Final report for GNC23-380
Project Information
Flea beetles (Coleoptera: Chrysomelidae) are major agricultural pests, feeding on Brassica vegetables, greens, and solanaceous crops. Their larvae are minor root pests, while adult foliar feeding leaves characteristic shotgun patterned damage, often significantly reducing crop quality and yield. Current control measures include insecticides and exclusion netting to prevent adult feeding. However, due to the small size of these insects, fine netting poses additional issues, including temperature extremes and exclusion of beneficial insects. Additionally, netting does not protect the crop from newly emerging adults from the root zone. While chemical pesticides can knock down flea beetle populations, there are few options for organic growers when controlling this pest.
Biological control is an option for growers who focus on sustainable farming methods and reducing pesticide use. Currently, several commercially available species of entomopathogenic nematodes (EPN) are recommended for control of flea beetle larvae. However, this technique fails to address the high mobility of adult beetles, which emerge from overwintering locations and disperse into the crop. Thus, applications of EPN at the crop root zone alone have little effect on foliar damage. Because of this overwintering behavior, trap cropping to intercept adult beetles seeking hosts in early spring has also been used. Trap crops alone may reduce feeding damage on the main crop but fail to reduce pest populations without pesticide application. While some organic pesticides are registered for use against flea beetles, efficacy when treating the trap crop alone remains low. These outcomes suggest that management of flea beetles must provide control within the crop and in the landscape, attacking multiple life stages.
To address this problem, we aimed to develop a technique for controlling flea beetles in high tunnel Brassica crops using a combination of EPN and trap crops. We hypothesized that the cultural control provided by the trap crop would interact with the biological control services provided by EPN in a synergistic manner; i.e., adult flea beetles would be intercepted by and preferentially feed and oviposit on the trap crop, increasing effectiveness of the targeted EPN application to the soil to treat flea beetle larvae. Specifically, we used commercial EPN products that are readily available to growers, rather than lab strains that are used in a lot of EPN research. This management strategy is sustainable, requires low-inputs, and is compatible with organic production, all while targeting multiple life stages of the pest and providing more complete crop protection.
Our evaluation included experiments conducted in the greenhouse, at research high tunnels at Meigs Horticultural Farm, part of Throckmorton Purdue Agricultural Center in Lafayette, IN, as well as on-farm, working closely with high tunnel grower collaborators. We implemented this trap crop and EPN system first in the greenhouse at a small scale, to assess beetle feeding preferences to affirm trap crop choice, and determine the effect of EPN applications on soil-dwelling flea beetle larvae. In the high tunnels, we implemented the trap crop design at a larger scale, placing the trap along the entire length of the high tunnel to physically intercept beetles. Our data collection included sticky card and visual observation counts of adult flea beetles, and feeding damage assessment.
Our educational approach to this project includes not only close work and on-farm demonstrations with grower collaborators, but presenting to and communicating with larger grower audiences in order to increase the dissemination of our experimental findings. Over the course of the project, we shared information with a total of over 100 attendees of grower conferences and field day demonstrations, and reached further audiences through an online extension bulletin. We worked most closely with our grower collaborators and their farm support staff, who demonstrated interest in adopting trap cropping, EPN, or both, to their high tunnel cropping systems.
Our findings from this project ultimately support the use of trap cropping to prevent feeding damage to high tunnel Brassica crops. However, the data we collected in this project as well as the current literature suggest that EPN have potential to be an effective tool for controlling flea beetles, but we plan to continue to investigate this specifically in Brassica specialty crop systems beyond the timeline of this project. Beyond the immediate scope of the project, we opened a conversation with growers about allocating high tunnel or adjacent space for non-crop plantings, including companion plantings that support other natural enemies, to ultimately minimize the use of insecticides. All growers at collaborating farms plan, to some extent, to adopt a similar system in their Brassica crops to prevent feeding damage from flea beetles. Additionally, non-collaborating growers who were audience members at field days and grower conferences showed interest in adopting some of these practices, even to different cropping systems.
Flea beetles are routinely identified by growers as a major pest, resulting in economic loss throughout most of the growing season. The ultimate goal of this project is to investigate means to decrease flea beetle pressures on Brassica crops and develop a system that is low-input and easily adapted by high tunnel growers. To address this problem, our research objectives are as follows:
Objective 1: Assess efficacy of Brassica juncea trap crop in combination with commercially available EPN species.
To begin the project, work conducted in the greenhouse and at the research farm will investigate the trap crop system and effectiveness of commercial EPN products to control flea beetles.
Objective 2: Cater low-input multi-generation control of flea beetles to grower high tunnels.
In the second half of the project, we will work with growers to modify the trap crop system to best meet their individual needs, and implement the system at their high tunnels, to test these management approaches in a less controlled environment than the research farm.
