Final Report for GNC15-205
Project Information
Since the invasive fly, spotted wing Drosophila (SWD) (Drosophila suzukii) was detected in the U.S., it has created serious problems in sustainable berry production. Few alternative controls beyond chemical control exist for control of this pest, and new management techniques are needed. Growing berries under protected culture is a new and growing method for season extension, but may also be a way to control this invasive fly and other pests. This project explored sustainable pest management in high tunnel raspberry production through the use of ultraviolet-reducing plastics and exclusion netting. We investigated the ability of plastics to prolong pesticide residual activity and to mitigate pest damage, especially in conjunction with SWD. We also investigated the utility of exclusion netting to control SWD and other pests in high tunnel raspberry production. Reducing ultraviolet light did not impact pest populations, including SWD, but did extend the residues of some chemicals on the leaves of raspberries. We also found that exclusion netting resulted in an average reduction of 73% and 4-week delay in the arrival of SWD in high tunnels. Implications of these results are discussed below. This aids in the development of sustainable and cost-effective production methods for berry growers and helps ensure supply of these nutritious fruits into the marketplace.
Introduction:
The demand for fresh, sustainable, and local food is increasing with berries becoming especially popular. As a result, the number of berry growers in the U.S. increased by 20% in 2012 compared to 2008 (USDA-NASS 2014). However, growers, particularly in northern regions, face many challenges in their production. While almost half of the berry farms in the U.S. are located in the North Central and Northeast, over 80% of raspberry production comes from the Pacific Northwest (USDA-NASS 2014). One of the key limiting factors for these northern growers is the climate, including excessive rain, short growing seasons, and harsh winters (Hanson et al. 2013).
Raspberry growers face many insect pests including Japanese beetles, cane borers, raspberry sawflies, aphids, and spider mites (Funt 2013). Additionally, growers face a new threat from the invasive fruit fly, spotted wing Drosophila (SWD) (Drosophila suzukii). SWD was first detected in the western U.S. in 2008, and was later detected in the Midwest in 2010 (Lee et al. 2011; Isaacs et al. 2011; Asplen et al. 2015). The crops of concern are soft-skinned fruit including raspberries, blueberries, and strawberries, with raspberries being the most susceptible crop (Burrack et al. 2013, Bellamy et al. 2013). In 2014, SWD caused estimated economic losses of $159 million in U.S. raspberry production (Burrack et al., unpublished data). Currently, the main control method is limited to pesticide applications. Because SWD has rapid population growth, growers often spray insecticides weekly during harvest, abandoning their sustainable integrated pest management programs (Lee et al. 2011). There is an urgent need to develop sustainable management practices to decrease insecticide dependence in this system.
High tunnels are a promising solution to both the climatic and pest management hurdles that these growers face (Gaskell 2011, Hanson et al. 2011). These large and unheated protective structures are covered in plastic during the growing season, allowing for earlier harvest in the spring and later harvest in the fall (Demchak 2009). This extended growing season allows raspberry production to become more lucrative, selling before or after the peak in production. Berries grown in high tunnels also tend to have increased shelf-life and decreased disease and pest incidence, making the fruit eligible for sale at farmers markets or grocery stores for premium prices (Demchak 2009, Hanson et al. 2011). However, the advantages of high tunnels need to be better explored, especially given the high initial investment which can deter growers (Conner et al. 2010). High tunnels allow for the possibility of light manipulation through the use of specialty plastics covering the structures, and for physical management options, like exclusion netting, to control insect pests.
The objectives were to determine if protective structures can allow for innovative and sustainable pest management practices, including the use of specialty plastics and physical exclusion. More specifically, our objectives were to:
- Determine the effect of ultraviolet light penetration under protected culture on pests, natural enemies, and pollinators
- Determine the effect of varying levels of ultraviolet light penetration on pesticide degradation over time
- Determine the effect of exclusion netting on high tunnel raspberry for controlling SWD and other insects
Cooperators
Research
Insect communities and abundance
In 2015, this research took place at the Horticultural Teaching and Research Center (HTRC) in East Lansing, MI. In 2016, this research took place at HTRC and at a commercial farm in Coloma, MI. At HTRC, the south half of the three 7.6 x 60 m Haygrove high tunnels (Haygrove Ltd, Herefordshire, UK) were covered with diffuse Luminance® plastic (Visqueen, Stevenston, UK) and the north half were covered with diffuse Lumisol® plastic (Visqueen, Stevenston, UK). Each tunnel had a mix of summer and fall bearing red raspberries (cv. ‘Polka’, ‘Joan J’, and ‘Himbo Top’). At the commercial farm in Coloma, three 7.6 x 122 m Haygrove high tunnels were covered in Luminance and three were covered in Lumisol plastic. Summer and fall-bearing red raspberries were grown, one tunnel per variety (cv. ‘Prelude’, ‘Joan J’, and ‘Himbo Top’).
