Final report for GNC23-370
Project Information
The overuse of antibiotics in agriculture has led to a public health crisis in increased antibiotic resistance and toxicity in our managed livestock and agricultural land. Thus, research into how antibiotics impact fundamental aspects of animal husbandry is essential to address this crisis. Western honeybees (Apis mellifera) are globally managed for their pollination services of our agricultural crops, contributing hundreds of billions of dollars to the global economy. The continual rise in annual commercial honeybee colony losses have raised major concerns about the future of agricultural crop yields and food security. The reliance on these important pollinators demands a constant evaluation of current honeybee management strategies and the mechanisms in which they may support colony health.
One such management strategy is the treatment of colonies with oxytetracycline (OTC), an antibiotic approved for livestock use, a strategy that has historically been the best strategy to prevent or treat devastating bacterial foulbrood diseases in colonies. OTC has been shown in previous literature to be detrimental to individual honeybee health and behavior in a lab setting. For example, fanning is an essential behavior vital to the success of honey bee colonies because it is thermoregulatory and maintains internal colony temperatures at 95°F for proper brood development. My previously published work, obtained in a laboratory setting, demonstrates that fanners treated with OTC are less likely to fan and are unable to quickly respond to heat stress. Such a disruption could have drastic effects at the colony level; however, whether the detrimental effects of OTC on individual honey bees can scale to affect whole colony health in a field realistic setting remains unclear. Understanding these dynamics is critical to properly assess OTC as a honey bee disease management strategy. Therefore, the objective of this study was to investigate the effects of preventative OTC treatment on honeybee colonies in a field-realistic setting.
Our central hypothesis for this NCR-SARE project was that OTC treatment would impact colony performance and health over the beekeeping season. To test this hypothesis, we performed a field-based study in partnership with a local Wisconsin beekeeper, tracking the health and thermoregulation of 24 honey bee colonies, half of which were treated with OTC following FDA guidelines, from May through October 2024 in Wisconsin. We found support for our hypothesis that OTC impacts colonies: OTC treated colonies were more productive than non-treated colonies, likely because OTC treated colonies reared a larger workforce to collect more food earlier in the season. Our results provide a critical, nuanced examination into the benefits and risks of OTC treatment on honey bee colonies and have broad implications for the future of honey bee management. This research, which is the first NCR-SARE graduate student project that investigated OTC effects on honey bee colonies, has already been used to educate Wisconsin beekeepers on the pros and cons of OTC use, increasing local beekeeper awareness of this important management strategy. Furthermore, this research has been presented at international science conferences, won scientific talk competition, and is currently being prepared for scientific publication; thus, this research has already contributed much to our knowledge on the relationship between OTC and honey bee health both scientifically and locally.
These data are currently being prepared to be submitted to a scientific journal for peer review and publication. Once available, we will provide the publication link!
Because of research exposing the effects of antibiotic use on honeybee health, in 2017, the Food and Drug Administration’s (FDA) Center for Veterinary Medicine passed the Veterinary Feed Directive (VFD) initiative, which requires beekeepers to obtain a veterinary prescription to purchase antibiotics like OTC. However, many beekeepers have expressed concern that this initiative will lead to diseases like American Foulbrood to resurge and cause increased colony loss. Thus, there appears to be a lack of proper education on the purpose and usage of antibiotics like OTC and all the effects they have on honey bees. This lack of clear education may continue to fuel misunderstanding and off-brand use by beekeepers, and honey bee livelihood and even environmental health may continue to worsen as a result. Therefore, the following outcomes are expected on completion of my project:
Learning outcomes (through talks and presentations):
- Honey bee researchers will better understand the effect of OTC on colony health, productivity, and thermoregulation in a field-realistic setting
- Beekeepers will understand the benefits and risks of prophylactic OTC use for colony health and behavior and for the larger environment
Action outcomes (after attendance of talks and presentations):
- Beekeepers will slow or stop overreliance on antibiotics except when treating active diseases
- Beekeepers will be more aware of what factors can impact their colony management success
- Beekeepers will experience reduced financial strain in replacing their colonies annually
- Beekeepers will reduce their personal contributions to antibiotic pollution in the environment
Cooperators
- (Researcher)
Research
We have provided several figures and photos to aid in illustrating our methodologies. Please refer to the cited figures and/or photo when referenced in-text. Additionally, throughout this progress report, we use terminology that is common in beekeeping and honeybee research that may not be clear to anyone outside the field. Please see the table below for the definition of these terms. We hope this table clarifies unfamiliar terminology.