Research
Preliminary Work: Crop Selection
Flash collard greens (Johnny’s Selected Seeds, Winslow, ME; Brassica oleracea) were selected as the cash crop to be used in the greenhouse and research farm experiments conducted to meet Objective 1. This crop was selected due to its relative heat tolerance and tendency to be slow to bolt, making it compatible with the high tunnel environment in mid-summer, the peak activity time for flea beetles. Mighty Mustard® Pacific Gold (Johnny’s Selected Seeds, Winslow, ME; Brassica juncea) is advertised by the manufacturer as a cover crop that may additionally be effective as a trap crop for crucifer-feeding flea beetles (Pacific Gold Blend 2019). Of the Brassica species listed, B. juncea has the second highest concentration of allyl isothiocyanate according to Cole (1976), and the highest concentration of phenylethly-isothiocyanate, two known flea beetle attractants (Table 1). Comparatively, B. oleracea, the cash crop, is lower in these two attractants, creating a stark contrast between the cash and trap crop, thus increasing the odds of differential flea beetle attraction. Wild turnip (B. rapa) was originally planned as the trap crop, but additional research suggested that beetles would be more attracted to a B. juncea trap crop over longer distances due to the chemical composition of the plant.
Table 1. Comparison of species-specific concentrations of allyl isothiocyanate and phenylethyl-isothiocyanate, two glucosinolate products identified as attractants for Phyllotreta adults by Wittstock et al. (2003) for six Brassicas as outlined in (Cole, 1976). n/a = data was not provided.
|
Species |
Allyl isothiocyanate concentration (ug/g) |
Phenylethyl-isothiocyanate concentration (ug/g) |
|
Brassica chinensis (now B. rapa subsp. chinensis) |
1 |
5 |
|
B. juncea |
24 |
7 |
|
B. napus |
n/a |
1 |
|
B. nigra |
32 |
4 |
|
B. oleracea |
1 |
2 |
|
B. rapa |
n/a |
3 |
Objective 1: Assess efficacy of Brassica juncea trap crop in combination with commercially available EPN species.
1.1: Affirm trap crop choice and EPN infectivity of beetle larvae with greenhouse feeding and oviposition bioassays.
Hypothesis: Adult flea beetle feeding and preference will be correlated with host chemical composition, specifically that isothiocyanate concentration makes a host plant more attractive.
Prediction: Both species of flea beetles will preferentially feed and oviposit in juncea over B. oleracea.
Materials & Methods:
Materials:
Flash collard (B. oleracea) and Pacific Gold mustard (B. juncea) seeds were sourced from Johnny’s Selected Seeds (Winslow, ME), and seeded in 72-cell trays (Greenhouse Megastore, Danville, IL) filled with a seed starting mix (Berger BM2; Berger, Saint-Modeste, QC, Canada). Six replicates were conducted at one time, three per beetle species, each occurring within a BugDorm-6 (MegaView Science Co., Ltd., Taiwan). Adult beetles used in bioassays were collected from a collaborating farm experiencing an infestation and were aspirated from plants in the field. Field-collected beetles were used in bioassays the same day of collection. Commercial EPN products were sourced from Koppert Biological Systems US (Howell, Michigan). Two products of two different EPN species were used: ‘Larvanem’ (Heterorhabditis bacteriophora), and ‘Capsanem’ (Steinernema carpocapsae).
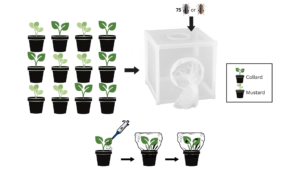
Experimental Design 1.1.1 - Beetle Feeding Preference:
Flash collards (B. oleracea) and Pacific Gold mustard (B. juncea) were seeded in the greenhouse. When plants were approximately 4 weeks old, they were transplanted into 4-inch round pots filled with potting medium (Berger BM7 Bark Mix; Berger, Saint-Modeste, QC, Canada). Pots were labeled with plant type, EPN treatment, and time duration (Table 2). 12 plants (one of each treatment combination) were randomly placed per BugDorm, with 6 total Dorms used. 75 field collected flea beetles were placed in each bug dorm. 3 Dorms contained Phyllotreta striolata, and 3 contained P. Cruciferae (Figure 1). BugDorms containing plants and beetles were kept under greenhouse conditions and pots were be watered as needed while minimally disrupting plants and soil. Adult beetles were left to feed and oviposit in the pots.
Table 2. Variables manipulated and treatment levels used in feeding bioassays.
|
Variable |
Levels |
|||
|
Plant |
B. oleracea |
B. juncea |
|
|
|
EPN Treatment |
S. carpocapsae |
S. feltiae |
H. bacteriphora |
Control |
|
Duration |
4 days |
10 days |
|
|
Data Collection 1.1.1 – Feeding Preference:
Data were collected at 4 and 10 days after beetle introduction. At each date, the number of beetles observed on each plant was recorded, and the percent of foliar damage was assessed on two randomly selected leaves per plant. After 4 days, half of the 12 pots per BugDorm (those assigned the 4-day treatment) were removed from the Dorms. Data were collected from the remaining 6 pots per Dorm after 10 days.
