Final report for GNE17-151
Project Information
Silage leachate is potent wastewater produced from the fermentation of corn and forage. Silage leachate contains high concentrations of organic acids; thus, it is acidic and has low redox potential. Disposing of this leachate can be a challenge for farm operators. In the current study, we investigated improving the quality of silage leachate. We had three objectives: I) to reduce the chemical oxygen demand (COD), II) find a relationship between the COD removal and residence time, and III) to remove nutrient from the leachate.
For COD reduction, an anaerobic Continuous Stirred-Tank Reactors (CSTR) digestor design was adapted and used. The essays used for this purpose contained anaerobic sludge to inoculate the reactors. Silage leachate was added to the reactors gradually at the rate of 50% of the volume of the reactor. The COD reduction was monitored in 6 residence times. For reduction of phosphorus, in a different set-up, limestone chips were applied. Finally, for the reduction of nitrate, flow-through woodchip-containing bioreactors were used. Separate set-ups had to be considered for each of these pollutants, due to the nature of the wastewater.
The CSTR successfully reduced COD from the leachate. The COD reduction followed first-order kinetics. The COD reduction increased with increasing residence time with 50% removal observed in 120 h. With the application of limestone, phosphorus was removed, and pH increased. It is worthy of note that increased pH inhibited the removal of COD. Thus, the reduction of COD and phosphorus should happen in a series of reactors. Finally, our results showed the removal of nitrate followed first-order kinetics, and the entire removal of nitrate happened within 24 h. Overall, our study showed, using a series of reactors, the quality of the silage leachate was improved.
Our overall objective was to improve the quality of the silage leachate to help farmers to avoid fees and penalties, thus, to reduce the farm’s spending. The current research addressed three of five required themes for the NESARE graduate student grant: “the reduction of environmental risks”, “the reduction of costs”, and “the conservation of soil, the improvement of water quality and the protection of natural resources”.
Our specific objectives were:
- To reduce chemical oxygen demand (COD), thus, biological oxygen demand (BOD) of the silage leachate.
- To determine an optimum residence time required for the COD removal.
- Nutrient removal
To achieve these objectives, field and laboratory experiments were performed.
The fermentation of silage produces acidic leachate with low redox potential. This potent leachate is rich in nutrients and organic matter. Several studies observed that biological oxygen demand (BOD) and chemical oxygen demand (COD) exceeded 49000 mg L-1 (Gebrehanna et al., 2014). These characteristics pose a danger to the environment and infrastructure of the farms. The corrosive runoff from the silage bunkers dissolves the concrete and burns the vegetation along its route. The direct discharge of the silage to the water bodies, cause an immediate drop in the dissolved oxygen. In comparison to other agricultural wastewaters, silage bunker leachate has the poorest water quality, although small in volume. Only1 L of silage leachate depletes the dissolved oxygen (DO) of 10,000 L of water below the tolerance of fish (Gebrehanna et al., 2014). Large scale fish-kill was reported due to the leakage of the silage leachate (Schade, 2005).
The leachate also contains high concentrations of nutrients. The excess nutrient leachate to waterbodies is concerning. Excess nutrient causes algal blooms, which leads to the release of toxins and odors and ultimately can lead to hypoxia. Lake Champlain algal blooms is an example of the undesirable consequences of such excess nutrient in the water bodies of the Northeast, USA (LCBP, 2008; Morse and Munroe, 2011).
Given the extremely poor quality of the leachate, federal and state regulatory provisions were taken to ban the discharge of silage leachate from the concentrated animal feeding operations (CAFOs) into streams and waterways (NYSDEC, 2017). Also, farmers frequently face fees and fines because of the disposal of insufficiently treated silage leachate. The current safety measures require the silage leachate to either be stored, if low flow occurs, or otherwise for the wastewater to be conveyed to vegetated buffer areas (NRCS, 2013). Small farms, however, do not have storage lagoons.
