Final report for GNE18-186
Project Information
Synergistic microbial endophytes have a long history of enhancing plant growth. However, many important crops species have very few known mutualists that can non-pathogenically impact plant growth and production, and many of these are limited to leguminous crops. Therefore, to assess if vegetable crops such as pepper and tomato can benefit from bacterial mutualists, we tested inoculation of these crops with the bacterium Gluconacetobacter diazotrophicus (Gd), a host generalist species isolated from sugarcane. We measured the total yield of harvestable fruit, quality of fruit based on USDA Agricultural Marketing Services guidelines, and total plant growth. We found that in bell pepper , inoculation with Gd increased the number, total fresh, and total dry fruit weights per plant, the total dry matter accumulation of aerial tissues, and the total cumulative biomass of all tissues. However, fruit quality was severely impacted both in treated and control plants. In hot pepper, inoculation increased the total number of peppers but not the total weight of fruit, meaning each fruit was individually smaller. Additionally, more fruit exhibited surface imperfections known as 'corking' with inoculation than with controls. In tomato, no significant difference was observed as a result of inoculation in fruit quality, quantity, or plant biomass accumulation. In conclusion, we found that inoculation of peppers effected an increase in the total number of fruit produced in both cultivars, but with differences in average fruit weight and leaf and stem production despite both hosts being C. annuum. Sweet pepper production could benefit from inoculation, as average pepper weight also increased and more leaf tissue was formed to drive photosynthesis and metabolite formation. Producers are unlikely to derive any benefit from inoculation of hot peppers as lower prices are paid for 'corked' peppers as they are rated as damaged by the USDA-AMS and no net biomass increase was achieved. Tomatoes in this experiment were plagued by blossom end rot which was wholly unexpected given the hydroponic growing media.
Objectives: The goal of this research was to evaluate the effect of inoculation of non-legumes with the BNF bacterium, Gluconacetobater diazotrophicus (Gd) . My specific objectives were to:
1. Detail the impact of Gd inoculation of tomatoes and peppers on vegetative plant growth, fruit yield, and fruit quality.
2. Assess crop maturation and fruit abortion differences as a factor or inoculation.
3. Evaluate the economic value of Gd inoculation using USDA-AMS grading standards and pricing.
An objective not achieved in the duration of this experiment due to COVID-19 Impacts was
4. Evaluate the nutritional macromolecules, micronutrients, secondary metabolites and piquancy components of assessed fruit.
With an growing population placing increased demands on agricultural output, new techniques to improve crop production and nutrition are more of a necessity than ever. Coupling this with continued pressure for sustainability, environmental stewardship, and nutritionally dense products, novel approaches such as bacterial modification of plant growth are one such avenue of increasing production. Taking a page from the well established and agronomically successful incorporation of Rhizobia in soybeans, new species of plant endophytes are being employed outside of their traditional hosts. One potential endophyte, Gluconacetobacter diazotrophicus, has shown promise in recent trials. Not only can this bacterium non-pathogenically infect myriad agriculturally important crops, it has shown the capability to fix atmospheric nitrogen, produce plant growth promoting hormones such as Indole-3 Acetic Acid (IAA), Gibberellins A1 and A3 (GA1, GA3), and to increase plant water use efficiency (WUE).