|
Terminology |
Broad Definition |
|
Deep |
A wooden box that contains wooden frames where bees can build their comb, store their resources, and rear their young. Single deep colonies have 1 of these boxes, while double deep colonies have 2, etc. Typically, beekeepers keep their colonies to 2-3 deeps. |
|
Frame |
A wooden support that represents a single “slice” of a colony. Bees build their comb onto these supports. There were 8 frames per deep in our study. |
|
Super |
A wooden box that is commonly half the size of a deep that beekeepers place on top of colonies during nectar flows. These boxes act as extra storage reserves for bees to store their excess honey and are harvested by beekeepers at the end of the nectar flow. |
|
Porch |
Another term for the entrance of the colony. The porch is where honeybee foragers will come and go as needed, and where fanners and guards can be seen performing their jobs. |
|
Cells |
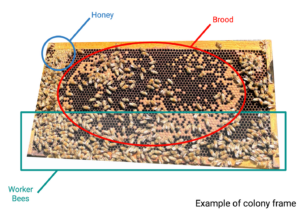
Refers to the holes within the honeybee comb where resources are stored, and honeybee offspring are reared. |
|
Brood |
Another broad word for offspring or young. A queen will lay an egg inside of a cell. These eggs hatch into larvae and are reared by worker bees inside of the comb. |
|
Uncapped brood |
Honeybee offspring that are in the earliest stages of development. During this time, because workers are directly feeding and caring for brood, their cells are kept open for the workers to access them. |
|
Capped brood |
Honeybee offspring that are in the final stages of development. Cells are covered in a wax cap covering to protect the young bees as they undergo their final metamorphosis into adult bees. |
Field site set up
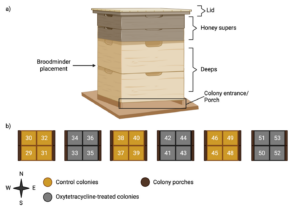
On May 1st, 2024, twenty-four honey bee (Apis mellifera linguistica) colonies from Concord Farms were installed at a field site located in Oconomowoc, Wisconsin (43° 6' 24.1662", -88° 37' 18.8904"). Colonies were kept on six pallets with four colonies per pallet (Figure 1; Photo 1). Colonies were originally set up as one deep with 8 frames, and all colonies were given a second deep on May 16th for the rest of the study. We chose to limit colonies to two deeps to control for colony size as a strong factor in determining colony success. On May 17th, all colonies were assigned a numbered tag (29-53) to keep track of colony identity (Figure 1), and the lids of colonies designated for antibiotic treatment were painted yellow and marked by bright orange duct tape. We chose to alternate treatment regimen by pallet: whole pallets consisted of colonies that received the same treatment to minimize potential contamination between colonies on the same pallet (Figure 1). On May 24th, we installed BroodMinder-T2SM internal hive temperature sensors in the center of twenty of these colonies (colonies 30, 31, 34, and 35 did not receive temperature monitors) in-between deeps to continuously collect internal hive temperature data. Ryan Stern of Concord Farms managed these colonies as necessary to maintain as field-realistic settings as we could. Ryan noted when and how he managed the colonies after the field season ended, and we will control for these management moments in our final analysis for journal publication by incorporating them as a random effect into our statistical models. Additionally, colonies 29, 38, and 48 (all of which were control colonies) died in the middle of our field experiment. We therefore removed these colonies from our final analyses, bringing our total colony number to 21 (9 controls, 12 treatment). Colonies remained in the same configuration, orientation, and location through the duration of the experiment.
Oxytetracycline (OTC) treatment regimen
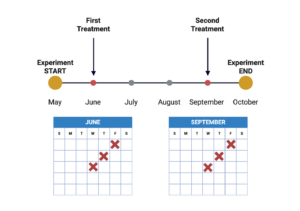

Preventative oxytetracycline treatment was given in two rounds, once at the beginning of the season (June) and again near the end of the season (September), as directed by FDA guidelines (Figure 2). On June 3rd, all colonies received their first round of treatment, where half of the colonies received 28.35g of powdered sugar (application group referred as “control”) and the other half received 28.35g of powdered sugar and 200mg of oxytetracycline hydrochloride (Oxytet® Soluble), which was procured from a local veterinarian (application group referred as “antibiotic”). 200mg per colony is the approved oxytetracycline dosage for whole honey bee colonies by the FDA. Both applications were dusted at the edges of the frames of the top deep of all colonies (Photo 2), and the regimen was repeated for a total of 3 doses with 5-day intervals in-between across the month of June and again in September, as advised by FDA guidelines (Figure 2).