UV light was measured once weekly on the inside and the outside of each tunnel with a UV light meter (MU-200, Apogee Instruments Inc., Logan, UT). Adult SWD were sampled using yeast-sugar traps (Leach et al. 2016) on a weekly basis with two traps placed in the middle row, 10 m from the edge on either side of each treatment replicate. During harvest, larval SWD were sampled by taking 25 fruit within 5 m of each trap location and immersing them in a salt solution, left for one hour before sifting the solution through a reusable coffee filter and counting the eggs and larvae using a stereomicroscope (Leach et al. 2016). One yellow sticky trap 14 x 23.5 cm (Scentry MultiGuard, Great Lakes IPM, Vestaburg, MI) was placed just above the crop canopy at each trap location. Traps were replaced weekly and the number and type of insects on each trap were identified to family. Pollinators were sampled during peak bloom using both visual observations and collections. For a total of 10 min, trained observers would walk through each treatment and count the number and type of bees pollinating raspberry flowers.
Statistical analyses
Both sites had different pest and natural enemy communities, so sites were separated for analysis. There was no significant interaction between the data from the two years and sites, and so 2015 and 2016 data were combined within each site. Each site had three replicates of each treatment, treated as blocking in the analysis. An analysis of variance was used to compare plastic treatments for insect and natural enemy abundance directly. Tukey’s Honest Significant Difference was used for post-hoc comparisons. Data were analyzed using R (3.2.2., R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Pesticide degradation
This research was conducted in small-scale tunnels covered with different plastic types at the Horticultural Teaching and Research Center in East Lansing, MI. The small-scale tunnels were 1.2 m x 3 m and covered eight potted raspberries (cv. “Polka). The hoops were constructed out of conduit, using a hoop bender (QuickHoops™, Johnny’s Selected Seeds, Fairfield, ME) and plastic was secured the sides of the tunnel using a 0.6 m x 3 m lumber on each side, raised 0.3 m above the ground to allow for airflow. In 2015, we constructed 12 tunnels with 4 replicates covered in three plastic types in a randomized complete block design: Luminance, Lumisol, and an uncovered control. In 2016, we constructed 16 tunnels with 4 replicates covered in four plastic types in a randomized complete block design: Luminance, research-grade clear UV-blocking (Visqueen, Stevenston, UK), research-grade clear UV-transparent (Visqueen, Stevenston, UK), and an uncovered control. The uncovered control was left uncovered unless rain was forecasted, which they would then be temporarily covered in plastic to keep the plants dry. In 2015, one application of insecticides was completed in late August. The insecticides were combined as a tank mix and applied to the plants at the equivalent of 50 gallons of water per acre using a CO2 powered backpack sprayer operating at 50 psi with a single head boom and TeeJet 8003VS spray nozzle. Chemicals were applied at their labeled rate per acre: Actara (4 oz), Admire Pro (8 oz), Asana XL (9.6 oz), Assial (5.3 oz), Brigade (6.4 oz), Delegate (6 oz), Entrust (6 oz.), Exirel (20.5 oz), Malathion (32 oz), and Mustang Maxx (4 oz). Twenty-five leaves were sampled from under each tunnel before the application, immediately after the application dried, and at 1, 3, 5, and 7 days after treatment (DAT). Samples were placed at 0°C immediately and then sent to Synergistic Pesticide Lab (Portland, OR) for residue analysis.
In 2016, the same chemical list was used, except that all chemicals excluding Actara, Delegate, and Entrust were applied at a reduced rate of 50% of the labeled rate, to improve the quantification of the samples. Two applications were applied in early July and early September and leaf samples were subsequently taken after each application at 1, 3, 5, 7, and 14 DAT.
