Final report for GNC16-229
Project Information
As human populations continue to increase, it is imperative that we develop agricultural production that efficiently utilizes natural resources and ecosystem services. Insects can provide many ecosystem services to agriculture, ranging from pollination to pest suppression, and can be promoted by providing floral resources in the margins of crop fields. However, the effects that these floral plantings have on beneficial insects are not well understood, especially in conventionally managed agroecosystems. Additionally, there is little research on how well commercially available floral plantings establish on-farm, and if they also provide resources for undesired herbivores.
Working with RD Offutt, a large potato grower based in Minnesota, we compared how floral plantings in the margins of conventionally managed potato fields affected pollinator and predator abundance, as well as biological control of Colorado potato beetle (CPB). We also assessed how well floral plantings established, and what impact they had on herbivores. Using sweep net sampling, pollinator transects, and passive trapping, the abundance of various arthropods was measured both in the margins and in adjacent potato fields. Sentinel prey in the form of CPB egg masses were used to determine the level of predation on CPB. We quantified how well floral plantings established by measuring floral species richness and the amount of floral cover in the margins of fields. Field work took place from 2017-2019.
Floral plantings led to significant increases in floral cover and floral species richness compared to unmanaged margins, although there was significant variation between margins. Floral plantings significantly increased pollinator abundance in field margins. Greater floral cover and floral richness were also strongly correlated with increased pollinator abundance. Predator abundance was increased by the presence of floral plantings, but not affected by increasing floral cover. This indicates that while predators benefit from the plantings, it is not due to the increased presence of flowers per se. Within nearby potato fields, floral plantings did not increase the abundance of pollinators or predators, and may act to concentrate beneficial insects more than exporting them to surrounding crops. Sentinel prey egg masses in floral margins were preyed upon more than egg masses in control margins, but the effect was not significant. Egg masses placed in the crop were preyed upon to a much less degree, and the presence of flowers had no impact on predation rate. The abundance of herbivores was unaffected by the presence of floral plantings, either in field margins or in nearby potato fields.
Our results indicate that floral plantings can promote pollinators and predators in commercial agricultural settings, and that floral plantings do provide increased floral cover and richness without promoting herbivores. However, the beneficial effects of floral plantings may not extend into adjacent crops. Floral plantings of this kind appear to function well as a conservation tool by significantly increasing the abundance of many insect taxa in agroecosystems.
Overall Goals
Our overall goals are to determine if floral plantings provide services to growers by: 1) Creating margins with a greater abundance and variety of flowers, 2) Conserving pollinators in agroecosystems, 3) Conserving predators and increasing the predation of key pests in adjacent fields, and 4) Not providing a refuge for pest insects or leading to greater number of herbivores harmful to adjacent crops
Objective 1: Determine how well floral plantings on the margins of agricultural fields establish after planting, and if they result in a greater richness of flower species and a larger area covered by blooms throughout the season. This will allow us to measure if floral plantings actually provide an increase in floral resources, or if growers might be wasting their money on floral plantings that provide no tangible change from margins that are unmanaged.
We will test this objective by conducting floral transects to determine number of flower species present and to quantify the amount of area taken up by blooming flowers (this is a way to quantify both abundance and size of flowers simultaneously). Agricultural margins with and without floral plantings will be compared to assess if floral plantings really do provide additional floral richness and cover.
Objective 2: Determine how floral plantings impact pollinator communities in the margins and in adjacent fields. This will allow to quantify if floral plantings are effectively conserving a greater number or variety of pollinators in agroecosystems.
This objective will be tested by conducting pollinator transects, trapping pollinators with bee bowls, and collecting pollinators from sweep net and pitfall trap samples. The abundance and community composition of pollinators will be compared between margins with and without floral plantings. Additionally, the abundance and community of pollinators in fields adjacent to margins with and without floral plantings will be compared to determine if pollinators are moving into the crops.
Objective 3: Determine how floral plantings affect predator communities and biological control of Colorado potato beetle, the main pest of concern in potato fields. This will help us measure if floral plantings provide an additional ecosystem service by increasing the predation of key pests in fields adjacent to the plantings.