Experimental Design 1.1.2 – Beetle Oviposition Preference and EPN Treatment:
Upon removal from the BugDorms, all adult beetles were removed from the plants, and the potted plants were placed in 1020 trays (Greenhouse Megastore, Danville, IL). Plants continued to be watered as needed by filling the trays with about one centimeter of water to be taken up by plants through holes in the bottom of pots. After 2 weeks to allow for hatching and maturation of flea beetle offspring, EPN treatments were applied. EPN products were stored in the refrigerator until application. The total contents (50 million per pack ‘Larvanem,’ 25 million per pack ‘Capsanem’) were weighed to estimate the weight of each product needed for the intended application rate. To verify infectivity of the product, approximately 100 ij (EPN infective juveniles) by weight were mixed with 1mL of deionized water. This was applied to Petri dishes lined with Whatman 1 filter paper (90mm diameter) containing Galleria mellonella larvae at an application rate of approximately 20 ij per larva. G. mellonella mortality and subsequent EPN extraction using modified White traps (modified from Orozco et al., 2014) were used to determine product viability.
In the greenhouse, approximately 500 infective juveniles in 1mL of water were pipetted directly into the soil of each pot. Following EPN application, a perforated bag was secured over the top opening of each pot containing the plant using a rubber band. Plants were incubated and watered from below until emergence of adult flea beetles. First emergence was approximately 6 weeks after beetles were released into the BugDorms, and the experiment was concluded at 9 weeks, one week after any new emergence ceased.
Data Collection 1.1.2 – Beetle Oviposition Preference and EPN Treatment:
At the conclusion of the experiment, the number of beetles that emerged from each plant into the perforated bag were recorded. Beetle emergence was used as a proxy for oviposition preference (controls) and EPN efficacy.
1.2: Model low-input control of flea beetles in collard greens using a high-glucosinolate mustard trap crop.
Note: Originally, this objective included an EPN treatment, with the ultimate goal of using the trap crop alongside EPN applications to the soil to control the soil-dwelling flea beetle larvae. In 2024, adult flea beetle numbers in and around the high tunnels were extremely low, especially in the early season, the key time for implementation of this trap crop/EPN system, which aims to target the offspring of the initial overwintering generation. This was despite infestations of beetles at other parts of the research farm, and infestation of crops grown in the same high tunnels in summer 2023. In spring 2025, flea beetles were collected from growers experiencing infestations using a D-vac and released at the high tunnels. Retention rates of these beetles were very low, despite two releases at a rate of approximately 200 beetles per tunnel. Therefore, the below methods and subsequent data and discussion reflect the trap crop data alone, without the EPN component.
Experimental Design:
12 high tunnels at Meigs Horticultural Farm at Throckmorton Purdue Agricultural Center (Lafayette, IN) were used for the experiment. Flash collard greens were seeded per distributor instructions in 72 cell 1020 trays in the greenhouse in late March. One week after germination, seedlings were thinned to one per cell. Once seedlings had two pairs of true leaves, or after about 6 weeks, and when outside daytime temperatures were consistently above 10°C, they were transplanted into the high tunnels. All tunnel beds were covered with black plastic mulch, and landscape fabric was used to cover the soil between beds to prevent weeds. Seedlings were transplanted in four plots of ten plants each in one external bed of each of the 12 tunnels. Half of the tunnels, randomly selected, had a potted trap crop of mustard placed along the exterior wall, which was open during the summer season. Treatment assignment was blocked by tunnel type. Mighty Mustard® Pacific Gold was seeded in the greenhouse several weeks prior to anticipated emergence of overwintering flea beetles. The trap crop was seeded directly into 2-gallon trade pots containing a 1-inch layer of pea gravel at the bottom to help with water drainage and to provide weight and support to the pot to prevent the trap crop from being damaged by wind when placed outside. On top of the gravel, the pots were filled with BM7 potting mix, and the top 1 inch of the soil was a BM2 germination mix, in which the mustard seeds were directly placed. Before anticipated beetle emergence from overwintering, trap crops were placed directly adjacent to the bed of collards, outside the tunnel wall (Figure 2).

To help mitigate the growth of weeds around the potted trap crop, a layer of landscape fabric was laid down under the pots. Trap crops were reseeded as needed. Trap crops were be irrigated by extending a drip line outside of the tunnel to lay over the top of the pots, allowing irrigation at a rate and frequency matching that of the high tunnel beds. Additionally, after significant damage to the cash crop due to a complex of Lepidopteran pests in 2024, Agree (Bacillus thuringiensis) was applied to the collards bi-weekly or as needed based on scouting.
Data Collection:
Data collection occurred weekly for the duration of the experiment. First, yellow sticky cards were set in each cash crop plot at plant height (four per tunnel), replaced weekly. Tunnels with trap crops also had four sticky cards placed in the trap crop. Relevant insects on yellow sticky cards were identified to family, and all flea beetles or other Brassica feeding insects were identified to species. Timed visual observations counted the number of flea beetles observed per collard crop plot per tunnel recorded in one minute. For tunnels with a trap crop, 1-minute observations were taken in the trap crop row as well. In 2025, observation data was expanded to include four observations per tunnel: one in each collard plot, and four in each trap crop row.
Objective 2: Cater low-input multi-generation control of flea beetles to grower high tunnels.
Three high tunnel grower farms were treated as individual case studies for the implementation of the trap cropping system. Because of such stark differences between the farms, data from the three cannot be combined. Prior to implementation, significant planning went into working with each grower to best cater the system to meet their individual farm needs and landscape. Each of the case studies will be described below, with the farms noted using identifiers.