This project aimed to explore the inoculation of two agronomically important fruit crops in the Northeastern US, tomatoes and peppers, by the bacterium G. diaz. Tomatoes represent a high value crop that has come under attack in recent years for nutrient dilution at the expense of yield, as well as high exogenous fertilization requirements. Peppers are a very high value crop that are increasingly being sown in controlled environments to ensure year round fresh availability, and as with tomatoes require extensive, unsustainable fertilization regimes. This research hoped to examine meeting the needs of production while limiting the concomitant costs, both financial and environmental, through replacement of exogenous N fertilization via bacterial inoculation. Simultaneously, we aimed to assess potential increases of production over standard practices, in both biomass and any effected secondary metabolite changes in fruit from bacterially sourced plant growth regulating hormones (PGRH). If even a single one of these three results was attained, the benefit to crop production for northeast farmers invested in these two crops may be substantial. Equally important, as bacterially sourced nitrogen is 'loss-proof' and may decrease exogenous N requirements, the environmental benefits could lead to greater sustainability
Research
To assess inoculation effects of G. diazotrophicus on C. annuum, a factorial experiment combining 6 levels of nitrogen fertilization and 2 levels of bacterial presence began on 10/25/2018. All experimental units were self-contained in closed system Deep Water Culture (DWC) hydroponic buckets, purposely designed and built for this experiment. Each bucket was of the standard 18.9L size and was opaque to prevent algal growth in the root zone. Externally, each bucket had a clear plastic standpipe for rapid assessment of water levels inside the DWC, as well as to expedite drainage. Each DWC was lidded with an opaque lid, again to prevent light penetration. Each lid had a 76mm hole for the plant growth netcup basket, centrally drilled, and three small holes; two proximal to the standpipe and one distal. The first proximal of these, 18mm in diameter, was used for nutrient delivery and Electrical Conductivity (EC) and pH measurements, and was stoppered with a #2 rubber stopper. The next proximal hole, 6mm in diameter, was uncovered and served as an air pressure equalization port. The remaining hole was an entry port for a 4mm inner diameter, 6mm outer diameter air hose used to deliver aeration to the nutrient solution, ensuring root zone oxygenation. The air hose delivered approximately 10 L of ambient air per minute, ensuring oxygen saturation of the solution surrounding the roots. Solution was continuously aerated 24/7, EC maintained at <2.4 with addition of distilled water and pH adjusted with non-nutritive acid/alkaline drops.
In order to assess potential exogenous nitrogen replacement potential, six total levels of nitrogen fertilization were used as a co-factor. A recommended dosage of 190ppm N was the Controlled Environmental Agricultural Center standard (U. of AZ), with five other levels levels of nitrogen. Deficient levels were 25% of Recommended (%R), ~47.5 ppm N; 43.75 %R, ~83 ppm N; and 62.5 %R, ~119 ppm N. A suboptimal level of 81.25 %R was included at ~154 ppm N, as well as a supra-optimal level of 118.75 %R, ~226 ppm N. The control was the 100 %R, ~190 ppm N recommendation, for a total of six fertilization levels. Equal gradation will allow for regression if appropriate. All other nutrients were provided in equal measures and at sufficiency, and comprised as Phosphorus (P) 50 ppm, Potassium (K) 291 ppm, Calcium (Ca) 176 ppm, Magnesium (Mg) 60 ppm, Iron (Fe) 2.85 ppm, Manganese (Mn) 0.48 ppm, Zinc (Zn) 0.14 ppm, Boron (B) 0.48 ppm, Copper (Cu) 0.14 ppm, Molydenum (Mo) 0.10 ppm.
To assess inoculation effect, two levels of bacterial presence were incorporated, as 'Inoculated' or 'Not-Inoculated'. 'Inoculated' (INOC) seeds were treated with a surface wash and soak in Gluconacetobacter diazotrophicus in an adjuvant solution. 'Not-Inoculated' (NON) seeds were treated with a G. diaz free dH2O wash and soak in the same adjuvant solution. 50 randomly assigned seeds were inundated with 200 ul of respective solution in a 60 mm sterile petri dish, and periodically swirled to ensure full exposure. Both treatments lasted for 10 minutes to allow for suspended bacteria and material to adhere to the seed surface, after which excess liquid was drained and the seeds allowed to air dry at room temperature (~ 21 oC) for 14 hours. (The adjuvant solution is a proprietary mix of seed adhesion compounds and bacterial support components, whose exact composition is protected under an NDA.)
The bacteria for this experiment was cultured in sterilized liquid ATGUS media [yeast extract (2.7 g/l), glucose (2.7 g/l), mannitol (1.8 g/l), MES buffer (4.4 g/l, K2HPO4 (0.65 g/l), pH 6.5], and incubated for five days at 60 rpm and 30oC. Colony Forming Units (CFU) was determined by serial dilutions and plating in petri dishes (ATGUS solid media [agar (0.8% w/v), yeast extract (2.7 g/l), glucose (2.7 g/l), mannitol (1.8 g/l), MES buffer (4.4 g/l, K2HPO4 (0.65 g/l), pH 6.5]) as well as OD600 spectroscopy (0.6). The applied CFU was a minimum 1.8 x 107 CFU/ml, above industry recommendations for this bacteria.