In 2016, fruit residue samples were also taken in early October. Plants grown under the small tunnels did not produce fruit, so canes producing raspberries were taken from the Clarksville Research Center in Clarksville, MI. After the canes were cut, the leaves were removed and canes were then placed in buckets with water and floral foam. The fruits and stems were then sprayed with the reduced rate tank mixture as described above. Fifty fruits were harvested on 0, 1, 3, and 5 DAT. They were frozen immediately after collection and sent to AGQ Labs and Technological Services (Oxnard, CA) for residue analysis.
Statistical analyses
Residue samples were analyzed using a linear mixed model with repeated measures. The trial was setup in a randomized complete block design with four replicates and three (2015) or four (2016) treatments. Treatment differences were established using analysis of variance followed by Tukey’s Honest Significant Difference for post-hoc comparisons. Data were analyzed using R (3.2.2., R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Exclusion netting
Details on this objective are excerpts from the published report: Leach, H., S. Van Timmeren, R. Isaacs (2016). Exclusion netting delays and reduces Drosophila suzukii (Diptera: Drosophilidae) infestation in raspberries. Journal of Economic Entomology, tow157.
Fly-proof netting was installed on two high tunnels over raspberry plantings at a commercial, conventionally managed farm in Coloma, MI. Netting was also installed on one high tunnel over raspberry plants at an organic research farm in East Lansing, MI. All three netted tunnels were adjacent to a paired control tunnel that was open at the ends, and all six tunnels were covered by UV-blocking Lumite plastic coverings. To exclude D. suzukii from the three tunnels, 80 gram Tek-Knit netting (Berry Protection Solutions Stephentown, New York) was applied to the sides of the tunnels by suspending it using 16-gauge galvanized steel wire attached along its length using zip ties along the interior of the curved roof struts, and with shade clips (FarmTek, Dyersville, IA) that held the netting to the wire. To allow movement of pickers to the outside rows of raspberry, the netting was also secured to the sides of the tunnel using 40mm metal clips (Haygrove Tunnels, Mount Joy, PA) and to the ground using landscaping fabric staples. Netting was applied to the outside frame on the two ends of each tunnel, using a different door design to accommodate different needs for access. The research farm site had two 7.6 x 60 m Haygrove tunnels oriented north-south, with each containing organic summer and fall red raspberries (cv. ‘Polka’, ‘Himbo Top’, and ‘Joan J’). In the netted tunnel, an access point was installed in the south end using a small door frame with a magnetic closure. At the commercial farm, four 7.6 x 122 m Haygrove tunnels were oriented east-west with three rows of summer and fall red raspberries (cv. ‘Prelude’) in each. Two of these were netted, and both ends of the tunnels were fitted with two 2.4 x 2.1 m barn style wooden doors covered in netting, which allowed for access by a sprayer. Netting was installed on the tunnels in early June prior to activity of D. suzukii. Bumble bee colonies (Koppert Biological Systems, Howell, MI) were introduced for pollination, using three hives per tunnel at the commercial farm and two hives per tunnel at the research farm. Summer berry harvest occurred in late June through July and fall harvest occurred from the middle of August through early October.
Temperature probes (Hobo Pendant® Temperature Data Logger, Onset Computer Corporation, Bourne, MA) inside radiation shields were hung in the center of each tunnel, taking readings every hour. Two monitoring traps baited with a yeast and sugar mix (Van Timmeren and Isaacs 2013) were placed 9 m from the end of each tunnel at canopy height and were checked weekly. Traps were made from 32 oz. deli cups filled with 150ml of solution and a yellow sticky insert hung from the top. When ripe fruit were available to harvest, 25 ripe raspberries were sampled every week within 5 meters of each yeast-sugar trap and in the center of the tunnel. The weight and diameter of these berries was recorded, and the degrees brix was recorded using a portable refractometer (Model RHB-32ATC, Westover Scientific Inc., Bothell, WA). These fruit were then immersed in a salt solution as described above and the number of Drosophila eggs were counted, along with the number of larvae of each stage as described above. At regular intervals during summer and fall harvest, additional ripe fruit were sampled and the flies were reared to confirm the infestation as D. suzukii. All flies emerging from the fruit were identified as D. suzukii.