Farm A:
Farm Details and 2024 Pre-Sampling:
This farm produces both in protected culture and in field crops. Their protected culture crops are grown in one large, gothic style high tunnel, and in eight smaller caterpillar tunnels (dimensions 10m wide by 18m long). Annually, they grow Brassica crops including cabbage, kohlrabi, radishes, kale, arugula, and turnips. They have routinely identified flea beetles as one of their most difficult pests to manage, and often use fine mesh row covers to mitigate feeding damage to their crops.
Beginning in spring of 2024, sticky cards were set in high tunnel beds growing Brassica crops to assess and monitor flea beetle populations throughout the growing season. Sticky cards were labeled with the date, crop they were deployed in, and location, and were replaced every two weeks. Data collected from sticky cards were compiled into a bulletin that was shared with the grower in early 2025, depicting which of their crops attracted the most flea beetles, and the dynamics of the flea beetle population over the course of the growing season. These bulletins included additional information on other key pests and beneficials that were collected or observed, and used to help inform employees on the farm who work in the tunnels on insect identification.
Experimental Design:
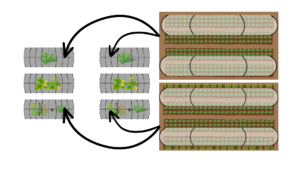
Four of the eight caterpillar tunnels were used for the experiment. The four tunnels were each assigned one treatment: EPN only, trap crop only, EPN and trap crop, and a control. The tunnels were configured in two rows of four, separated by an 8m wide gravel path. The tunnels used were not adjacent, and the tunnels between the experimental tunnels were planted with cucumber, a non-host item for flea beetles. Initially, this experiment was expected to be carried out in early June of 2025. The initial plan had to be altered in order to facilitate crops grown earlier in the season that were necessary for the grower collaborator’s community supported agriculture program. Therefore, the EPN application was not in the correct time frame to target any soil dwelling larvae. It was, however, possible that the drench application had an effect on adults on the foliage. In two of the tunnels, the outer most row of each of the tunnels was planted with a mix of Brassica napus, Brassica rapa, and Raphanus sativus (Brassica Classic Trio Cover Crop Seed Mix, True Leaf Market, Salt Lake City, UT) in mid July.
An arugula cash crop was seeded two weeks after the trap crop, to ensure there was sufficient trap crop biomass to provide protection to the vulnerable arugula seedlings. The cash crop was seeded in rows in four beds in the four experimental tunnels. To protect some of the crop and make sure that there was enough viable arugula to be included in the CSA, fine mesh stretched over low tunnels was placed over the outermost rows of arugula, in tunnels with a trap crop treatment, this was the rows adjacent to the trap (Figure 3).
EPN Application:
‘Larvanem’ (H. bacteriophora) and ‘Capsanem’ (S. carpocapsae) were applied at an application rate of 250,000 ij/m² (500,000 ij/m² combined) to the trap crop and arugula or arugula alone, based on treatment. EPN applications occurred six days after the arugula was seeded. The application occurred in the evening to prevent the impact of strong UV light on the viability of the product immediately following application. EPN products were stored in a cooler for transport until application and were mixed in a watering can with ½ gallon of water per square meter of bed space. Tunnels were irrigated using drip irrigation during the experiment.
Data Collection:
The experiment ran for four weeks, during which data were collected 2 to 3 times weekly. Sticky cards were first deployed in the trap crop at germination. Three yellow sticky cards (3” x 5”, BASF, Florham Park, NJ) were placed in each trap crop row to sample flea beetles. After the arugula sprouted, three sticky cards were placed per bed of uncovered arugula (six total arugula sticky cards per tunnel). Sticky cards were replaced weekly. Timed visual observations also occurred at each sampling date, where the number of flea beetles in a 1m plot were counted for one minute. Three observations occurred per row, per date. 6 arugula leaves were selected randomly from each observation area and the number of shotgun flea beetle feeding holes were counted.
Farm B:
Farm Details and 2024 Pre-Sampling:
Farm B operates year-round, providing produce to an on-site farm store. They grow both in field crops and in 4 full-size high tunnels, one of which is modified with supplemental heat to allow year-round production of cold-sensitive crops. Growing Brassica crops year-round has resulted in severe flea beetle infestations, resulting in crop loss. Like Farm A, pre-sampling occurred in 2024 and was used to create informational material for the grower and farm employees.
Experimental Design:
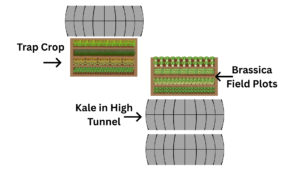
Sampling with sticky cards and visual observations in 2024 and spring of 2025 showed that flea beetles were first inhabiting the northwestern corner of the property, suggesting that adults were overwintering in a wooded area outside of the cropland nearby. In 2025, rows of arugula, radish, turnips, and baby kale microgreens were planted in a northwestern field plot in mid-April. Flea beetle activity was noticed almost immediately, and this plot became overwhelmed quickly. Working with the grower, we made the decision to leave this plot to grow and continue through flowering to serve as the trap crop for this site. To the south of the trap crop plot, there were several other Brassica green field plots and a large polyculture high tunnel (30m long by 9m wide) growing two beds of kale, serving as the cash crop (Figure 4).