Fanning observations
To assess the behavioral component colony behavioral thermoregulation, we counted the number of fanners present on the entrance of all colonies three times per week. Fanners are a specialized worker whose job is to thermoregulate the colony by rapidly flapping their wings to generate airflow (Egley and Breed 2013; Lindauer 1955). Although fanners fan throughout the colony, they are easily identifiable on the porch of the colony with a distinct posture: they stand still with their abdomens are angled downward and heads facing in the direction of the colony entrance and actively fan their wings (Egley and Breed 2013).
We noted where fanners were standing on the porch (southern side, northern side, or center of the porch) and measured the porch surface temperature per porch location using a temperature gun (Fluke® 568 Infrared Thermometer). We also used a lux meter (EXTECH 401025: Light Meter) to measure light intensity on the porch, as heat and light are interlinked, and we were interested in the role light may play in triggering fanning behavior. We recorded the time when we made each of these observations and measurements, although we chose to perform most of these measurements in the afternoon at peak temperature of the day as we were interested in measuring peak fanner output. During these fanning observations, we also downloaded data via Bluetooth from all Broodminder-T2SM monitors inside colonies. All data was written on data sheets that were then transcribed into an Excel file that was saved to Marquette University’s cloud-based Microsoft SharePoint OneDrive service for long-term storage.
Hive inspections

Twice per month, all colonies were subjected to a full hive inspection (Photo 3). Colonies and honey supers, if present, were first weighed using a luggage scale (CONAIR TravelSmart©). We then opened colonies and estimated the proportion of workers, pollen, honey, and brood that covered each frame (See Figure 3 for example frame and estimation). These estimations were repeated for both deeps for all colonies. The data collected from hive inspections, which are data that professional beekeepers also collect, helps to characterize colony composition in several different ways. For one, colony weight can be a direct measurement of the amount of food and bees inside the colony, but it can also indicate overall colony changes. For beekeepers, such inspections are critical in detecting changes in colony composition that could contribute to changes in colony weight. All these methodologies for measuring colony strength in honey bees are standard within the field (Delaplane et al. 2013). All data was written on data sheets that were then transcribed into an Excel file that was saved to Marquette University’s cloud-based Microsoft SharePoint OneDrive service for long-term storage.
Statistical Analysis of Field Data
All data analysis and visualization were performed in the program R (v. 4.5.1) and RStudio (v. 4.3.2). We removed colonies that had died in the middle of the season (colonies 29, 38, and 48) during our analysis to avoid confounding factors from influencing our results. It is noteworthy to highlight that all colonies that perished during the study were colonies from the control group. For data analysis, we created several generalized linear mixed models with the appropriate distributions and tested all models using an ANOVA Type II Wald Chi-Square Test, and post-hoc comparisons were conducted using each model’s estimated marginal means with a Benjamini-Hochberg correction to control for false discovery rate across all our tests. When assessing differences in the thermoregulatory capabilities of colonies between treatment groups, we downloaded local climate data from the National Oceanic and Atmospheric Administration (NOAA) Watertown Municipal Airport, WI, U.S. station #USW00054834 (43° 10' 0.012", -88° 43' 0.0114"), which was the closest NOAA station to our field site. We paired this NOAA database with every timepoint gathered in our Broodminder data as well as our fanner observation data to model how climate data impacted our results.
Worker gut sample processing & DNA extraction
OTC is known to disrupt the honey bee gut microbiome, but it has not been tested whether OTC treated at the colony level meaningfully disrupts worker microbiome. Therefore, to begin to assess the impact of OTC on the honey bee gut microbiome, throughout the duration of the field study, whole honeybee workers were sampled indiscriminately from directly inside the colonies before, in between, and after treatment. These samples were then stored at -80ºC once back in the lab for later processing. Sampling occurred three times in June, once in July, three times in September, and once in October, totaling 9 different collection events spread across the summer and providing a snapshot of their gut microbiome at those times. Workers that were sampled during the treatment months of June and September were then randomly selected to be processed to minimize batch effects. After thawing on ice, whole guts of every individual honeybee collected in a sample were dissected into 1mL phosphate buffer solution using sterile technique following the guidance of previous studies (Zheng et al. 2018).
