Final report for GNC16-226
Project Information
Organic sweet potato growers are challenged by sustainable soil management, which impacts the occurrence of soilborne disease, weed competition, and other pest issues that consequently reduce the tuber yield and/or quality. Being organic, their options are limited for pest control, especially fungal disease that affects the roots and/or tubers. The proposed research aims to develop a strategy of using cover and/or green manure crops, and to evaluate their effects on soil and plant health, as well as disease and weed suppression. The proposed experiment was conducted at a Certified Organic producer’s field based in central Missouri. Effects of rapeseed and cereal rye, both as cover or green manure crop, were assessed in a randomized complete block design for a one-year period. Soil samples were collected for soil organic matter, pH, nutrients, and microbial analysis by using phospholipid fatty acid (PLFA) analysis three times over the course of the study. The PLFA assessed various groups of fungi and bacteria for their diversity and density. Evaluations also included weed coverage, sweet potato plant vigor, disease occurrence (Fusarium root and stem rot), and sweet potato yield. Results from this experiment would increase our knowledge on how to best incorporate cover crops into organic sweet potato production by improving soil health as well as suppressing weed and disease. The expected outcome included development of a strategy for improving sustainability and productivity of organic sweet potato. The project was also evaluated by survey at field days which sampled the farming community for their opinions and acceptance towards this project.
The proposed research would result in an increased knowledge and understanding of:
- the effect of cover and/or green manure crops on soil properties
- the effect of cover and/or green manure crops on soil microbial population/activity;
- The effect of cover and/or green manure crops on sweet potato quality and yield,
Specifically, the learning outcomes included increased awareness for both farmers and agriculture professionals on the use of cover crops in organic production systems, as well as enhanced skills and motivation to adopt such a practice.
The action outcomes would include implementation of such a principle and practice in production from farmers who were exposed to and influenced by this project. Agriculture professionals who were involved or aware of this experiment could also be stimulated for performing further research in this area which would consequently facilitate productions of extension publications and field production guides. Additionally, farmers who subsequently benefit from reduced losses and improved economic wellbeing in organic sweet potato production will most likely support research in this nature and consequently enhance the partnership between researchers and stakeholders. This will ultimately result in the sustainable use of resources and hence, improved environmental stewardship and quality.
Cooperators
Research
Experimental Site
Field plots were established at the Share-Life Farms (Longitude: -93.2225, Latitude: 39.1341) located in Central Missouri, 6.5 miles north of I-70 in Saline County. Share-Life farms is a central Missouri Community-Supported Agriculture (CSA) farm that has been Certified Organic continuously since 2004. The soils are silt loam, topography is upland loess and the elevation is 764 ft (233 m).
Experimental Design
Treatments were arranged in a split-plot with the whole plot variable being the use of cover crop species as cover or green manure, in addition to control. The sub-plot variable was the cover crop species cereal rye (Secale cereale L.) or rapeseed (Brassica napus I.). Individual whole-plot measured 6 m × 6 m, subplot measured 3 m × 6 m, and the total plot area measured 18 m x 34 m. Whole plot variables were arranged in a randomized complete block design with 3 replications, with a 7.6 m-wide alleyway between blocks.
Plant Material and Planting
Organic rapeseed and cereal rye seeds were obtained from Johnny’s selected seeds (955 Benton Ave, Winslow, ME 04901) and GrowOrganic (Peaceful Valley Farm & Garden Supply, 125 Cydesale Court, Grass Valley, CA 95945) , respectively. The sweet potato slips “Beauregard” were purchased from Southern Exposure seed exchange (P.O Box 460, Mineral, VA 23117).The rapeseed and cereal rye were established on the 3rd of October 2016 at rates of 7 lbs and 100 lbs/acre respectively, using a mechanical hand planter (Figure 1). The sweet potato slips were planted on the 22nd of June 2017 using a planter (Figure 2), at an intra and inter row spacing of 30 cm and 150 cm, respectively.