To monitor activity of other arthropods in the tunnels, 14 cm by 23.5cm yellow sticky traps (Scentry MultiGuard; Great Lakes IPM, Vestaburg, MI) were suspended above the plant canopy in the middle of each tunnel and replaced weekly. Arthropods captured were identified at least to family and later sorted by functional group. Direct leaf observations were also conducted weekly on 25 randomly selected raspberry leaves in each tunnel. Leaves were evaluated for percent leaf damage and suspected causes of the damage. The numbers and types of arthropods found on the leaves were also recorded.
Statistical analyses
For the high tunnel experiment, fruit quality, temperature, and immature and adult D. suzukii data were analyzed using analysis of variance to compare netted and open tunnels, followed by Tukey’s Honest Significant Difference for post-hoc comparisons. A Student’s t-test was used to analyze data from the direct leaf observations and yellow sticky traps. Data were analyzed using R (3.2.2., R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Insect communities and abundance
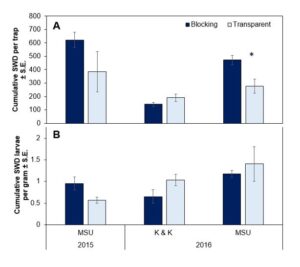
In both years across the two sites, no significant difference was found in SWD populations under the different plastic types from both adult capture and fruit sampling (Figure 1). Additionally, no statistical difference was found among insect pests on the yellow sticky traps or in direct observations for the key pests at each site. Common pests varied between sites, but generally included small sucking pests, like aphids, thrips, leafhoppers, and mites. Other common pests included raspberry sawfly, raspberry beetles, and sap beetles.
The majority of the natural enemies that were found on the traps and from direct observations were lacewings, spiders, syrphids, lady beetles, minute pirate bugs, and predatory mites. From the yellow sticky traps in 2015, there was no statistical difference in natural enemy abundance between the two treatments. From the direct observations in 2015 and 2016, there were also statistically similar numbers of natural enemies in the treatments.
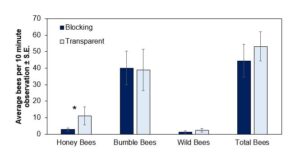
Across both years, there was no difference in the number of bumble bees, wild bees, or total number of floral visits among all bees between the two plastic types at both locations. However, there were significantly fewer honey bees observed making floral visits under the UV blocking treatment compared to the transparent treatment in the ten-minute observations conducted (F [1,8] = 8.19; p = 0.02) (Figure 2). Wild bee numbers were statistically similar across both treatments, but with a trend for fewer under the blocking treatment.
Pesticide degradation
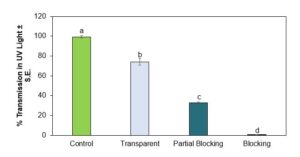
The amount of ultraviolet light under each of the treatments differed among the plastic types, with the full blocking material excluding the most UV light (Figure 3). The ten insecticides tested in 2015 and 2016 were found to degrade differently between the UV-blocking and UV-transparent or uncovered treatments. However, the time of year that the trial was carried out and the insecticide type caused different patterns in their degradation across the plastic types.
In 2015, the blocking treatment was significantly higher than the open uncovered treatment for nine out of the ten insecticides tested over the entire experiment. Spinosad, used in Entrust, an organic insecticide, was not found to degrade differently among the treatments (F [2,53] = 2.74; p = 0.07). The partial transparent treatment often did not differ between the two other treatments for most the chemicals. In some cases, as with acetamiprid (Assail), the partial transparent was statistically similar to partial blocking plastic, and both differed from the open control (F [2,53] = 8.74; p < 0.001). For imidacloprid (Admire Pro), the partial transparent plastic resulted in residues that were statistically similar to the open control, and both treatments had lower residues than the blocking treatment (F [2,53] = 19.09; p < 0.001). Overall in 2015, the partial blocking plastic significantly increased the residues across all of the sampling dates.
Similarly in 2016, the July leaf trial results showed that the fully or partially blocking materials had significantly higher residues than the transparent or open control for nine out of the ten chemicals tested. Malathion did not show a difference among any of the plastic types (F [3,71] = 0.25; p = 0.86), partly because it degraded very quickly among all treatments. For all chemicals except for imidacloprid (Admire Pro), the blocking treatment was statistically similar to the partial blocking treatment. For all chemicals except acetamiprid (Assail), the transparent treatment was statistically similar to the uncovered control.