We performed all dissections under anaerobic conditions to preserve microbial viability for later plating assays. After dissection, we homogenized guts using a sterile pestle and vortex in 1mL PBS, then we pipetted an additional 1mL PBS to all samples. We then split the sample into two 1mL Eppendorf tubes, one for storing as a glycerol stock for later plating assays and the other for DNA extraction. All samples were kept frozen at -80ºC until all bee samples from June and September were dissected and split.
We then used a Qiagen PowerSoil Kit to extract DNA from gut samples from all colonies and at all 6 time points. We randomly selected and pooled 5 bee guts from the same colony collected at the same time point to create “colony-level” samples, and we also extracted individual bee guts. DNA samples were then all sent to Argonne National Labs for Illumina MiSeq 16S rRNA amplicon sequencing.
16S rRNA sequencing & analysis
We are currently awaiting sequencing data. Once received, data will be run through the publicly available DADA2 pipeline in R (Callahan et al. 2016) to assign bacterial taxonomy in our gut samples before running the appropriate statistical analyses to identify gut bacteria present in each sample. Worker microbiomes will be compared across colony and individual samples, treatment groups, and time of season sampled to parse out how the microbiome was affected during treatment duration and whether the microbiome recovered afterwards.
Referenced literature for methods:
Callahan, Benjamin J., Paul J. McMurdie, Michael J. Rosen, Andrew W. Han, Amy Jo A. Johnson, and Susan P. Holmes. 2016. “DADA2: High-Resolution Sample Inference from Illumina Amplicon Data.” Nature Methods 13 (7): 7. https://doi.org/10.1038/nmeth.3869.
Delaplane, Keith S., Jozef van der Steen, and Ernesto Guzman-Novoa. 2013. “Standard Methods for Estimating Strength Parameters of Apis Mellifera Colonies.” Journal of Apicultural Research 52 (1): 1–12. https://doi.org/10.3896/IBRA.1.52.1.03.
Egley, Rachel L., and Michael D. Breed. 2013. “The Fanner Honey Bee: Behavioral Variability and Environmental Cues in Workers Performing a Specialized Task.” Journal of Insect Behavior 26 (2): 238–45. https://doi.org/10.1007/s10905-012-9357-1.
Lindauer, M. 1955. “The Water Economy and Temperature Regulation of the Honeybee Colony.” Bee World 36 (5): 81–92. https://doi.org/10.1080/0005772X.1955.11094876.
Zheng, Hao, Margaret I. Steele, Sean P. Leonard, Erick V. S. Motta, and Nancy A. Moran. 2018. “Honey Bees as Models for Gut Microbiota Research.” Lab Animal 47 (11): 317–25. https://doi.org/10.1038/s41684-018-0173-x.
We have provided graphed data to illustrate our results. Please refer to the referenced figure uploaded as part of this report when cited in the text of this results section.
OTC treatment alters end-of-season colony resource composition
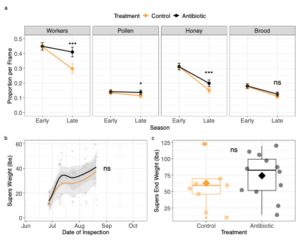
First, when modeling what factors predicted proportion of resource found on a given frame of a colony, we found a significant three-way interaction between treatment, resource type, and season, demonstrating that OTC effects significantly depended on time of season, and those effects differed by the resource type (Figure 4a; GLMM, X2 = 8.8324, p = 0.0316). Differences emerged only in the late season: OTC-treated colonies had higher average proportion of workers (z-ratio = -4.732, p < 0.0001), pollen (z-ratio = -2.023, p = 0.0430), and honey (z-ratio = -3.141, p = 0.0017) of frame coverage compared to control colonies (Figure 3a). Specifically, in the late season, OTC colonies had on average 41.2% coverage of worker bees on their frames while control colonies only had 29.6%. Control colonies had on average 11.3% of pollen stores on their frames, while OTC colonies had 13.6%. Moreover, OTC colonies had 19.7% of their frames covered in honey, while control colonies only had 15% honey coverage. Despite these differences in honey stores in colony deeps, we found no significant differences in the amount of excess honey the colonies produced in honey supers (Figure 4b & 4c; GLMM, X2 = 1.001, p = 0.317). This result could be due honey super weight being highly variable, as upon examination of average honey super end weights (Figure 4c), OTC-treated colonies tended to have heavier honey supers (45.86 ± 30.46lbs) compared to control colonies (34.03 ± 29.43lbs). Regardless, across all colonies, we found that proportion of resource stored in frames significantly depended on time of season (GLMM, season main effect; X2 = 285.64, p < 0.0001) and the type of resource (GLMM, resource main effect; X2 = 27726.96, p < 0.0001).