Termination of cover crops
The cereal rye and rapeseed were first terminated on the 24th April, 2017 using a mower (Figure 3) followed by a second time on the 6th of June, 2017 using a roller crimper (Figure 4), due to the regrowth of the cover crops. A tiller (Figure 5) was used to incorporate the cover crops into the soil for plots that are designated to be green manure plots, on both the 24th of April and 6th June following termination.



Measurements
Data was collected for visual assessments of cover crop plant vigor and coverage once monthly in February, March and April 2017. Plant vigor was estimated based on a 1-5 scale where 5 was highest, 3 and 1 were average or lowest, respectively. The coverage was estimated as the percent area of the sub plot covered by the cover crops. The percent weed cover was estimated once monthly from February to October, using a 61 cm x 61 cm frame (Figure 6) placed 3 times randomly in each subplot.

Soil samples were collected three times: after planting cover crops (10th October, 2016), after terminating cover crops but prior to planting sweet potato (22nd June, 2017), and before sweet potato harvesting (6th November, 2017). From each sub-plot, ten random soils cores were collected by a steel probe with diameter of 10 centimeters to a depth of 15 centimeters. The cores were separated from rocks by passing through a 2 mm sieve. Soil samples from each sub plot were then mixed thoroughly, and stored at room temperature for further analysis.
Soil samples were analyzed for OM, pH, and plant available nutrient including P, K, Fe, Ca, S, Mn, and NH4-N and NO3-N. Soil microbial community structure and biomass were determined by analyzing soil phospholipid fatty acid (PLFA) following the procedure described by Bligh and Dyer (1959), and later modified by Petersen and Klug (1994). Briefly, soil samples between 2 and 2.5 g were added to Teflon-lined screw cap cultures tubes and freeze-dried before extracting the total lipids. The total lipid extract was then fractionated into glycol-, neutral, and polar lipid fractions, and the polar lipid fraction was trans-esterified with mild alkali to recover PLFA as methyl esters (Ibekwe et al. 2002). A gas chromatograph equipped with flame ionizer detector was used to separate, quantify and identify the methyl esters against an internal standard. The peaks of the samples were then compared with the database of known microbial fingerprints to determine the molar responses of taxonomic microbial groups of bacteria, fungi, gram-positive bacteria, gram-negative bacteria, actinomycetes, and mycorrhizae, and the ratios of fungi to bacteria (F/B) ratio, cyclopropyl 17 to monoenoic precursor (Cy17/pre) ratio, and saturated to monounsaturated fatty acid (Sat/mono) ratio. Total PLFA concentration (p mole g−1 soil) was also determined as the total microbial biomass.
During growth, the sweet potatoes were visually assessed for plant vigor and insect damage on a monthly basis from the time of transplanting in June to right before harvesting in October 2017.
The sweet potatoes were harvested over several days from 6th of November to 13th November, using a potato digger (Figure 7). A week after harvest, the numbers of tubers from each sub plot were counted and graded based on the USDA market grade standards, and marketable (at least 3.81 cm in diameter and free of blemish, spot and disease – Figure 8) and culls (roots of any size that exhibited wounds, breakage, or severe cracking that would reduce storage life – Figure 9) (USDA, 1981) were separated. The tubers were also assessed and recorded for disease symptoms.



Statistics
All data collected were subjected to ANOVA using SAS (9.4), and significant means were separated by Fisher’s Protected LSD at P<0.05.
References
- Bligh, E. G., & Dyer, W. J. (1959). A rapid method of total lipid extraction and purification. Canadian journal of biochemistry and physiology, 37(8), 911-917.Balesdent, J., Chenu, .C, Balabane M. (2000). Relationship of soil organic matter dynamics to physical protection and tillage. Soil Till Res 53:215–230
- Petersen, S. O., & Klug, M. J. (1994). Effects of sieving, storage, and incubation temperature on the phospholipid fatty acid profile of a soil microbial community. Applied and environmental microbiology, 60(7), 2421-2430.Ranjard, L., Richaume, A. (2001). Quantitative and qualitative microscale distribution of bacteria in soil. Res Microbiol 152(8):707–716.