EPN Application:
‘Larvanem’ (H. bacteriophora) and ‘Capsanem’ (S. carpocapsae) were applied at an application rate of 250,000 ij/m² (500,000 ij/m² combined) to the trap crop in early May when flea beetle populations were high. EPN were applied using a drench from a watering can, and beds continued to be irrigated using 6-inch sprinklers spaced every meter in the beds.
Data Collection:
One sticky card was deployed in each of the plots (one plot per crop) that made up the trap crop bed. Sticky cards were also placed in each Brassica field plot, and two sticky cards placed per bed of high tunnel kale. Sticky cards were replaced every two weeks, and accompanied with 1-minute visual observations. Three 30cm deep soil cores were taken from each trap crop plot two weeks post-application and combined into one composite sample per trap. These were transported to the lab in a cooler and set up in 12oz deli cups baited with five Galleria larvae. Infected cadavers were set in modified White traps to await EPN emergence to confirm infection.
Farm C:
Farm Details:
Farm C was added to the project in 2025, and thus had no pre-sampling in summer of 2024. This farm is smaller in size and production compared to the others, selling just at a local farm stand and market. Produce is grown in two high tunnels (18m long by 5m wide) and field plots. The farm prioritizes using regenerative, low-disturbance practices, and is certified naturally grown, conforming to the highest organic standards. Both tunnels are polycultures, including arugula, cabbage, bok choy, and mustard green Brassicas in addition to tomatoes, peppers, and cucurbits.
Experimental Design:
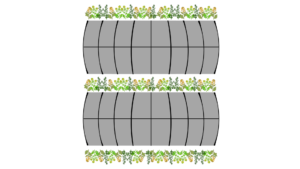
Both high tunnels, situated adjacent to each other with 3 meters between them, were used for the experiment. Brassicas were grown in polyculture in both tunnels during the growing season. In the last week of April, two rows of the Brassica Classic Trio Mix were seeded on each side of the high tunnels, and two rows were seeded in the middle (Figure 5).
EPN Application:
Only ‘Capsanem’ (S. carpocapsae) was applied to Farm C, because ‘Larvanem’ (H. bacteriophora) was not OMRI listed at the time of the experiment, although the process of listing the product is ongoing. EPN were applied using a watering can to the Brassica mix trap crop outside of the tunnels at a rate of 500,000 ij/m². EPN were applied early in the morning to avoid UV light.
Data Collection:
Three sticky cards were deployed in each pair of trap crop rows beginning at germination. Sticky cards were also deployed in each plot of Brassicas inside the high tunnels. One-minute visual observations were conducted at each sticky card location as well at each farm visit. Sticky cards were collected and replaced at each visit, every 2 to 3 weeks. Composite samples were taken from the trap crop root zone two weeks post-application to be used in lab bioassays as described above.
References
Cole, R. A. (1976). Isothiocyanates, nitriles and thiocyanates as products of autolysis of glucosinolates in Cruciferae. Phytochemistry, 15(5), 759–762. https://doi.org/10.1016/S0031-9422(00)94437-6
Mighty Mustard (2019, November 11). Pacific Gold Blend. Mighty Mustard®. https://mightymustard.com/varieties/pacific-gold/
Orozco, R. A., Lee, M.-M., & Stock, S. P. (2014). Soil Sampling and Isolation of Entomopathogenic Nematodes (Steinernematidae, Heterorhabditidae). Journal of Visualized Experiments : JoVE, 89, 52083.https://doi.org/10.3791/52083
Wittstock, U., Kliebenstein, D. J., Lambrix, V., Reichelt, M., & Gershenzon, J. (2003). Chapter five Glucosinolate hydrolysis and its impact on generalist and specialist insect herbivores. In J. T. Romeo (Ed.), Recent Advances in Phytochemistry(Vol. 37, pp. 101–125). Elsevier. https://doi.org/10.1016/S0079-9920(03)80020-5
Results
Objective 1:
1.1.1: Data collected from repeating these experiments twice in summer 2024 support B. juncea as a trap crop for collard greens (B. oleracea), as the average number of beetles observed as well as the proportion of damage was significantly higher on B. juncea at both time points (Figure 6). Data for the two flea beetle species are combined because there were no significant differences between the two.
Figure 6. More flea beetles preferred to inhabit and feed on B. juncea than B. oleracea.
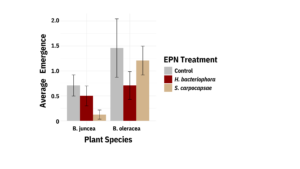
1.1.2: Data presented are summed across both flea beetle species, as trends did not differ. However, significantly more beetles emerged from B. oleracea than B. juncea, though EPN surprisingly had no significant effect on emergence (Figure 7).