While most research focuses on the management of silos, few studies investigated the treatment of the silage leachate. Anaerobic digestion of the silage leachate was investigated previously by adding limestone chips to digesters in a laboratory experiment (Barry and Colleran, 1982). By increasing the pH of the silage leachate to 8, they observed a COD reduction of 86 to 89 % in 3 days. In the current study, we plan to build upon this study and investigate the treatment of silage leachate using anaerobic digestion. We also tested the COD reduction in different residence times. Together with the estimated flow rate from (Cropper and DuPoldt, 1995; NRCS, 2013), this information can provide guidelines for the size of the digestors in field settings. Finally, we investigated the removal of nutrients, specifically P and N, from the leachate.
Cooperators
- (Researcher)
Research
1 Chemical oxygen demand (COD) reduction
1.1 Silage leachate
Silage leachate was obtained from a dairy farm in Tompkins County, NY. The silage in the bunker in this site was covered with plastic sheets. The leachate was drained using tiles that are connected to a storage tank. The storage tank content was ultimately pumped to the manure pit. The chemical oxygen demand (COD) of the silage leachate was 20500 mg/L.
1.2 essay experiment
Anaerobic Continuous Stirred-Tank Reactors (CSTR) set-up was used for reducing the COD of the silage leachate. Six 250-ml flasks were filled with 100 ml anaerobic sludge obtained from the Ithaca Wastewater treatment plant. The chemical oxygen demand (COD) of the sludge was 28600 mg/L. Treatments included essays with and without activated carbon (1 g) in triplicates. In the first stage, 3 ml silage leachate was added to the essays. The essays were, then, sealed, and anaerobic conditions in the essays was provided by replacing the headspace with nitrogen gas. Controls were also considered for comparison.
The essays were put on the magnetic stirrer for constant mixing and were kept at 28 ° C. For conditioning the reactors, in 5 consecutive days after that, 5ml more leachate was added to the system each day. The step by step addition of silage leachate to the batches was done so that the essays would be conditioned. After this stage, the experiments began by replacing 50 ml of the mixture of sludge and leachate with 50 ml leachate (first shock). The COD concentration was measured every 24 h for 5 days. After the 5th day, the second shock was introduced to the system by replacing 50 ml of the mixture with 50 ml of the leachate. Similarly, the COD concentration was measured every 24 h for 5 days. We used 40% replacement ration to assure the increase in pH with COD reduction does not increase to the level that the transformation of ammonium to ammonia inhibits bacterial activity.
2 Nutrient removal
2.1 phosphorus (P) removal
Six batches were constructed using 208 L plastic drums in Harford dairy research farm, Cornell University (Figure 1). Sampling ports were located on the side of the barrels. The batches were sealed with venting occurring through a bottle submerged in water. Three of the batches were filled with limestone, and the three other batches were used as controls. One ton of limestone chips were used to fill the three barrels. The average size of the limestone chips was 1 cm2.
The batches were filled with two series of the silage leachate acquired from two different dairy farms. The first series was taken from a 1000 gallons underground tank used for the silage leachate storage in Harford Cornell dairy research center. The leachate in the pit may have been mixed with rainwater. The second series of leachate was acquired from a farm in the Tompkins’s County area. After filled with leachate, daily samples were taken from the batches. The samples were analyzed for total phosphorus (P) and reactive P content using the ascorbic acid method. The pH and COD concentrations were measured.

2.2 Nitrogen (N) removal
Assuming that after the improvement of water quality, ammonium can oxidize to nitrate, an experiment was conducted to reduce nitrate through denitrification. Synthesized polluted water was injected to 12-L up-flow chambers containing woodchip in duplicates. Woodchip can facilitate denitrification. The influent contained 11.5 mg/L nitrate. The removal of nitrate was monitored in 4 residence times of 2, 4, 8, and 24 h.
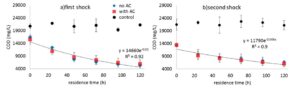
1-COD Reduction
The COD concentrations in the essays in different residence time is shown in Figure 2. With introducing both shocks, a reduction in COD was observed in the essays over time. This reduction followed the first-order kinetics. In 120 h, on average, a 50% reduction of COD was observed in both experiments. The addition of activated carbon did not create a significant difference. It is of note that pH was kept below 7 to assure the transformation of ammonium to ammonia did not occur. Ammonia is toxic to methanogenesis (Yenigün and Demirel, 2013; Sung and Liu, 2003).