- [USDA] U.S. Department of Agriculture. (1981). U.S. Standards for Grades of Sweet potatoes.
Cover crops
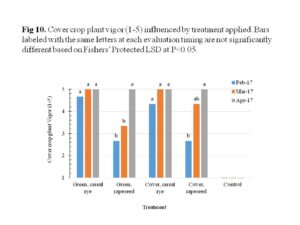
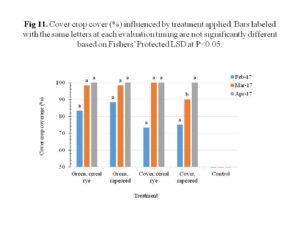
The cover crops overall performed well, with cereal rye showing greater initial plant vigor than rapeseed. The cereal rye maintained good plant vigor, while the rapeseed improved over time with the highest plant vigor before termination (Figure 10). The plant cover on the other hand showed no significant differences between treatments, except for the rapeseed used as cover crop which was lower during the evaluation in March (Figure 11). When it was time for termination, both cereal rye and rapeseed were flowering and covered the entire plots. Figures 12 through 15 illustrate this.




Weeds
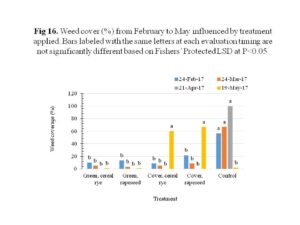
There were a number of weed species established in the plot area during the course of the study, including grasses like yellow foxtail, and broadleaf weeds like Amaranth, clover, dandelions, morning glory, henbit, purslane, Queen arms lace, crownvetch and milkweed. The weeds were significantly suppressed when the cover crops were still in the field from October, 2016 to 24th April, 2017 and no significant differences were observed between cover crop species used (Figure 16). The cover crop plots showed significant increase of weed cover in May, 2017 because these plots were not tilled. In comparison, minimal weed cover was found in green manure plots and control plots since they were tilled first on 24th April and a second time on 6th June.

Once the cover crop was terminated, the weeds increased significantly later in June through October, with the highest coverage found in the control plots (Fig 17). Regardless of species, plots with cover crops tilled into the soil as green manure showed significantly less weed cover from July to October (Figure 17). There were no differences in weed suppression between cover crop species for the most part except for June, just after termination of the cover crops. Figure 18 to 20 illustrate the weeds in all plots in August 2017, one and half months after the sweet potato slips were transplanted and established.



Soil microbes (Phospholipid Fatty Acid Analysis)
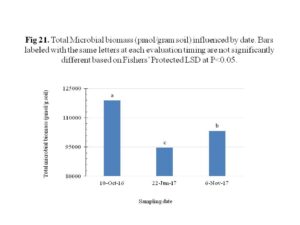
The total microbial biomass was significantly influenced by sampling date, with the highest numbers being observed at the 1st sampling on 10th October 2016. The numbers decreased significantly during the second sampling on 22nd June, 2017, possibly due to the tillage done in preparation for planting of sweet potatoes. It has been shown that tillage reduces soil macroaggregate content which provides an important microhabitat for microbial density, diversity, and activity (Ranjard and Richaume 2001; Six et al., 2002). The last sampling carried out on 6th November was performed on undisturbed soil right before harvesting; this may explain the 9% increase of microbial biomass since the second sampling (Figure 21). There was however, no significant effect of the treatments on the total microbial biomass, which was different from early reports where increase in microbial biomass was observed following utilization of cover crops (Mendes et al., 1999; Wang et al, 2007; Finney et al., 2017). It is likely that several seasons of cropping are necessary before such a change can be found.