This objective will be tested by collecting predators via sweep net and pitfall trap sampling. The abundance and community composition of the predators will be compared between margins with and without floral plantings, and in the potato crops adjacent to the floral plantings or to unmanaged margins. Additionally, we will place Colorado potato beetle egg masses in the margins themselves, and at the edge of the crop. The number of eggs missing or damaged over a 24 hr period will be compared between floral and unmanaged margins. This will allow us to more directly quantify predation of Colorado potato beetle.
Objective 4: Determine how floral plantings affect herbivore communities in the margins and in adjacent fields. This will allow us to determine if floral plantings are providing an ecosystem disservice by attracting or harboring a greater number or variety of herbivorous or pest insects.
To test this objective, we will use sweep net and pitfall sampling to compare the abundance and community composition of herbivores between margins with and without floral plantings, and in potato crops adjacent to the floral plantings or unmanaged margins.
Research
In order to quantify floral planting success, how pollinator and predator communities were affected, and the level of predation of Colorado potato beetle, we sampled as many fields as possible and sampled throughout the entire growing season. We utilized multiple sampling methods to try to get a comprehensive measurement of beneficial insect communities and attempted to mitigate confounding factors by comparing floral and unmanaged margins within the same field.
Experimental Design
Field work was conducted in central Minnesota, where a large commercial producer of potatoes (RD Offutt) established ~500 acres of floral plantings around 42 of their center-pivot irrigated potato fields in 2015 and 2016. Floral plantings were created using a commercially available seed mixture marketed by Syngenta and Pheasant Forever (Operation Pollinator, Honey Bee and Monarch Butterfly Partnership), consisting of 7 species of grass and 35 species of flowering forbs. Floral plantings were seeded in the unmanaged margins of the fields, in sections ranging from 0.1-5 acres. Between 1-3 of the 4 available corners around the potato fields had been planted with floral seed mixture, depending on the location, while the remaining corners were left unmanaged.
Fields in the study area were on a 3-year rotation of potatoes, corn, and soy or kidney beans. Potato fields were conventionally managed, with aerial applications of fungicide on a biweekly basis, and an in-furrow neonicotinoid treatment at planting. Fields that were planted with potatoes in a given year, had at least one corner margin planted with flowers and at least one margin left unmanaged, and had margins at least 400 meter apart were selected and all accessible margins sampled. To control for variability between fields, floral margins were compared to control margins on the same field. Six fields were sampled in 2017, eight fields in 2018, and six fields in 2019. Sampling occurred approximately once a month from early June-September for a total of five sampling dates per year. In 2018, sampling only took place early June-August (i.e. four sampling dates) due to consistent rain and cold temperatures in September. Sampling took place in the margins of the fields, and inside the fields. Within the potato fields, we sampled at the edge of the crop, and 10, 30, and 50 meters into the field. This gave us a total of 5 sampling locations for each side: Margin, Edge, 10m, 30m, and 50m. For the early June and late September sampling dates we only sampled the margins and did not sample inside the fields because the potatoes had not grown large enough and because the potatoes had been harvested, respectively.
Sampling Methods
Floral Transects: Floral transects were conducted in the margins only, and were used to quantify floral species richness and floral cover. We went to the middle of the margin of the field (at least 20 meters from the edge of the crop in all cases) and ran a tape measurer out 15 meters from the center of the margin. We placed a 1x1 meter square made of PVC at 0, 5, 10, and 15 meters along the tape measurer and identified and counted every blooming flower within the square. All 4 locations were summed to get an overall measurement of the floral composition of the margin. The abundance of each flower species was multiplied by the average area that a flower of that species takes up to get a measure of floral cover. Species richness data was supplemented by doing a visual scan of the margin to ensure no common species were left out of the transect. If a species was seen to be common in the margin, but not found in the transect, the species was recorded and added to the overall richness count.
