treatments with time. The larger LOC increase happened at the treatment number 4 (Jack bean and Sunnhemp). For the observation in the field, the sunnhemp was not so present in the plots. It is probable that the increase in LOC was influenced by the abundant presence of Jack Bean. Control samples also demonstrate an increase in LOC, probably because control plots were covered with weeds some of the time.
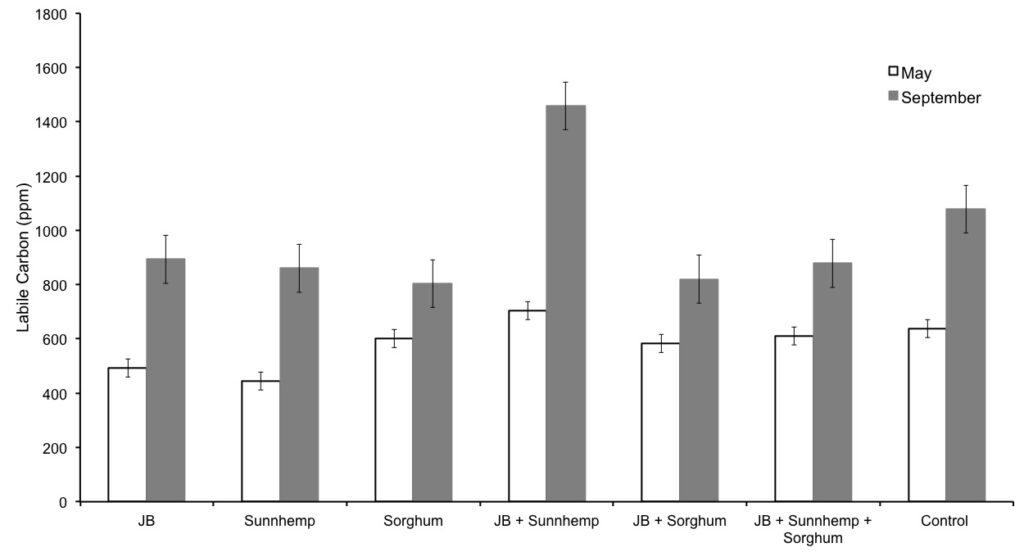 Figure 1. Average of Labile Soil Organic Matter of each treatment during May and September with standard error.
Figure 1. Average of Labile Soil Organic Matter of each treatment during May and September with standard error.
For RSOC the treatment to control ratio was calculated and compared (Figure 2). The RSOC ratio allowed normalizing any differences between field environmental conditions (environmental conditions may affect sample collection), the extraction efficiency of RSOC and sample storage conditions. The recalcitrant carbon was higher for the May observation than for that of September. May’s observation responded to the incorporation of CCs with a better germination of sunnhemp and less heavy rains that could have move or washed the soil. We need to focus on long term for an overview of the production of RSOC. The lignin and other components that are considered to be part of the recalcitrant carbon pool take more time to be available for the analysis (Haynes, 2005). It is probably that the factor of poor germination and long periods of rain during the months in which was necessary the incorporation of the CCs affects the results. Lignin bio- degradation, for instance, is hindered under anaerobic conditions, a cause of waterlogging (Rovira and Ramón-Vallejo, 2002). Another possible factor is the period of wait for taking the sample; maybe it is important to wait more than two weeks after CCs incorporation to take the soil samples.
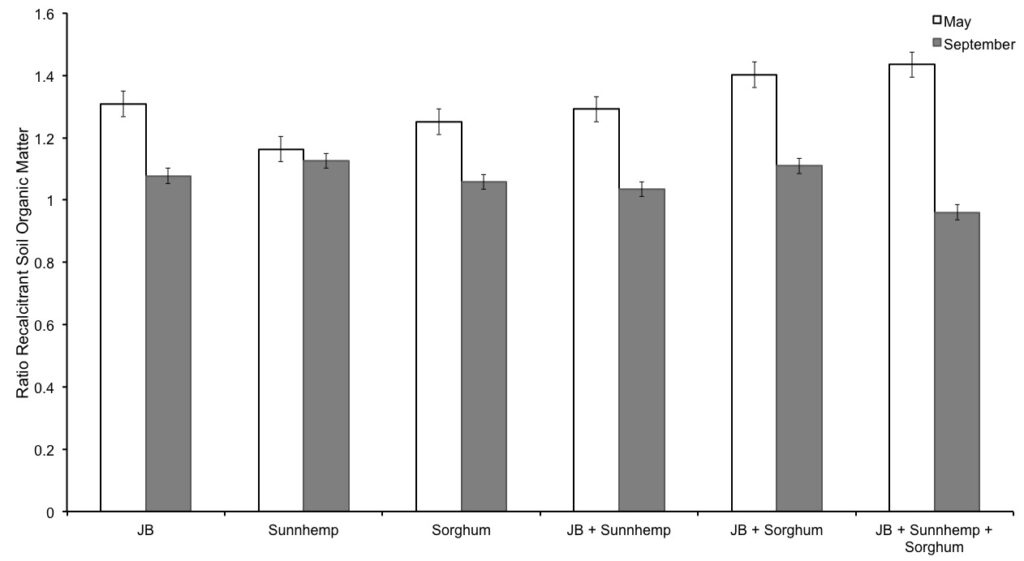 Figure 2. The ratio of each treatment compared with control over time.