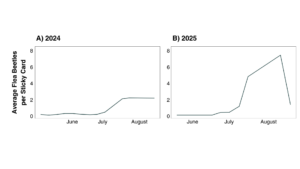
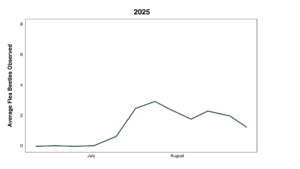
1.2: Flea beetle populations at the research farm were very low in both 2024 and 2025, despite introduction of collected flea beetles in 2025. Specifically, these numbers were very low during the spring, when an EPN treatment would be most effective, able to target soil-dwelling larvae that are the offspring of the overwintering generation. Populations increased in August, suggesting that second generation adults were entering the cropping system from other parts of the farm (Figures 8 & 9).
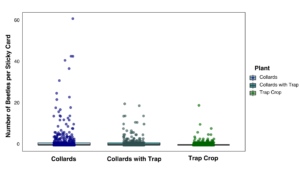
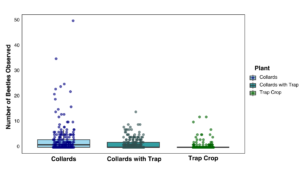
In 2024, the trap crop study data showed that based on visual observations, far more beetles were found in the mustard trap crop compared to the collard greens, corroborating the greenhouse bioassay data. However, the sticky cards placed in collard plots with an adjacent trap crop collected the most flea beetles (Figure 10).
Combined data from both years are presented in Figures 11 and 12 for beetle counts per sticky card and visual observation data. Overall, more flea beetles were observed and captured on sticky cards in collard plots without a trap crop than the trap crop alone or collards with an adjacent trap crop. The data include a lot of zeros due to overall low counts, with several instances of high counts in one individual date, specifically later in the summer, driving the trends visualized below.
Objective 2:
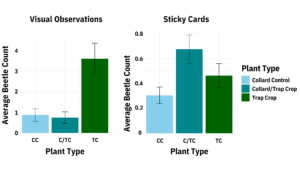
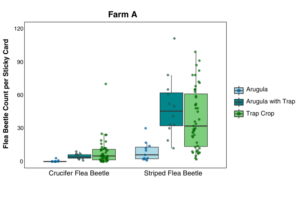
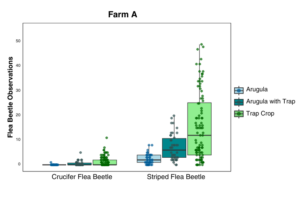
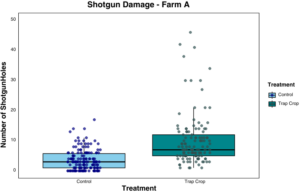
Farm A: Data were analyzed using Poisson GLMM with sample location and date as random effects. The flea beetle population, which was sampled by sticky cards and through visual observations, was mostly composed of the striped flea beetle, Phyllotreta striolata. Significantly fewer crucifer flea beetles were collected on sticky cards in arugula alone than either arugula with a trap crop or the trap crop alone (p = 0.001). Significantly fewer striped flea beetles were collected on sticky cards in arugula (p ≤ 0.024), and the arugula with a trap crop had significantly more beetles than the trap crop alone (p = 0.0002), (Figure 13). Flea beetle counts differed by crop and species, with both crucifer and striped flea beetles significantly more abundant on the trap crop than Arugula (p = 0.001). The interaction between plant type and beetle species was also significant (p = 0.03), (Figure 14). Arugula in high tunnels without the trap crop had significantly fewer shotgun feeding damage holes than arugula with the trap crop (p<0.0001), (Figure 15).
Farm B:
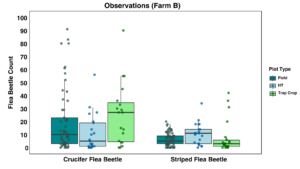
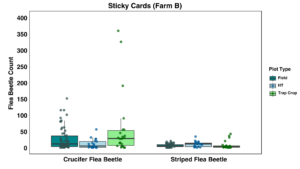
There was a trend for fewer crucifer flea beetles to be observed in the high tunnel than in the field plots or the trap crop, but differences were not statistically significant (Figure 16). Significantly more crucifer flea beetles were captured on sticky cards in the trap crop than in the high tunnel (p <0.001), but there was no significant trend for the striped flea beetles (Figure 17).
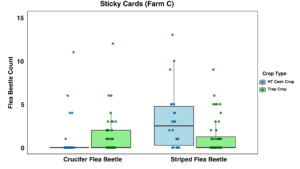
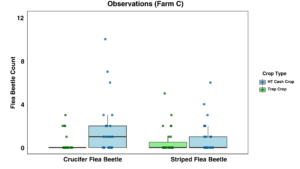
Farm C: On Farm C, there were no statistically significant observed differences between beetle counts or sticky card captures in the brassica trio trap crop rows or the cash crops in the high tunnels (Figures 18 and 19).