Pollinator Transects: Pollinator transects were conducted in the margins and at all locations in the crop (edge, 10m, 30m, 50m), and were one of the methods used to quantify pollinator abundance, richness, and community composition. Transects consisted of timed, slow walks through the field or margin, hand netting with a mesh aerial net every bee or syrphid fly that was observed. Netted insects were transferred to a kill jar filled with soapy water. Each transect was timed. Timing would continue until a pollinator was netted, at which point the timer would be stopped. Once the pollinator was inside the kill jar, the timer would be started again. In this way, the time of each transect was measured in "searching" time while eliminating the time it took to handle pollinators. Pollinator transects in the margins lasted for 5 minutes of searching time. Pollinator transects in the crop (edge, 10m, 30m, 50m) lasted for 3 minutes of searching time.
Sweep Nets: Sweep nets were used to collect insects in the margins and all locations in the crop (edge, 10m, 30m, 50m) and were one of the methods used to quantify pollinator and predator abundance, richness, and community composition. Using large, heavy canvas sweep nets, we would slowly walk while sweeping the net through the vegetation in front of us using back and forth pendulum swings. The swings were timed so all vegetation in front of us would be swept with the net once as it was walked over. The sweep net transects consisted of 100 swings total. The contents of the net were then transferred to a plastic bag and placed inside a cooler filled with ice to kill or immobilize the captured insects.
Pitfall Traps: Pitfall traps were used to passively trap ground dwelling arthropods in the margins and all locations in the crop (edge, 10m, 30m, 50m) and were one of the methods used to quantify predator and pollinator abundance, richness, and community composition. Using a golf hole cutter, an area the size of a solo cup was excavated. A 16 oz red solo cup was placed in the hole, and buried to the brim. The cup was then filled a quarter full with soapy water. The trap was left out for between 18 and 24 hours, and the contents collected. Water in the trap was filtered through a square of fine mesh to separate out the captured arthropods, which were then placed in a portable freezer.
Sentinel Prey: Colorado potato beetle egg masses were placed in the margins and at the edge of the crop to directly quantify predation. Egg masses were collected in late June and early July of 2017 and 2018 from potato fields managed by RD Offutt in the same area as our study fields. Potato leaves with egg masses were picked and placed in a portable freezer. Egg masses were selected that were lemon-yellow in color to ensure they were of similar age to one another. After the eggs were frozen, the leaf surrounding the egg mass was cut away, leaving a small square of leaf the size of the egg mass. This was then affixed to a piece of white card stock with double sided sticky tape. The number of intact and damaged eggs was counted and recorded on each of the egg mass cards, and the egg mass cards were then re-frozen. The cards were taken out into the field, and attached to the post of a small fluorescent pink marking flag with a paper clip so they could be easily found again. The egg mass cards were adjusted to be approximately 20-30 cm above the ground, and were left out for 18-24 hrs. In 2017, eggs were placed in the margins of the fields on 2 occasions: August 22-23, and September 20-21, and at the edge of the crop on 1 occasion: August 22-23. In 2018, sentinel prey were placed in the margins of fields and at the edge of the crop on July 27-28 and on August 29-30. In 2019, sentinel prey were deployed during all sampling dates. The egg masses were collected and the number of intact and damaged eggs was counted again.
Identification and Analysis
Insect Identification: Insects and other arthropods from pollinator transects, sweep nets, pitfall traps, and bee bowls were separated out from vegetation, soil, and other debris in the lab. Pollinators from all sampling methods were pinned and labeled, and then identified to family in the case of syrphid flies and bee flies, and to genus in the case of bee species. One exception was honeybees, which were identified to species. Predators were separated into 2 broad groups: those that are known to prey on Colorado potato beetle, and other predators that are not known to eat Colorado potato beetle. Predators were identified to the lowest relevant taxonomic level, often to family, but occasionally to genus or species. Predators known to prey on Colorado potato beetle were more often identified to species or genus. Herbivores were also identified to the lowest relevant taxonomic unit (family). Colorado potato beetle were identified to species and analyzed separately.
Analysis: Results were analyzed using generalized linear mixed effects models (GLMM) with the glmmADMB package in R. The total number of pollinators, predators, or herbivores per sampling location per field per year were summed together to estimate overall abundance for different taxa. This was done both for insects found within margins, and for those within adjacent potato crops. Arthropod abundance, floral cover, and number of eggs missing as were used as response variables, and treatment (presence of absence of floral margins), floral cover, and sampling location as fixed effects. Field identity and sampling date were used as random effects to account for abiotic and landscape variability between fields, and repeated sampling throughout the year respectively. Data were overdispersed with high variance, and negative binomial was selected as the distribution with a log link function. In select cases with richness data, a Poisson distribution was used instead. Models of best fit were selected via comparison of AIC values.