Sampling date also significantly affected the fungi, mycorrhizae, gram positive, gram negative, anaerobes and eukaryotes groups (Figure 22). The fungi and gram positive bacteria decreased overtime, while the AM Fungi and gram negative increased overtime. The fungal and bacterial densities may have been influenced by tillage which has been demonstrated to affect soil temperature and humidity, thus influencing strongly soil microbial abundance and in particular fungal development (Frey et al., 1999; Spedding et al., 2004). Additionally, tillage causes direct tissue damage to the fungi leading to a reduction in their abundance at soil surface (Balesdent et al. 2000; Six et al., 2002). No till systems have been demonstrated to have a positive impact on soil physical and chemical properties (Six et al., 2002), and on soil microbial biomass and activity (Sapkota et al., 2011).
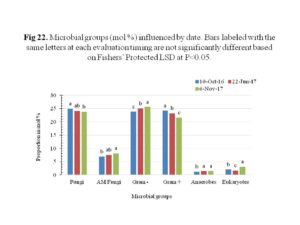
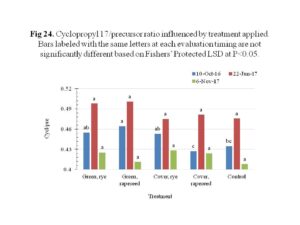
Community change and stress indicator ratios to depict shifts in populations and stress respectively, were determined and significant sampling timing effect were observed (Figure 23). Stress indicator ratios including the saturated to monounsaturated fatty acids and cyclopropyl 17/precursor were developed to detect soil microbial shifts in response to unfavorable conditions, such as extreme temperatures and pH, suboptimal substrates and water, and toxin accumulations (Bank et al., 2014). The ratio of saturated to monounsaturated fatty acids decreased over time while the cyclopropyl 17/precursor increased at the second sampling date and then decreased. The cyclopropyl 17/precursor showed significant differences among treatments for the sampling on 10th October 2016 (Figure 24). It appeared that rapeseed used as cover crop showed reduced cyclopropyl 17/precursor ratio than other treated plots. This effect, however, was only observed in one sampling date shortly after establishment of cover crops on the 3rd of October 2016. Collectively, the ratios give indication of overall decline in stress conditions for the bacteria overtime, hence the decrease.
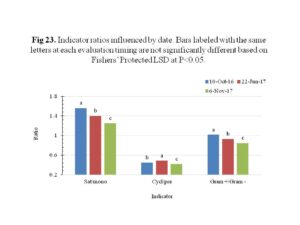
The ratio of gram positive to gram negative bacteria showed a decrease over time, since the gram negative bacteria increased while the gram positive bacteria reduced. The gram positive versus gram negative bacteria likely reflects the dominant metabolic strategy used by each of these bio marker groups. Specifically, gram positive bacteria tend to utilize more recalcitrant C sources, while gram negative bacteria tend to exploit more labile C sources (Fierer et al., 2007).
Soil chemical property analysis
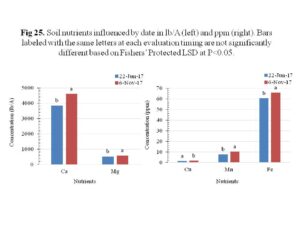
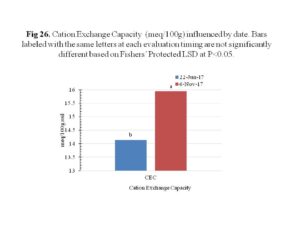
Soil chemical property analysis was performed to determine the pH, Cation Exchange Capacity (CEC), nitrate, ammonium, phosphorus, potassium, magnesium, calcium, sulphur, manganese, copper and iron. The calcium, magnesium, copper, manganese and iron had significant increments over time (Figure 25). The CEC was also significantly influenced by the sampling date, with a significant increase over time (Figure 26). Collectively, these results indicated that cover crops, regardless of species or method of termination, did not affect soil chemical properties significantly.