Discussion:
Objective 1.1: The collected data affirm the use of B. juncea as a trap crop for Phyllotreta flea beetles as the feeding preference was evident for both species of beetle. Additionally, though with the different treatments replication of control groups is low, there may be an oviposition preference that is the opposite of the feeding preference for the beetles, as more beetles emerged from the pots with collards than the mustard trap crop pots. A better understanding of movement of gravid females between the trap crop and cash crop can better inform placement of EPN treatments to control soil dwelling larvae. Additionally, EPN products that are marketed for control of flea beetles may not actually be effective in this system. We observed no statistically significant difference between the number of beetles emerging in from the different EPN treatment pots. Some literature suggests that the nematocidal properties of brassica crops that are used as green manures for plant parasitic nematode control can actually deter EPN as well. Further, much of the existing research on EPN for flea beetle control uses lab reared strains, rather than the commercial products available for growers to purchase. Future greenhouse experiments will assess whether exudates from healthy brassica plants or secondary metabolites released due to plant damage negatively impact EPN. I also plant to assess whether beetle diet (i.e. type of brassica they are feeding on - with varying chemical composition) impacts ability of EPN to infect and kill the host insect. I plan to continue to investigate this in the future, repeating replication to try to capture the true effect of multiple commercial EPN products on flea beetle larval mortality. I will work on additional methodology, such as floating out beetle larvae from the soil to determine exact pre-application counts, to refine the experiment with the ultimate goal of gathering information that can inform growers using EPN products in their specialty crop production systems.
Objective 1.2: Overall, through both years of the experiment, flea beetle populations, specifically in the spring and early summer, were too low to collect quality data. Because populations did not increase until later in the summer, there was no reason to apply EPN to the trap crop and plots. An EPN application would only target the soil dwelling larvae, and in Indiana, flea beetles do not complete two full generations per year, so larvae are only present in the soil from around May to June, after the overwintering population mates and lays eggs. However, from 2024 to 2025 we saw differences in trends in the trap cropping data. In 2024, more flea beetles were observed on the trap crop than the plots, and sticky cards in the plots of collards with an adjacent trap crop collected the most beetles. The overall data trends were driven heavily by 2025 data, where in August there were large numbers of flea beetles colonizing collards in the control tunnels which did not have a trap crop. This may be in part due to releases of flea beetles in the 2025 season. Flea beetles were collected from grower farms and released at the research tunnels twice in early June 2025. Release points were established so that released beetles had equal opportunity to travel to tunnels representing both trap crop treatments. While the beetles did not appear immediately after release, it is possible that they colonized other plants nearby, such as cruciferous weeds, to travel to the high tunnels later in the season. Additionally, and anecdotally, while the beetles collected from the farm were a mix of crucifer and striped flea beetles, the majority of beetles recovered at the research farm were crucifer flea beetles. The beetle species distributions could be related to crop preferences of the beetles, or behavioral and ecological differences between the two species that are usually regarded as a species complex, despite evidence in the literature of differences in their annual period of activity.
Objective 2: Farm A yielded the most promising results from the trap crop study, despite the fact that the EPN application was not properly timed to have an effect on soil dwelling larvae. In this study, there was a significant effect of the trap crop, though it was the opposite of what was hypothesized. The arugula in the high tunnels without the trap crop had very few shotgun feeding holes, and fewer beetles were observed or collected on sticky cards there. This could imply that while the trap crop has an effect, the proximity of the trap crop to the cash crop is important. It is possible that flea beetles were able to move easily from the trap crop to the arugula in the same tunnel, but would not pass the tunnels of non-host plants (cucumber) that stood between them and the control arugula tunnels. This has implications for push-pull strategies, using an unattractive plant in conjunction with the trap to enact behavioral change on the pest. The arugula grown in high tunnels was very high quality, which was lauded by the grower who usually had trouble growing any brassicas without insect netting. This system was desirable to the grower, who plans to use a trap crop in the future to control flea beetles.
On Farm B, we had less control over experimental design compared to other farms due to the layout, which likely resulted in the suboptimal trends in the data collected. We did not see any effect of the EPN on the flea beetles, with populations continuing to climb as the year went on, but due to the large size of the farm and the number of plots growing host material for flea beetles, it was not feasible to isolate the experiment, and new migration into the crop was likely to be occurring. There was a slight impact of the trap crop according to observation data. I intend to continue to work with this grower in the future, and he has expressed great interest in trying other protocols and methodologies to provide low-input and sustainable ways to manage flea beetles because his farm tries to not apply any chemical insecticides at all.
At Farm C, we detected far lower flea beetle populations than the grower had reported in years past, and especially compared to the other two sites that were sampled. This resulted in data without strong trends. Additionally, we had trouble managing weeds in the trap crop at this site while adhering to management constraints due to certifications held by the farm. The trap crop was at times during the summer dwarfed by tall weeds, creating a polyculture that may have been less effective because any volatiles or cues given off by the trap crop could have been muddled with the other plants present. However, this grower expressed interest in continuing to use a trap cropping system in the future.
Educational & Outreach Activities
Participation summary:
Educational tools were created for grower collaborators based on on-farm data collection in 2024 and 2025. These were distributed in the spring following the field season where data were collected. In 2024, I participated in a field day at Throckmorton Purdue Agricultural Center located in Lafayette, IN, with an audience of growers, agricultural professionals, and academics. This included a demonstration of research in collaboration with other researchers, showcasing pest management practices in high tunnel specialty crop systems. In the demonstration, I talked with attendees about utilizing a trap crop inside or adjacent to the high tunnel, as well as including information on entomopathogenic nematodes. I showcased my research high tunnels where the trap crop system was implemented, and discussed identification of flea beetles and other common pests of cruciferous vegetables. This same year, I published an insect spotlight article in Purdue University's Vegetable Crops Hotline Newsletter, which has over 800 online subscribers and is open access, reaching an additional, larger audience. Data from this project were shared to an academic audience at the Entomological Society of America annual meeting held in Phoenix, AZ in November 2024, as well as the same meeting in 2025, in Portland, Oregon. I also gave a presentation to an audience of over 50 growers at the Indiana Small Farm Conference/Horticulture Expo in 2025 and 2026.