Figure 2. The ratio of each treatment compared with control over time.
DHA analysis
Data on DHA activity of the CCs treatments are presented in Figure 3. Compared with the control, DHA activity in May was higher in most of the treatments. However, DHA activity in September was not significantly different among treatments compared with the control. There was not a significant difference among the treatments in time, probably by uncontrolled external factors (rodents and rainfall) that may have affected the normal distribution of the data. DHA analyses were made with water-logged soil samples (had to be taken on high rain period). Gurabo’s clay soils, retain moisture for long periods of time. DHA analyses need to be carried out as soon as possible as to avoid compromising sample quality. Also, soil drying cannot be accelerated on an oven as soil biota would be adversely affected. The impossibility of drying was relatively constant for most treatments in September due to the moist soil (Figure 3). Figure 4 shows the relative ratio to control of labile, recalcitrant and DHA for the CC treatments. Recalcitrant carbon and DHA ratios were relatively constant in time for all treatments. Similar trends occurred for labile organic carbon, except for JB+Sunnhemp treatment that increased for approximately 30% (Figure 4).
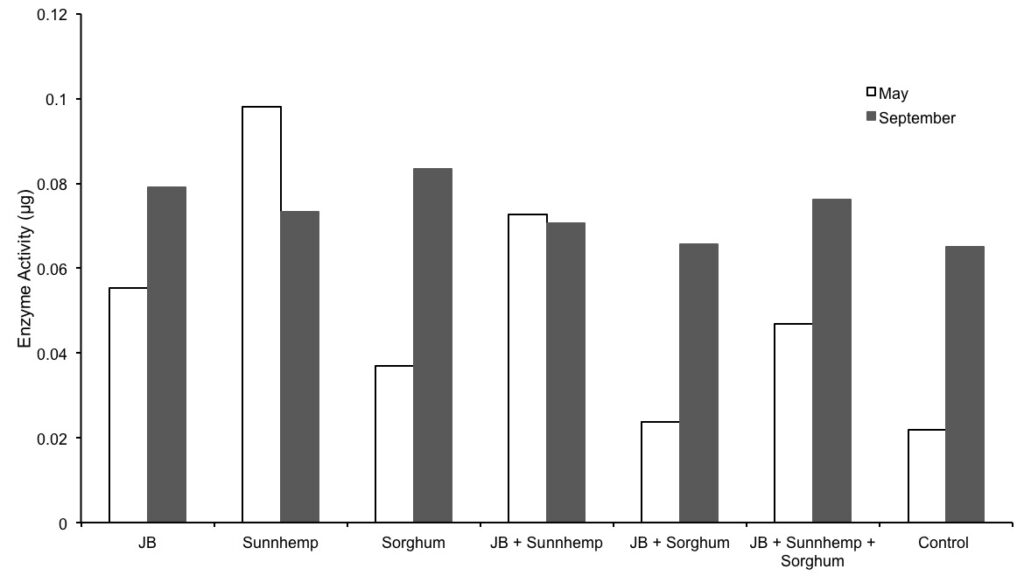 Figure 3. Dehydrogenase enzyme activity found during May and September.
Figure 3. Dehydrogenase enzyme activity found during May and September.