Objective 1: Establishment of floral plantings
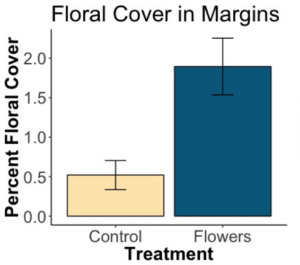
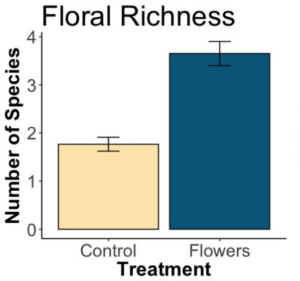
We wanted to determine if floral plantings resulted in more floral cover and a greater number of species of flowers present in margins compared with unmanaged margins. We found that floral plantings led to significantly increased area covered by blooms (a greater number of, and larger, flowers). Additionally, floral plantings led to significantly more species of flowers, with almost twice as many species present in floral margins on average.
There was a significant effect of treatment on floral cover (p=1.0e-11). The x axis refers to whether or not flowers were planted in the margins. “Control” means no flowers were planted, and the margins were left as they were. “Flowers” means the margin was sown with a flower seed mixture. The y axis quantifies how much area is covered by blooms as a percentage of total measured area, and accounts for both the number of flowers and the size of the flower. Floral margins had significantly more area covered by blooms throughout the season and across fields.
There was a significant effect of treatment on floral richness as well (p=2e-16). The x axis refers to whether or not flowers were planted in the margins. “Control” means no flowers were planted, and the margins were left as they were. “Flowers” means the margin was sown with a flower seed mixture. Floral margins had significantly more species of flowers present throughout the season and across fields.
Objective 2: Pollinator Conservation
The main purpose of floral plantings in agroecosystems is to provide resources to beneficial taxa, like pollinators. We wanted to determine if these floral plantings did indeed increase the abundance, or change the community composition of pollinators. Additionally, we measured pollinator presence in crops adjacent to the margins to determine if a greater number of pollinators moved into fields next to floral plantings.
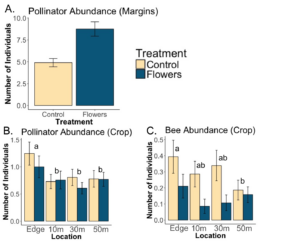
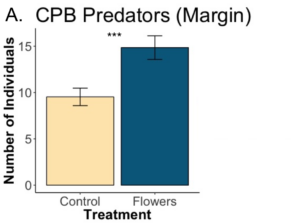
We found that a significant effect of floral plantings on the abundance of pollinators in the margins (p=5.6e-07). Additionally increasing floral cover had a significant and strong effect on pollinator abundance (p=0.0016). Floral plantings overall increased the abundance of pollinators, and floral plantings that led to greater amounts of floral cover increased pollinator abundance to a greater degree. Floral plantings in the margins had no effect on overall pollinator abundance in nearby fields, but did lead to significantly fewer number bees in the adjacent field (p=). Edges of potato fields had significantly more pollinators than locations further into the fields.
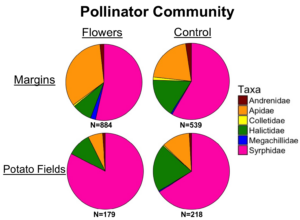
Pollinator communities had notable differences, with floral margins having significantly greater number of bumblebees, and bees in the genera Melissodes and Megachile. On a family level, floral margins had proportionately greater numbers of Apidae.
A. There was a significant effect of treatment on pollinator abundance in margins (p=5.6e-07). B. Within crops, there was no significant effect of treatment on overall pollinator abundance (p=0.47). C. Floral margins led to significantly fewer bees in nearby fields (p=0.0066). Letters denote significant overall difference between locations (p<0.05).