OTC treatment has no effect on overall colony weight gain
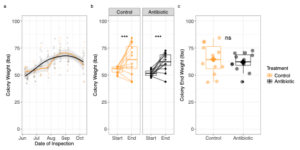
Treatment had no effect on colony weight gain the course of the summer (Figure 5, LMM, X2 = 0. 0322, p-value = 0.8347). OTC-treated colonies weighed on average 60.04± 9.96 lbs while control colonies weighed on average 59.28 ± 10.82 lbs at the end of the season (Figure 5b & 5c). Seasonality significantly affected weight gain, which was expected for healthy colonies (Figure 5a; LMM, X2 = 9.4050, p = 0.002164). However, we found no interaction between treatment and time of season (LMM, X2 = 0.6638, p-value = 0. 415). Additionally, all colonies gained a significant amount of weight when comparing the start and end of season weight (Figure 5b; Friedman’s Test, X2 = 0.17, p = 0.00427), but they all gained the same amount of weight regardless of treatment, as they weighed similarly at the end of the season (Figure 5c; Welch’s two sample t-test, t = 1.8304, p = 0.08736).
OTC treated colonies maintained their internal colony temperature regardless of ambient climate

When modeling which factors impacted internal colony temperature, we found a significant interaction between treatment and ambient wet bulb temperature (Figure 6, X2 = 162.4418, p < 0.0001), demonstrating that OTC treatment significantly influenced colony temperature depending on ambient climate. Specifically, internal colony temperatures of control colonies increased by 0.267°C for every 1°C increase in wet bulb temperature, while OTC-treated colonies experienced an increase of only 0.221°C per 1°C increase (Figure 6b). Namely, once ambient temperatures rose above 18.06°C, the internal temperature of control colonies rose 0.04°C faster than OTC-treated colonies (Figure 6b). These results suggest that the internal temperature of control colonies were more influenced by ambient climatic temperatures, and OTC colonies were able to maintain constant internal colony temperatures in response to rising ambient climate temperature.
Fanning output is dependent on thermal climate and not OTC treatment
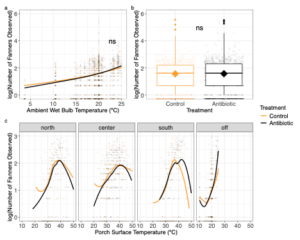
As expected, the number of fanners we saw at colony porches was significantly influenced by ambient wet bulb temperature, as the number of fanners observed increased as climatic temperatures increased (Figure 7a; X2 = 38.4376, p < 0.0001). Specifically, for every 1°C increase in ambient wet bulb temperature, we observed an 11.3% increase in the number of fanners across all colonies (Figure 7a). We found a significant interaction between porch temperature and porch location in driving fanning output (Figure 7c; GLMM, X2 = 44.7118, p < 0.0001). Namely, compared to the northern region of the porch, we were more likely to observe fanners in the porch center and rarely saw fanners off the porch and in the southern region of the porch when ambient temperatures were more reasonable (Figure 7c). However, when porch temperature climbed, there was more steep increase in the number of fanners observed at the southern porch compared to all other regions (Figure 7c). Furthermore, the number of fanners fanning off the porch and at other locations around the outside of the colony increased by 16% per 1°C increase in ambient climate temperature (Figure 7c). OTC treatment did not affect fanning output (Figure 7b, X2 = 0.3217, p = 0.5706). Interestingly, across all colonies, we saw number of fanners significantly drop off after porch surface temperatures reached over 40°C, suggesting that there may an upper thermal limit when fanners will engage in fanning.
Educational & Outreach Activities
Participation summary:
These data have been presented to the local scientific community several times. First, Justine has presented preliminary results as part of her annual graduate student seminar to Marquette University's Department of Biological Sciences (2025GradSeminar). Additionally, she competed in Marquette University Graduate School Student Poster Competition using some of the data gathered from this project (Nguyen2025GradPosterComp).