From the fruit residues tested in 2016, there were fewer differences between the plastic treatments. Most insecticides had higher residues under plastic treatments compared to the uncovered controls. Some chemicals, like acetamiprid (Assail) and malathion (Malathion) were not significantly affected by the plastic type. In some cases, residues from the blocking treatments were lower than the residues from the transparent plastics, such as with bifenthrin (Brigade).
Exclusion netting
Details on this objective are excerpts from Leach, H., S. Van Timmeren, R. Isaacs (2016). Exclusion netting delays and reduces Drosophila suzukii (Diptera: Drosophilidae) infestation in raspberries. Journal of Economic Entomology, tow157.
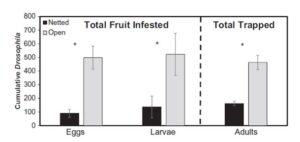
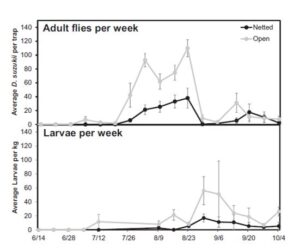
There were significantly fewer Drosophila eggs, larvae, and adults in the netted tunnels than the open tunnels at both sites (Figure 4). Over the entire season, there was an 82% reduction in Drosophila eggs (F =18.5; df = 1,16; p = 0.0002), a 74% reduction in Drosophila larvae (F = 4.7; df = 1,16; p = 0.02), and a 65% reduction in D. suzukii adults (F =30.0; df = 1,10; p = 0.0003) in the netted tunnels (Figure 4). Over the entire season, there were significantly fewer first instar larvae found in fruit in the netted tunnels (81.4 ± 67.8 larvae/kg) than the open tunnels (361.6 ± 206.6 larvae/kg) (F =4.8; df =1,16; p = 0.02). There were also significantly fewer second instars from fruit in the netted tunnels (45.2 ± 45.2 larvae/kg) compared to the open tunnels (162.7 ± 62.6 larvae/kg) (F= 3.6; df = 1,16; p = 0.03). Third instar larvae from fruit in the netted tunnels (9.0 ± 9.0 larvae/kg) was reduced compared to the open tunnels (22.6 ± 4.5 larvae/kg), but not significantly so (F= 0.8; df = 1,16; p = 0.18). Furthermore, the netted treatments delayed the arrival of D. suzukii adults by twenty-three days (Figure 5). The average first catch in the open tunnels was on July 8 (± 12.5 days) and the netted tunnels on July 31 (± 18.7 days). Larval infestation was delayed by twenty-four days with the average first larval detection in the open tunnels on August 16 (± 17.6 days) and the netted tunnels on September 3 (± 14.1 days). However, none of the netted treatments maintained zero adult D. suzukii captures in the traps or zero larval infestation in the raspberries, and later in the season the infestation built up inside the netted tunnels (Figure 8). Despite this, the overall level of infestation remained lower in the netted tunnels than in the open tunnels.
Netting of the high tunnels affected other arthropods present in this system, though the composition of the arthropods remained similar between the two treatments. Examination of the yellow sticky traps revealed that the abundance of pests, natural enemies, and pollinators was reduced in the netted tunnels compared to open tunnels. Pest insects and mites at the commercial farm were reduced by 44% (t = -7.58, p = 0.008). Natural enemies were reduced at both sites by 48% (t = -2.88, p = 0.02). Pollinators, excluding the supplemented bumble bees, declined by 77% although this reduction wasn’t statistically significant due to variability among sites (t = -1.78, p = 0.07). From the direct leaf observations, we observed similar reductions of pests (42% decline) and natural enemies (32% decline) but these were highly variable between sites and there was no significant difference between netted and open tunnels (t = -0.47, p > 0.32; t = -0.30 p > 0.38, respectively). We found no instances of insect abundance increasing in the netted tunnels, though some insects were more affected by the netting than others. Thrips, mites, and leafhoppers were reduced by greater than 50%, whereas aphids and raspberry beetles were reduced by fewer than 20% compared to populations found in the open tunnels, from both yellow sticky trap and direct leaf observation data. Spiders were the only natural enemy that appeared to be less affected by the netting with an average reduction of 21%. Average percent leaf damage throughout the entire season in the open tunnels (2.3 ± 1.3%) was slightly higher than the netted tunnels (2.2 ± 1.8%), but not significantly so (t = -0.05, p = 0.47).