In the future, these data will be used for additional academic presentations (conference TBD), and will be published in at least one peer-reviewed publication during the last year of my degree. Additionally, I plan to make further contributions to the Vegetable Crops Hotline, where I will share information on entompathogenic nematodes and trap cropping for flea beetle control.
Project Outcomes
This project resulted in three grower collaborators who expressed interest in continuing to use a trap cropping system to mediate flea beetle feeding damage to their high tunnel Brassica crops following the end of my experiment. Further, it has opened doors for additional years of experimentation to collect more data to hopefully more concretely be able to provide recommendations in the near future. Additionally, outside of the immediate scope of my project, I have the opportunity to speak to and with other growers at the Indiana Small Farm Conference, where I engaged in a Q&A session with growers who work in different systems but are interested in incorporating biological or cultural control methods. Between conference years, the number of growers interested in low-input control methods is growing across agricultural systems, especially in response to increased knowledge on the negative impacts of chemical insecticides, environmental concerns, and instances of pest resistance.
Economic Benefits
Flea beetles are a devastating pest that many growers struggle with, specifically when feeding renders crops unmarketable. In the high tunnel system especially, and for small-scale growers in general, there is an increased reliance on premium quality produce for direct-to-consumer sales. Improving pest suppression or diverting pest pressures to non-crop (trap) plants can reduces foliar damage, which protects yields. Additionally, this can help decrease costs associated with other labor intensive control methods such as the use of fine insect netting or insecticides.
Environmental Benefits
This project used no insecticides to control pests, and relied completely on a combination of biological and cultural control. Compared to other biological control agents as well, EPN pose very low risk to non-target organisms. A system that includes no insecticides minimizes environmental contamination and negative impacts to other important insects in the cropping system, such as pollinators. Trap crops can provide additional benefits to the crop environment as they can provide additional resources for other beneficial organisms.
Social Benefits
Biological and cultural control methods align with growing consumer demands for environmentally responsible crop production. Many high tunnel growers sell directly to local markets, where customers increasingly value reduced pesticide use. A goal of this project is to increase grower confidence in adopting biologically and culturally based pest management methods. Further, this can increase sharing of knowledge between growers at who collaborate at markets or through farm stores to distribute produce. This in turn strengthens local food systems by helping small and mid-sized farms maintain profitability while adopting sustainable practices.
While I plan to continue working in this system beyond the scope of the project described in this report, my ultimate goal is that the outcomes will provide growers with practical guidelines for implementing EPNs and trap crops under high tunnel conditions, supporting long-term reductions in chemical inputs, and improved economic resilience. By increasing adoption of integrated pest management strategies for Indiana high tunnel growers, this project contributes to the long-term sustainability of specialty crop production in the region. Ultimately, optimizing the informed use of these key tools will help growers to minimize the costs associated with managing their high tunnel crops. Beyond using a trap crop for pest control, the project helped to demonstrate the value of sacrificing space on-farm for non-crop plants, particularly those that provide some other ecosystem service. Anecdotally, the mustard trap crops used at our research farm were excellent resources for a number of pollinators, namely syrphid flies, which can provide pollination as well as biological control services.
Through this project, and specifically through disseminating results and speaking with growers, I have learned a lot about what sustainable agriculture looks like from a different perspective. I realized that we as scientists have idealized ideas about regenerative, "green," or low-input practices, but can sometimes be caught in a disconnect with the growers themselves who are, more than anything, concerned with the well-being of their crop, which is their livelihood. I have learned that sustainable for them doesn't just mean that there is optimal protection for the environment and minimized use of insecticides, but sustainable for themselves; feasible, easy to carry out, and able to turn a profit off of.
Specifically in year two of the project, working more closely with grower collaborators, I also realized how different sustainable agriculture can look between farms. Each grower or farm operates under different leadership, and with different thresholds and tolerances for pests, which is driven by their consumer base. For example, a grower who sells exclusively to a local clientele that is intimately familiar with the farm and their practices may be more willing to tolerate a degree of pest damage on crops compared to a grower that uses their crops to support a large, multi-farm CSA program. Inputs and labor are individual choices, and being able to practice sustainability, to any degree, relies on catering methods and models to each individual, which requires one-on-one work.
A collaborating grower who owns and operates a small-scale farm in northern Indiana (Farm A) felt very positively of the trap crop system we implemented, though the effect did not align with our original hypothesis about the treatments. The farm had not been able to successfully grow arugula in recent years due to severe flea beetle infestations that left the crop decimated. Any arugula that was attempted to be grown was seeded and immediately covered by a fine mesh. Using the trap cropping system, the arugula produced in the control tunnels had very little flea beetle damage, despite being uncovered. This arugula was able to be sold and distributed through the CSA, meeting consumer standards.