2-Removal of P
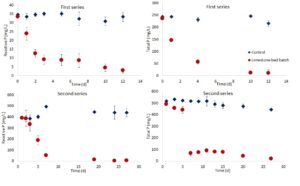
The result of the experiments indicated that limestone chips successfully removed phosphorus from leachate. In the first series of experiments, the pH increased from 6.1 to 7.5, and reactive P reduced from 30 mg/L to 3 mg/L. The total P in this series decreased from 235 mg/L to 10 mg/L (Figure 3).
During the second series of experiments, the pH raised from 4.3 to 7.5. The reactive P reduced from 514 mg/L to 4, and total P decreased to 19 mg/L from 400 mg/L. Overall, more than 95% of the reactive and total phosphorus was removed from the leachate by the bioreactors.
It is of note that although adding limestone was successful in the P removal, they did not help reduce the COD. In the first series, the batches reduced 54 % of the COD in 10 d. In the second series, however, only 23% removal of COD was observed in 30 days. The slow COD reduction was attributed to the high ammoniacal N concentration, which was transformed to ammonia in higher pH. The ammoniacal N concentrations in the first series and the second series were 300 and 1600 mg/L, respectively.
3-Removal of Nitrogen (N)
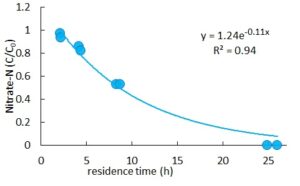
Woodchip was successful in the removal of nitrate. The removal of nitrate varied from 5% to 100%, with maximum reduction observed in 24 h. Nitrate removal following first-order kinetics. Such removal has been reported in other studies too which applied woodchip for the reduction of nitrate.
The current study investigated improving the quality of silage leachate by reducing chemical oxygen demand and nutrient content. We showed that CSTR design with the gradual addition of leachate successfully reduced the COD from the silage leachate. The COD reduction followed a first-order kinetic rate, increasing with increasing residence time. On average, a 50% reduction of COD was observed in 120 h.
Nutrient removal can be achieved through a series of treatment methods after the reduction of COD. By using limestone, phosphorus was removed from the leachate, although the reaction was slow. About 90% of phosphorus was removed in 12 to 30 days. Removal of nitrate, however, occurred faster. In 24 h nitrate was removed in its entirety.
Education & outreach activities and participation summary
Participation summary:
We have a manuscript ready to be submitted. the tile of the manuscript is: ” Effect of biochar amendment on nitrate-N removal in bioreactors"
Project Outcomes
The objective of this study was to improve the quality of silage leachate. In comparison to other agricultural wastewaters, silage bunker leachate has the poorest water quality. Though small in volume, it has great potential for environmental contamination. This is due to the combined low pH (high acidity), low pE (high electron activity), and high nutrient content, which is unique among wastewaters. Silage leachate direct discharge to the stream cause immediate depletion of the oxygen. Also, its containing nutrients pose a danger to the downstream water quality. Moreover, farmers have difficulties disposing of it.
Here, we provided methods to improve the quality of the silage leachate. If the reduction of COD is of interest, the anaerobic digestors with some modification can be used. We added the leachate to the digestor gradually, with injection not exceeding 50% of the volume of the reactor. The gradual addition allowed the reactors to be conditioned with the quality of the leachate. Wich such provision, our study would be able to help farmers to treat silage leachate.
The main lesson gained from this study was the application of the anaerobic digestors for the silage leachate treatment. We learned that cautions should be taken for such an application. Maintaining pH below 7 was crucial in the treatment since ammonia, which is toxic, can be produced in high pH values.
On a different note, this study increased my understanding of farm operations regarding the production of silage leachate. During this research, we visited many different farms and talked to farmers on various occasions. This research promoted communication with local farmers and provided us with the opportunity to observe the problems created by the silage leachate, including the damages to the concrete structures (Figure 5).

In addition, we noticed that management strategies taken by the farmers could immensely influence leachate production. For example, in Harford dairy research farm, which is equipped with machines to wrap the silo in the bags, minimal leachate production was observed (Figure 6). Whereas in another field where silo was stored in bunkers, more leachate was produced. We acknowledge that purchasing such machinery may not be feasible for all farmers. Therefore, finding practical ways to improve the quality of the leachate was, indeed, important.