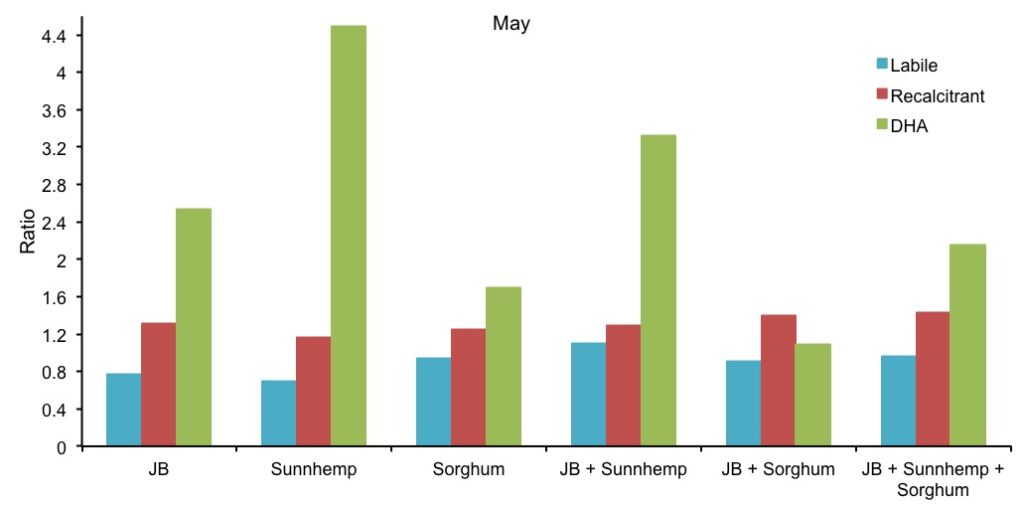
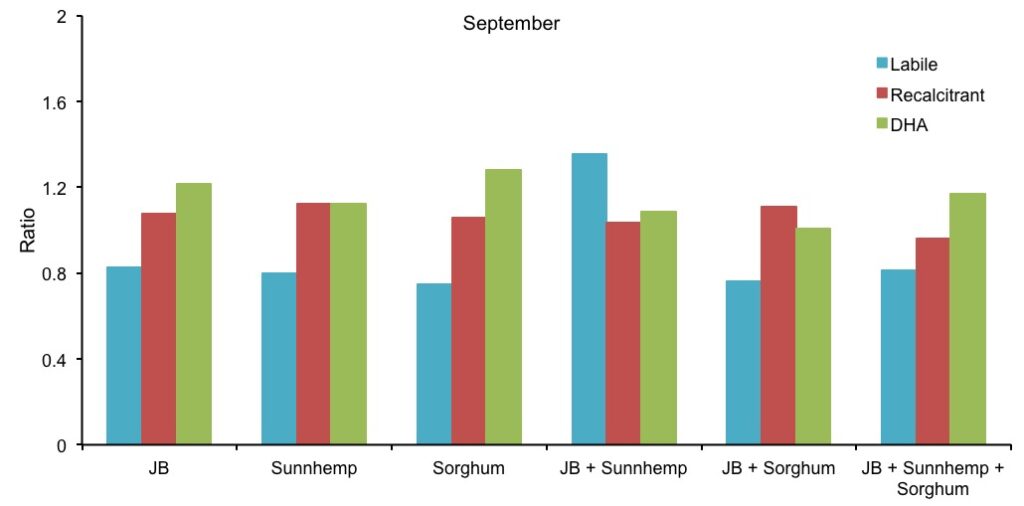 Figure 4. Comparison of the ratio of carbon labile, recalcitrant and DHA of each treatment with the experimental control.
Figure 4. Comparison of the ratio of carbon labile, recalcitrant and DHA of each treatment with the experimental control.
Soil Fertility Samples
From the soil sample made in May complete panorama of the soil at the moment for plantain planting. The results are summarized in Table 2.
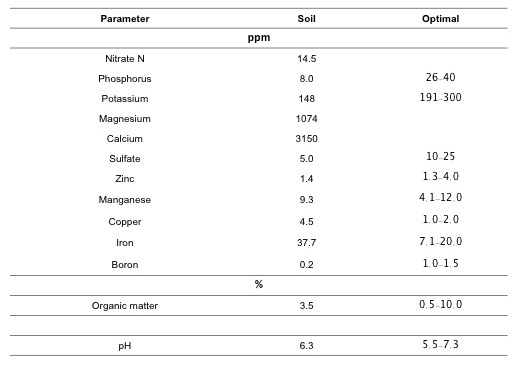
Table 2. Results from soil sample in the experiment plot.
Foliar Sample
Foliar samples of plantain, collected during flowering in January 2017, were sent to AgSource Laboratory. The data could be useful to evaluate the CCs effects in plantain (Table 3). Because the foliar samples were pooled, it is not possible to evaluate CC treatments on plantain nutrition at this stage of the project. However, in general, AgSource report showed low foliar concentrations of zinc, iron, nitrogen, phosphorus, potassium and sulfate at the study area (Table 3). Some of the foliar analysis data confirmed the soil deficiencies above mentioned.