There was little effect on fruit quality characteristics of raspberries grown under the netted high tunnels. The average weight (Netted: 2.7 ± 0.1g; Open: 2.5 ± 0.1g), diameter (Netted: 16.1 ± 0.2mm; Open: 15.7 ± 0.2mm), and brix (Netted: 7.5 ± 0.6 °Bx; Open: 7.1 ± 0.2 °Bx) of the fruit collected from the netted tunnels was not significantly different from the fruit collected from the open tunnels (F = 2.91, df = 1,4, p > 0.16). The temperature in the netted tunnels was not significantly different from the open tunnels (Netted: 21.7 ± 0.2 °C, Open: 21.6 ± 0.3 °C) (F = 0.04; df = 1,4; p = 0.83). Likewise, the average maximum temperature was similar in the netted tunnels (Netted: 42.9 ± 0.8 °C, Open: 42.2 ± 0.8 °C) (F = 0.39; df = 1,4; p = 0.56).
Figure 1. (A) The cumulative number of SWD adults found per trap (± S.E.) and (B) SWD larvae per gram (± S.E.) season long under UV-blocking or transparent plastics. Bars marked with an asterisk denote treatment differences at each site at alpha = 0.05.
Figure 2. The average number of bees seen per each ten-minute observation (± S.E.) under tunnels covered in UV-blocking or UV-transparent plastics between both sites and years. Bars marked with an asterisk denote treatment differences at alpha = 0.05. (Statistics: Honey Bees: F [1,8] = 8.19, p = 0.02, Bumble Bees: F [1,8] = 0.002, p = 0.97 Wild Bees: F [1,8] = 0.1 p = 0.77 Total Bees: F [1,8] = 0.28, p = 0.61).
Figure 3. The average percent transmission of UV light (± S.E.) under each of the plastic types covering the miniature tunnels used for the pesticide degradation study. UV light was measured using a handheld UV meter (Apogee MU-200). Bars marked with different letters denote treatment differences at alpha = 0.05 (F[3,12] = 571.3; p <0.001).
Figure 4. Cumulative number of Drosophila eggs and larvae per kilogram of raspberries (± S.E.) and the cumulative number of D. suzukii adults caught each week per trap (± S.E.) in open and netted high tunnels throughout the entire raspberry season. Bars marked with an asterisk denote life stages where treatments were significantly different at alpha = 0.05 (Leach et al. 2016).
Figure 5. The number of D. suzukii adults caught per trap (± S.E.) per week in open and netted tunnels (top) and the number of Drosophila larvae per kilogram of fruit (± S.E.) per week in open and netted tunnels (bottom) throughout the raspberry growing season. Summer berry harvest started in mid-June and ran through mid-July, whereas fall raspberry harvest began in August and ended in late September (Leach et al. 2016).
Educational & Outreach Activities
Participation summary:
Leach, H., Van Timmeren, S., & Isaacs, R. (2016). Exclusion netting delays and reduces Drosophila suzukii (Diptera: Drosophilidae) infestation in raspberries. Journal of Economic Entomology, tow157.
Integrated Strategies for Management of Spotted Wing Drosophila in Organic Small Fruit Production. MSU Extension Bulletin. Heather Leach, Matthew J. Grieshop, and Rufus Isaacs. October 2016. http://www.ipm.msu.edu/uploads/files/SWD/MSU_Organic_SWD_factsheet_Dec2016.pdf
Exclusion netting and harvest frequency for SWD management (with Heather Leach, Eric Hanson, Josh Moses, and Phil Fanning). Great Lakes Expo, Grand Rapids, MI. December 5, 2016, grower, industry, and agricultural consultant audience (80 people).
Potential effects of tunnel plastics on pesticides and pest management. Heather Leach, Eric Hanson, and Rufus Isaacs. Presented at the North American Berry Conference in Grand Rapids, MI. December 5, 2016, grower, industry, and agricultural consultant audience (50 people).
Rebuilding Raspberry IPM Programs after Invasion by Spotted Wing Drosophila. Heather Leach, Eric Hanson, Josh Moses, Steven Van Timmeren, and Rufus Isaacs. Presented at the International Congress of Entomology in Orlando, FL, 26 September 2016. Academic and research audience (100 people).