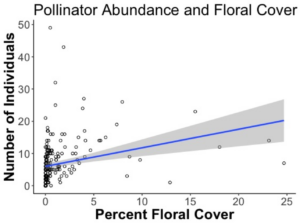
Pollinator abundance was also significantly increased in margins with increasing floral cover (p=0.0016), regardless of if they were seeded with floral plantings or not.
The community composition of pollinators was different between floral margins and control margins, although pollinators communities were broadly similar, with syrphid flies making up the largest part of the community, followed by bumblebees, honeybees, and species within the genera Melissodes and Lasioglossum. In adjacent crops, the community was dominated by syrphidae.
Objective 3: Predators and Biological Control of Colorado Potato Beetle
To determine if floral plantings provided benefits other than conserving pollinators, we wanted to measure predator abundance, richness, and community composition like we did for pollinators, as well as how predation of Colorado potato beetle was impacted by the presence of floral plantings.
We found that floral plantings led to a significant increase in the abundance of predators overall (p=0.0093). Additionally we found that floral plantings significantly increased the abundance of predators that are known to prey on Colorado potato beetle (p=). Within potato crops, there was no significant effect of treatment on overall predator abundance or CPB predator abundance.
Predator communities were largely similar within field margins, although flowers did lead to greater numbers of predatory hemipterans and a greater number of opiliones. Predator communities within potato fields were similar between treatments, although varied significantly from the community found in margins, with far more coleopterans. These results suggest that margins may not be exporting many predators to nearby crops, considering how different the communities are.
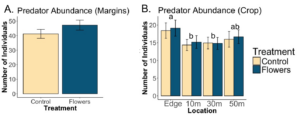
A. Flowers significantly increased the abundance of predators overall in field margins (p=0.0093). B. Flowers in field margins did not affect predator abundance within fields (p=0.86), although there was a significant effect of sampling location on predator abundance. Letters denote significant differences in overall predator abundance between locations (p<0.05).
Flowers significantly increased the richness of known predators of Colorado potato beetle in field margins (p=4.3e-08).
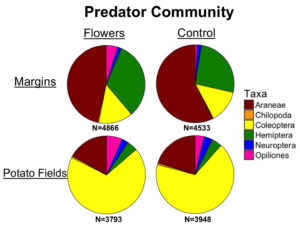
Communities of predators were largely similar within margins, but in-crop communities had significantly more coleoptera and far fewer araneae.
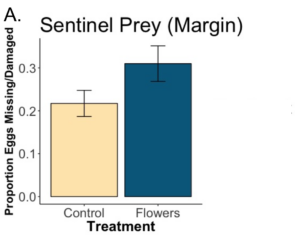
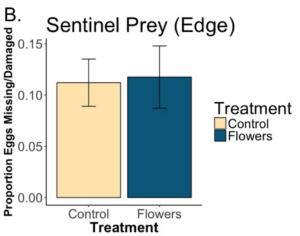
We found that while the predation of Colorado potato beetle eggs was greater within floral plantings compared to control margins, there was not significant effect (p=0.31). While the flowers are attracting predators that will consume Colorado potato beetle, it appears that they might not be preying on Colorado potato beetle to a greater degree. We found no difference in predation when we placed egg masses in the edge of the crop (p= 0.95). Our data currently indicate floral plantings do attract more predators of Colorado potato beetle, but biocontrol services do not extend into the adjacent crop.
A. Floral plantings did not lead to significantly more Colorado potato beetle (CPB) eggs consumed in the margins of fields (p=0.31). B. Floral plantings also had no effect on eggs consumed at the edge of crops (p=0.95). The x axis refers to whether or not flowers were planted in the margins. “Control” means no flowers were planted, and the margins were left as they were. “Flowers” means the margin was sown with a flower seed mixture. The y axis refers to the number of sentinel CPB eggs consumed or found to be missing after 24 hrs. When masses of CPB eggs were placed in the margins, far more were consumed when flowers were present.
Objective 4: Herbivore Communities
While floral plantings may provide benefits by promoting predators and pollinators, they also have the possibility to provide resources for detrimental pests and other herbivores as well. We wanted to measure the herbivore and pest community to ensure floral plantings were not providing ecosystem dis-services.