Sweet potato yield
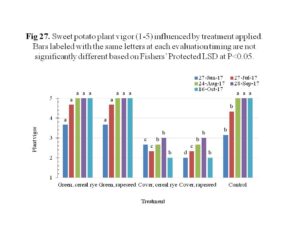
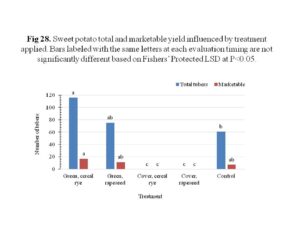
After transplanting, the sweet potato slips showed adequate growth in the plot area, with significantly higher plant vigor in the green manure plots regardless of crop species (Figure 27). The cover crop plots showed reduced plant vigor, likely due to the weed presence since these plots were not tilled prior to planting. The plant vigor was correlated to the sweet potato yield, in that the green manure plots and the control plots, both received tillage prior to planting, produced marketable tubers, while the cover crop plots had no yield at all (Figure 28). Compared to the control plots, plots received green manure, regardless of species, produced 48% or more total tubers. Plots that received cereal rye as green manure resulted in 2.4× of marketable yield compared the control. The weight of the marketable tubers was positively correlated to the marketable yield, with the green manure plots having higher yield compared to control which had lower marketable yield.
At harvest, all tubers were evaluated for possible disease or mice damages. The damages caused by disease or mice all followed the same trend of tuber yield influenced by treatment, with higher damage in the green manure plots.
Conclusion
- The cover crops regardless of species, significantly suppressed weeds before termination.
- No significant effects were observed on the total microbial biomass or soil chemical properties due to the treatments applied. It may take several seasons to observe changes.
- The sweet potato yield however, was 48% higher in plots that had the cover crops incorporated into soil as green manure, and the marketable yield double compared to control plots.
References
- Banks, M.L., Kennedy, A.C., Kremer, R.J. and Eivazi. F. (2014). Soil microbial community response to surfactants and herbicides in two soils. Appl. Soil Ecol. 74:12–20.
- Fierer, N., Strickland, M.S., Liptzin .D., Bradford, M.A., Cleveland, C.C. (2009). Global patterns in belowground communities. Ecol Lett 12:1238–1249.
- Finney, D. M., Buyer, J. S., & Kaye, J. P. (2017). Living cover crops have immediate impacts on soil microbial community structure and function. Journal of Soil and Water Conservation, 72(4), 361-373.
- Frey, S.D., Elliott, E.T., Paustian, K. (1999). Bacterial and fungal abundance and biomass in conventional and no-tillage agroecosystems along two climatic gradients. Soil Biol Biochem 31:573–585.
- Kabir, Z., O’Halloran, I.P., and C. Hamel, C. (1999). Combined effects of soil disturbance and fallowing on plant and fungal components of mycorrhizal corn (Zea mays). Soil Biol. Biochem.31:307–314. Knivett VA, Cullen J (1965) Some factors affecting cyclopropane acid formation in Escherichia coli. Biochem J 96:771–776
- Mendes, I. C., Bandick, A. K., Dick, R. P., & Bottomley, P. J. (1999). Microbial biomass and activities in soil aggregates affected by winter cover crops. Soil Science Society of America Journal, 63(4), 873-881.
- Pan, X., Richardson, M. D., Deng, S., Kremer, R. J., English, J. T., Mihail, J. D., & Xiong, X. (2017). Effect of Organic Amendment and Cultural Practice on Large Patch Occurrence and Soil Microbial Community. Crop Science.
- Sapkota, T., Mazzoncini, M., Bàrberi, P., Antichi, D., Silvestri, N. (2011). Fifteen years of no till increase soil organic matter, microbial biomass and arthropod diversity in cover crop-based arable cropping systems. Agron Sustain Dev.