Exclusion Netting for Spotted Wing Drosophila Control in Michigan Raspberries. Poster. Heather Leach, Tobias Marks, Eric Hanson, and Rufus Isaacs. Presented at the Entomological Society of America National Conference in Minneapolis, MN, 16 November 2015.
High Tunnel Fruit Production Tour. Presented to growers and interested public on insect management approaches for fruit grown under high tunnels (40 people). Extension event took place at the Horticulture Teaching and Research Center in Holt, MI, 5 July 2016.
Project Outcomes
We have found that commercially available plastics for high tunnels that vary in UV-transmittance are not likely to strongly impact the insect complex within raspberry systems, including SWD. Pollinators may be affected by the reduction in UV light, as seen with honey bees in this study, but more research on the effect of pollinators is needed. Pesticide degradation was found to differ on raspberry leaves depending on the amount of UV light exposure. Reduced UV light resulted in significantly higher residues overall when compared with transparent plastics or the uncovered control. This same trend was not as clear with the raspberry fruit, which may be due to the shorter duration of the study. Additionally, canes were cut from plants to supply fruit, and this may have affected the degradation over time. However, we observed increased residues when raspberry fruits were covered with plastics compared to uncovered, which may have implications for all high tunnel grown raspberries. Increased residual activity may extend the efficacy of the insecticides, resulting in fewer applications needed and greater time between sprays, which would ultimately save time and money for the grower and increase sustainability. Efficacy of these chemicals over time on SWD or other pests was not presented in this study, however, and remains to be tested.
Exclusion netting can be a viable way to reduce SWD and other pests without negatively affecting the fruit quality in high tunnel raspberry production. Moreover, we found that the exclusion netting resulted in a delay in the arrival of SWD, which could eliminate SWD in the summer crop of raspberries entirely. Delaying the arrival of SWD also reduces the amount of insecticide that needs to be applied during this time, reducing costs and risk to pollinators. This is one of the first alternative management tactics proposed for SWD that could be used in place of chemical management. We also found that chemical control and exclusion netting work best when combined, maximizing the possibility of marketable fruit. SWD has increased the amount of insecticides being applied in raspberry production, and this does not represent an adequate long-term management strategy. Exclusion netting and the potential of UV-blocking plastics to increase pesticide residual activity, among other new control measures, will help to make these systems more sustainable for the grower, consumer, and environment.
Economic Analysis
The cost to net the perimeter of one acre of high tunnels (four 7.6 x 122 m tunnels) was estimated using the cost of purchasing and shipping netting, the cost of labor for installation of the netting at $10/hr, the cost of supplies (hoop clips, etc.) to install the netting, and the cost of bumble bees for pollination services. The netting is expected to last 5 years (Tek-Knit) and this cost could be amortized over these years. We also expect labor costs to decrease as experience with the installation increases. No other economic analysis was performed as part of this research.
Farmer Adoption
There are no formal metrics on farmer adoption of this project. While surveys on plastic use by growers are limited, we don’t expect this research to change the trends in plastic type for insect control. However, interest in exclusion netting to control SWD continues to grow as other management techniques, including frequent applications of broad-spectrum insecticides, are not suitable for long-term or sustainable management. Growers have expressed interest in utilizing meetings from grower meetings, but hesitation comes from the large upfront costs and maintenance required to install and take-down the netting each year. Many commercial growers of raspberries and blueberries, particularly in the western U.S. and regions of Europe, are now utilizing exclusion netting on a large-scale.
Areas needing additional study
- More research should be focused on how pollinators are responding to environments with reduced UV light and whether this affects yield or fruit quality.
- Additional research should focus on how longer pesticide residues under UV-reducing plastics affect pest control and if they reduce the number of sprays needed .
- More information should be collected on the economic advantage of the netting, including long-term costs of labor and repair.
- Additional studies should also be performed on the success of exclusion netting in hotter regions, where fruit quality could be compromised due to heat.
- Future studies could focus on the combination of repellents or insecticide-impregnated netting to further suppress suzukii populations.
Information Products
- Exclusion Netting Delays and Reduces Drosophila suzukii (Diptera: Drosophilidae) Infestation in Raspberries
- Integrated Strategies for Management of Spotted Wing Drosophila in Organic Small Fruit Production
- Rapid harvest schedules and fruit removal as non-chemical approaches for managing spotted wing Drosophila
- Pest Management Approaches for Raspberry Growers with Heather Leach