Floral plantings did not increase the abundance of herbivores in field margins (p=0.9), or in adjacent potato crops (p=0.9705)
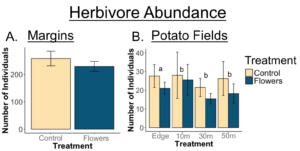
A. Herbivore abundance in margins was not affected by treatment (p=0.9). B. Herbivore abundance in nearby potato fields was not affected by treatment (p=0.9705), but the edge of the crop had significantly more herbivores than other sampling locations. Letters denote significant differences in overall herbivores abundance between locations (p<0.05).
Educational & Outreach Activities
Participation summary:
I presented my current research results at the Wisconsin Fresh Fruit and Vegetable conference in Wisconsin Dells in January 2018. Around 30 growers, researchers, and other presenters attended the talk.
Project Outcomes
This project has the potential to be very influential to future conservation efforts in agroecosystems and ecosystem service management, leading to greater agricultural sustainability.
Crop production takes up some 12% of all ice-free land. With so much land being used to produce food, a key part of sustainable agriculture is mitigating environmental impacts and conserving species within agroecosystems. One of the primary ways to do this is by creating stable habitat within agroecosystems to provide a constant source of food, resources, and refuge in what is normally a frequently disturbed and highly ephemeral landscape. While floral plantings are but one of many ways to accomplish this goal, they are commonly used. Yet their effects on beneficial species are poorly understood. In many instances, growers will plant margins with a floral seed mixture, but have little to no empirical data on what results can be expected. This is especially true of very large floral plantings in the margins of conventionally managed fields. Even less well understood are the ways that these floral plantings might provide tangible benefits to the grower in the form of ecosystem services like pest suppression and pollination.
My project seeks to help answer many of these questions were there is currently a lack of knowledge. By working on large, conventionally managed farms where floral plantings have been established, we can determine how these plantings perform for their intended goals of conservation in a real world setting. Do they actually provide a stable source of resources in agroecosystems that helps conserve species, or might growers be wasting their money? This project clearly deals with environmental benefits to farmers, but it also touches on economic issues and even some social benefits. If plantings do provide other ecosystem services and decrease the need for inputs such as pesticides, growers would have clear, tangible economic incentives to plant flowers. There are also potential social benefits such as good PR for growers that plant flowers to promote beneficial insects, and improved aesthetics on the farm. While this project directly deals with very important environmental aspects of sustainable agriculture, there is also a definite possibility of economic and social benefits to growers. Understanding how floral plantings promote pollinators, predators, and herbivores is critical to understanding all of these elements.
My knowledge and awareness of sustainable agriculture has increased substantially, and my views on it have become deeper and more nuanced. When I first started working on this project and beginning my research, I was mostly approaching sustainable agriculture from the view of an entomologist. I focused on how insects can provide benefits to growers and how growers could promote said in insects. While I knew sustainable agriculture encompassed many other things, I hadn't really given them much thought. Since then, I have learned quite a bit more, both directly and tangentially related to my research.
Having conducted research on very large, conventionally managed farms, my perspective on what counts as progress towards sustainable agriculture has shifted. Sustainable agriculture doesn't have to be practiced by only small-scale local or organic growers. Even large producers can, and are, incorporating sustainable practices into what they do. Sustainability can be compatible with conventional management (albeit, often with more difficulty), and more integrated practices are often more efficient and cost effective, although more knowledge-intensive. Additionally, by conducting my research on active farms, I've learned just how many factors need to be considered. Soil type, pathogens, fertilizer applications, dealing with fungi, concerns about weeds, aesthetics and public perception, even state regulations about moving around roads. All of these influence my own research into floral plantings and beneficial insects, and these are all things that must be considered by growers who may want to implement this practice.
In addition to learning about all of the different factors that can influence my own project, I've learned more about other projects through SARE and other programs. The range of different topics and approaches is huge, and has made me appreciate just how much there is to research and learn in order to produce food and fiber in more sustainable ways.
In total, I've learned a huge amount, and I'm sure I will continue to learn as my career continues.