- Six, J., Feller, C., Denef, K., Ogle, S.M., Sa, J.C.D., Albrecht, A. (2002). Soil organic matter, biota and aggregation in temperate and tropical soils—effects of no-tillage. Agronomie 22:755–775.
- Spedding, T.A., Hamel, .C., Mehuys, G.R., Madramootoo, C.A. (2004). Soil microbial dynamics in maize-growing soil under different tillage and residue management systems. Soil Biol Biochem 36:499– 512.
- Wang, Q. R., Li, Y. C., & Klassen, W. (2007). Changes of soil microbial biomass carbon and nitrogen with cover crops and irrigation in a tomato field. Journal of plant nutrition, 30(4), 623-639.
Educational & Outreach Activities
Participation summary:
A field day took place at Share Life Farms on August 16 2017. Invited speakers, included Dr. Robert Kremer a Soil Microbiologist from the University of Missouri, Dr. Jaime Pinero and Entomologist from Lincoln University, Dr. Reid Smeda A Weed Science Specialist from the University of Missouri, Mr. Tim Reinbott the Director at the South Farm Research Center and Ms. Kathi Mecham an Extension Specialist. Topics included soil health, insect control and pollinator plants, weed control and fertility in organic production systems. In addition to this, results were shared from the sweet potato research both conducted at Share Life Farms and other locations on other aspects of sweet potato production that would benefit farmers such as cultivar differences in terms of yield and quality and the benefits of processes such as curing after harvest. Despite heavy rainfall, the field day attracted more than 40 people representing farmers, industry and government representatives, community gardeners, and students and extension specialists from 17 Missouri towns (Figure 29 and 30). The majority of participants expressed satisfaction in attending the field day, and 96% of them stated their willingness to change their current practices based on what they learned. Brochures were prepared to share with field day attendees, addressing issues related to the research as well as other aspects of organic production. Lastly, an oral presentation of this work was presented at Agronomy Society of America/Crop Science Society of America/Soil Science Society of America annual meeting in Tampa Florida during the Soil Health Symposium, with about 30 agricultural professionals in attendance. The results will also make a core part of my graduate thesis.


Project Outcomes
This research resulted in better understanding of cover crops and how to best incorporate this practice into organic sweet potato production. The farmers appreciated more the use of cover crops during production, and were also motivated to adopt the practice. The results from this study have been shared at meetings and field days, which would likely stimulate the desire to conduct more research on this very important topic. The sustainable use of resources however, has no doubt a huge impact on the environment. Hopefully, in the future we can continue our research on how to reduce losses and increase yield and quality in organic production system.
I have always valued sustainable agriculture but had never conducted any controlled, scientific research on the methods and practices employed in sustainable agriculture until the start of this project. My knowledge and skills of collecting valid, useful data and conducting on-farm research was increased through the course of this project. It was a rewarding experience working with farmers and a huge learning experience for both my advisor and I, as this was our first time to work with cover crops as relating to organic sweet potato production.
Our project was designed to be as grower-focused as possible, which is why we incorporated a measurement of crop growth and yield as influenced by cover crops. I learned so much interacting with Mr. Jim Thomas regarding organic production, it was nice to see things from the famer’s perspective. During the course of the project, we interacted with several farmers and other agricultural research specialists, one thing we learned is that farmers want to see that the methods or technologies being proposed should benefit them not only environmentally but economically. Our project did not clearly demonstrate the benefits derived from using cover crops to enhance soil health, plant health and sweet potato yield, however there were no negative impacts of the cover crops and there were some numerically notable differences between treatments. It may just take several years before effects are clearly seen. Our project also highlighted the need for further study into the relationships between cover crops and long-term soil health, as well as crop performance and tolerance to insect and disease pests.
We would just like to thank SARE for the opportunity given to be able to carry out this study. If this experiment could have been performed for the second year, it would likely allow us to draw a conclusion on the utilizing of cover crop on soil long-term health. Nevertheless, the experience gained from this project will enable us to do an even better job that may have greater impact to our farming community.