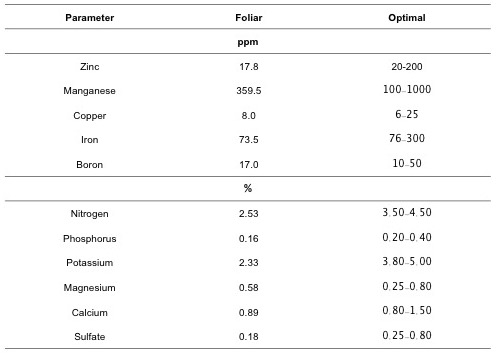
Table 3. Results from foliar sample in plantain of the experiment plot.
SMB analysis
The soil microbial biomass was estimated by the heterotrophic most probable number at the four stages of the experiment (prior treatment, seed addition, grown crops, after treatment) for the seven treatments. The preliminary evaluation for SBM indicates a very low microbial biomass in the study area. As expected, similar results were reported at the beginning of the treatments (seed addition) as is shown in Table 4. The SBM value increased in some treatments during the growing of the cover crops and after cover crops' incorporation. However, the value for the ‘after treatment’ in the control sample seems to be an artifact (sample contamination) that was not corroborated because only one pooled sample was prepared at the laboratory.
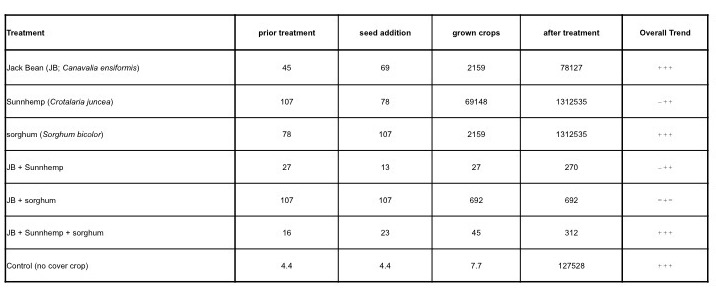
Table 4. Soil microbial biomass estimated by heterotrophic most probable number.
Educational & Outreach Activities
Participation summary:
We have been actively partnering with the USDA-NRCS Caribbean Area on Soil Health promoting activities and workshops. Outcomes from this research have been periodically shared with other farmers and cover crop stakeholders in Puerto Rico, USVI and the US.
At present, we have been actively participating in the newly formed Southern Cover Crop Council. Therefore, these findings will serve as a foundation base for continuing research efforts, and developing cover crops adaptation works for the Southern region, especially in the tropics.
Project Outcomes
This study has shown that compared to the control, the CC treatments for the farm in Gurabo had a positive trend for the DHA activity on both soil sampling periods (May and September). The increased DHA activity compared to control plots pinpoints to an improved soil quality as a result of the use of cover crops. In the Gurabo farm, the most successfully established cover crop was Jack bean (Canavalia ensiformis). Rodents were a major pest that significantly affected the Sorghum establishment, as well as the high rainfall that occurred during 2016.
The RSOC is built up usually on a long-time frame, therefore, it was not possible in the short period of this project to observe a significant effect of the CC treatments, however a trend towards RSOC increase was observed. The LOC are those components of the soil organic matter that are more accessible for the soil microorganisms. Therefore, LOC is a good indicator for microbial activity as well as the DHA activity. Thus, for this short report both indicators, LOC and DHA activity, were used to evaluate the effects of the CC treatments in soil quality. Both parameters seem to indicate the best cover crops were Jack bean and sunnhemp. Of these two CCs, the sunnhemp was more susceptible to changes in the climatic conditions.
Potential Contributions
We have been able to determine that sorghum and Crotalaria are more difficult to establish in heavy clay soils than Jack Bean. Germination rates and rodent damages consequently have affected the establishment. In addition, the soil compaction and heaviness prevented the emerging of many Crotalaria and sorghum seeds, which survived the rodent attack and germinated. Whereas, Jack Bean germination strength pushed up soil clog achieving full germination. Also, Jack Bean has the advantage of shade tolerance under plantain canopy, thus providing a better stand on the second CC planting. These observations have made possible a continuing improvement on how we manage cover crop species use in plantain production systems. Therefore, from a practical standpoint, Jack Bean is more suitable for this type of soils.