This research has also been presented to the international science community: Lead investigator Justine presented a talk on this work at the 2025 Joint Annual Meeting of the Entomological Society of America in Portland, Oregon. Justine presented the data (2025EntSocTalk) as part of the 10-Minute Student Competition for President’s Prize in the Apiculture section, where she won first place (Photo 4 & 5). These data are currently being prepared to be submitted as a scientific article to journals like Journal of Apiculture Science or Journal of Economic Entomology once sequencing data has been obtained and analyzed.


These data have been discussed with various visitors to the Cook lab at Marquette University. Additionally, Justine has presented the results gathered from this project to two local Wisconsin beekeeping associations (Brown County Beekeepers and North Shore Beekeepers Association). Attendees took an anonymous pre- and post-presentation survey via a Google Form. All attendees surveyed were either hobbyist beekeepers or interested in becoming hobbyist beekeepers. A summary of pre- and post-presentation survey question related to this project is below.
Pre-Presentation:
- How knowledgeable would you rank yourself in terms of your knowledge on the usage of antibiotics for treating honeybee colonies? (Rate from 1-5)
- Average response: 2.31
- How concerned are you about honeybees being exposed to antibiotics, pesticides, and/or pollution in their environment? (1 - I haven't thought about it at all; 5 - I currently take action to reduce chemicals in our environment)
- Average response: 3.41
- If you keep honey bee colonies, do you use antibiotics (ex: Terramycin, Tylosin, etc) in your colonies?
- 21 out of 22 pre-presentation responses was “No, I don’t use them/haven’t used them at all”
- If you answered no in the previous question, please feel free to share why you choose not to use antibiotics in your colonies.
- Responses ranged from “don’t see a need”, “no access”, “have not looked into usage”, “keeping hive clean of synthetic treatments”
Post-Presentation:
- How satisfied were you with the presenter's ability to communicate scientific information? (1 - Not satisfied at all - they did a poor job; I learned nothing; I couldn't understand what they talked about; 5 - Very satisfied; they did very well and I understood and learned a lot!)
- Average response: 4.86
- How satisfied were you with the presenter's presentation skills (talking pace, organization, visuals)? (Rate from 1-5)
- Average response: 4.71
- How satisfied were you with the presenter's ability to answer questions? (1 - never answered questions directly or entirely; 5 - they always answered questions clearly)
- Average response: 4.86
- How likely are you to recommend someone listen to the presenter speak on a honeybee-related topic? (Rate from 1-5)
- Average response: 4.6
- After seeing the presentation, how concerned are you about honeybees being exposed to antibiotics, pesticides, and/or pollution in their environment? (1 - don't care, won't be changing my behavior; 5 - I WILL take action to reduce them in our environment)
- Average response: 3.5
- After seeing the presentation, if you're a beekeeper or planning to become a beekeeper, how likely are you now to use antibiotics?
- Majority of responses answered, “Will only use them when there is an active disease outbreak”
- Selected anonymous comments from attendees on presentation:
- “Glad an institution is doing this research in my own backyard and interested in its direction.”
- “I am very new, and it’s just good for me to learn all these different topics and information about bees. I never realized this was a thing.”
- “Justine was very informative, interesting presentation”
- “I am interested to hear how the gut biome research turns out and how it is/will be done.”
- “Very interested, knowledgeable speaker! Enjoyed her and the presentation!”
Project Outcomes
Previous literature suggests that prophylactic antibiotic treatment is an overall detriment to individual honeybee health, as it increases mortality, disease susceptibility, and gut dysbiosis. However, because prophylactic antibiotic treatment is given at the colony level, it is difficult to replicate how much antibiotics individual bees consume in the lab. Thus, the concentration of antibiotics given for lab-based studies may not be necessarily relevant at the colony level, where the consumption of antibiotics per individual may be sparse. Furthermore, in response to rising concerns about the spread of antibiotic resistance and its detriments on honeybee colonies, many beekeepers have adopted an all-or-nothing response, where they either continue to misuse antibiotics in their colony or stop using them altogether. As a result, many beekeepers appear to be unaware of the adverse and beneficial effects antibiotics have on honeybee livelihood.