Obtained plantain yields on this research have proven to be outstanding, even with sub-ideal fertility conditions as demonstrated by soil fertility and foliar samples. Harvested plantains have achieved “high quality” marketable standards. It is important to state that no nematicide was applied through the plantain crop cycle. This is a very important outcome that confirms CCs nematode suppression effect obtained in previous works with plantain intercropped with Jack Bean by Morales-Mallery (2006). Principal consequence of not using chemical nematicide is the adverse effect of nematodes on plantain plants that fall to the ground because of deficient root anchorage caused by nematode root damage. This reveals a very needed alternative solution for local plantain industry, as presently (2016 to 2017) there are no chemical nematicides available in the market; DupontTM Vydate® L is the only effective chemical nematicide registered for use in banana and plantain in Puerto Rico and has been out of market due to the cease of product manufacturing.
In addition, chemical fungicide applications were reduced by 78% (4 of 18 recommended applications were needed to make in order to keep control of Sigatoka disease). These results have been previously observed in our experience using CCs, and keep repeating. This phenomenon needs to be further studied to determine Sigatoka disease suppression.
Therefore, cover crops use demonstrates advantages from an economic standpoint to the farmer especially on nematode and Sigatoka disease suppression topics. The soil health benefits of increasing soil biota and increasing trends on organic matter have been demonstrated in this research.
Another contribution was the beginning of propagation of mycorrhizae. Extraction of mycorrhizae spores was performed from soil samples taken from the Jack Bean and sunnhemp treatments. After spore extraction, maize was sown on sterilized soil and inoculated with mycorrhizal spores. We have achieved maize root colonization by micorrhizae (Figure 5). We plan to use this mycorrhizae on future In-vitro plantain plantlets.
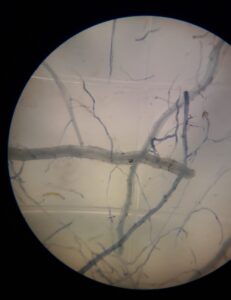
Figure 5. Dyeing of mycorrhizae in maize roots observed under dissection microscope.
Future Recommendations
Continuing Research
A major observation was that plantain foliar damage by Sigatoka fungus was reduced, even during high humidity conditions due to the continuous rains over the growing season. This phenomenon should be further assessed in another research, to see if the presence of cover crops has any relation with this observation of Sigatoka disease reduction.
Cover crop establishment
In this project, a Precision Vacuum Seed Planter was used to sow CCs seeds. It was extremely difficult to achieve a good stand of cover crops especially with sunnhemp and sorghum small seeds. Heavy clay soils as the one where the research was done, did not provide the conditions to allow an efficient use of this equipment, as when very hard soil on dry conditions did not allow good seed to soil contact, and also the technical difficulty associated with waterlogged conditions when rained, thus affecting seed establishment and germination. Also, the line-pathway pattern of seed distribution allowed an easy-to-forage situation for rodents, who just followed the seeding line of sorghum. We recommend using broadcast technique for sunnhemp and sorghum just prior to a rain event, so that germination rates could be better. We assume that with the higher density of used seeds losses by rodent damage could be compensated.
Recalcitrant Soil Organic Matter
RSOC production needs to be assessed on long term analyses. The lignin and other components that are considered to be part of the recalcitrant carbon pool take more time to be available for the analyses.
References
Haynes, R.J. 2005. Labile Organic Matter Fractions as Central Components of the Quality of Agricultural Soils: An Overview. Advances in Agronomy. 85: 221-268.
Matos GI, Junior CS, Oliva TC, Subtil DF, Matsushita LY, Chaves AL, Lutterbach MT, Sérvulo EF, Agathos SN, Stenuit B. 2015. Bacterial community dynamics and ecotoxicological assessment during bioremediation of soils contaminated by biodiesel and diesel/biodiesel blends. Commun Agric Appl Biol Sci.80(1):157-61.
Morales-Mallery, R. 2006. Manejo de nemátodos fitoparasíticos utilizando productos naturales y biológicos. Master Thesis. Universidad de Puerto Rico-Mayagüez.
Rovira, P., V. Ramón-Vallejo. 2002. Labile and recalcitrant pools of carbon and nitrogen in organic matter decomposing at different depths in soil: an acid hydrolysis approach. Geoderma 107 (2002) 109–141.
Sheahan, C.M. 2012. Plant guide for sunn hemp (Crotalaria juncea). USDA-Natural Resources Conservation Service, Cape May Plant Materials Center. Cape May, NJ. 08210.
Walkley, A., B.I. Armstrong. 1934. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil Science. 37 (1) 29-38.