Altogether, our results provide a nuanced view into the effect of OTC on colony dynamics. It is essential to continue to investigate current honey bee management strategies in the face of rapid honey bee population declines. Such examinations are essential to properly inform beekeepers of the pros and cons of different strategies and protect them from large financial losses on their beekeeping operations. Our study has already been used to inform beekeeping communities on the nuanced advantages and disadvantages of using antibiotics and bridge gaps in their understanding of honey bees and the biology of their diseases. This work thus contributes a nuanced view into OTC treatment as a viable strategy in honey bee colony management strategies, and more thorough, field-realistic studies like this project will be required to draw accurate conclusions about strategies if we are to continue to rely on these important pollinators for our food security.
This project will contribute to future agricultural sustainability by 1) providing a holistic assessment of FDA approved dosages of antibiotic use on honeybee colonies in Wisconsin and communicating our results to the broader scientific community and 2) educating beekeepers on the benefits of appropriate antibiotic usage and dangers to its misuse. This project will improve the economic viability of beekeeping by potentially reducing number of colonies lost to foulbrood diseases and increasing overall honey production. It may also reduce unnecessary antibiotic misuse when beekeepers are equipped with the knowledge gained from this project. This project will also potentially remove biases that are present within the beekeeping community surrounding the usage of “chemicals” like antibiotics in their colonies.
Our results characterize the effects of OTC treatment on several honey bee colony health metrics within ecologically and agriculturally relevant contexts, filling a crucial gap in our understanding of the realistic effects of oxytetracycline on bees. In summary, we found that preventative OTC treatment on whole honey bee colonies did alter multiple aspects of colony health in a seasonally dependent, nuanced manner, lending support for our hypothesis. Yet, our findings contradicted our original predictions that OTC treatment would negatively affect colony health overall; instead, OTC treatment appeared to have a slight beneficial effect.
First, OTC treated colonies were more productive than control colonies, rearing more workers to collect and store more pollen and nectar by the end of the season (Figure 4). Curiously, there was also no significant difference in proportion of brood reared on frames between treatment groups (Figure 4), even though OTC treated colonies had more workers covering frames than control colonies (Figure 4). It is possible that queens from both colonies produced similar amount of brood, but the number of larvae that survived to adulthood was significantly different between colonies, as most diseases that OTC treats affect larva. We also found no significant difference in overall colony weight gain between treatment groups (Figure 5). However, it is important to note that worker population and resource amount both factor into influencing overall colony weight. Weighing colonies alone does not provide a complete picture of colony health, which is why it is essential to pair weight and resource estimation metrics from hive inspections together to make an informed decision. Thus, although there was no significant difference in overall colony weight across treatment, exactly how colonies grew was statistically different when considering our hive inspection data (Figure 4). Such nuances are lost when examining at colony weight as a singular metric. Furthermore, we found a significant interaction between treatment and ambient wet bulb temperature on influencing internal colony temperature (Figure 6b), where OTC treated colonies were significantly less influenced by ambient wet bulb temperature, suggesting that they were able to respond more efficiently to rising ambient temperatures than control colonies (Figure 6b). The difference in internal colony temperature maintenance between treatment groups may have not had to do with colony fanner output, as there was no difference in the total number of fanners observed throughout the experiment (Figure 7). Instead, because honey provides thermal mass and therefore acts as a thermal buffer between extreme temperature fluctuations to the colony, internal temperatures of OTC colonies may have been more resistant to ambient climate changes because OTC colonies stored more honey in their frames than control colonies (Figure 4).
Before conducting this study, I expected prophylactic antibiotic treatment to have a negative effect on honeybee colonies based on the literature. I have a long history working with the consequences of anti-microbial resistance and antibiotic toxicity in the environment, so I was heavily biased towards antibiotics being detrimental for bees overall. However, after working with the colonies and analyzing the field data, I now can appreciate how antibiotics, when used properly and in the recommended dosages, can be beneficial to colony health and success. I previously would have advocated for stopping the usage of antibiotics altogether, but now I understand that like much of biology, antibiotics can be a beneficial tool, so long as we do not abuse its usage. Realistic, sustainable agricultural change begins by fully understanding the nuanced effect that human activity has on our environment, and this project highlighted the importance of taking research into the field and testing it within relevant contexts.
Conducting this project has taught me much about field work, beekeeping, collaborating with the local community, and how honeybee colonies behave throughout the season. It also taught me about what beekeepers care about in terms of colony health and success, which has made a more well-rounded honeybee researcher and has positioned me well to educate the Wisconsin beekeeping community on this subject. This project has only bolstered my passion for applicable science that involves the community and is designed with the community in mind and has shaped how I think about the future of my research career. Managing this project likely has led to my successful hiring as a post-doctoral researcher.