Seeds were sown in tared 30mm x 30mm x 35mm AO36/40 rockwool cubes (Grodan, Inc.) with 2 seeds placed per pre-drilled hole. A small plug of rockwool was pressed into the hole, over the seed, to ensure contact. Seeds were germinated in a Growth Chamber (Conviron Systems, Inc.), 16h at 25oC, 8h at 22oC, until seedings emerged. Plants were provided only with tap water to maintain moisture of rockwool; no nutrient solution was added. Upon emergence, a 16h 'day' (Photosynthetic Active Radiation [PAR] ~210 umol, 25oC) and 8h 'night' (darkness, 22oC) was maintained until roots began to penetrate the rockwool base. Again, only N-free tap water was provided at this time. Upon root emergence, the plants were randomly assigned to individual Deep Water Culture buckets according to inoculation status, were seated in 76mm rockwool blocks seated in 76mm round plastic netcups, and placed into the predrilled bucket lid holes. After 96 hours in situ, plants were thinned to a single plant per bucket.
Greenhouse conditions were maintained to within acceptable growing parameters, PAR ~220-236 @ 16h day, 8h night; Temperature 62oC-74oC; humidity ambient. Nutrient solutions were changed twice per growing cycle and recharged with an additional 19L of nutrient solution, for a total of three total doses of nutrient solution averaged at about 40 days between doses.
Plants were grown according to crop and then fully harvested, fruit, root and shoot material. Sweet pepper was harvested at 119 days after planting (DAP), hot pepper 119 DAP, and tomato 126 DAP. All fruit was sectioned, counted, individually weighed, and graded by USDA-AMS guidelines. All plant portions were separately dried at 60oC for 48-72 hours to ascertain dry matter (DM). Statistics on measurements and responses was performed via ANOVA with SAS Software (Copyright 2017 SAS Institute, Cary, NC), specifically PROC MIXED, GLM, and GENMOD using Tukey-Kramer LSD for detection of significant differences.
Trial List:
NOTE: All three plant cultivars, sweet pepper, hot pepper, tomato, were subject to the same experimental conditions, nutrient solutions, and inoculation techniques. Harvest varied by crop as reported.
Crop #1: Pepper, sweet (Capsicum annuum var. 'California Wonder'), One trial, repeated
Crop #2: Pepper, piquant (Capsicum annuum var. 'Jalapeno Early'), One trial, repeated
Crop #3: Tomato, determinate (Solanum lycopersicum var. 'BHN 589'), One trial, repeated
Files:
Tomato-Layout Greenhouse Placard for Tomato Round #1, #2
SebringSeminar2019 Presentation of Preliminary results as part of Penn State Plant Science Spring Seminar Series



Crop #1: Sweet peppers
Nitrogen level was significant for all responses, but did not interact with inoculation. All responses hence with will be regarding pooled inoculation status. Sweet pepper yields increased by 8.8 peppers per plant with inoculation (p<0.0004), from an average of 39.8 peppers in NON plants and 48.6 in INOC plants, an increase of 22%. The total fresh weight of all peppers harvested also increased with inoculation by 12.1% (p<0.0357), with no differences detected in mean or median fresh fruit weight. Dry weight was similarly increased with INOC by 13.1% (p<0.0305) over untreated plants. Root material accumulation was not impacted by inoculation status, but leaf and stem tissue increased 25.8% over controls from 118.7 g DM in NON to 149.4g in INOC (p<0.0001). Overall biomass, the aggregate total of fruit, shoot, and root DM, was subsequently greater with inoculation than with controls (p<0.0039). Thus, inoculation of sweet pepper can increase fruit number and biomass accumulation leading to more harvestable yield, with the following caveat.
For sweet pepper, responses to nitrogen did not indicate any nutritive equivalency was achievable by replacing some fertilizer N with biologically sourced N. As such, no evidence of N fixation was observed in INOC plants. The net results demonstrate that at any particular fertilizer level the presence of G. diaz can increase the yield of peppers produced, both in count and biomass. However, assessment of quality revealed significant aberrations in fruit quality across all treatments, which was likely a result of the growing conditions. Large fractions of both control and treatment fruits exhibited internal proliferation, excessive folding and enveloping, and poor blossom end closure. Quality imperfections such as these reduce USDA-AMS quality to off/sub-par grade which can result in lower commodity prices at best, or outright rejection and disposal at worst. These defects were universal and not statistically significant between treatments, indicating the experimental set-up led to their emergence. Pepper cultivation in DWC is a common practice and the fertilizers used were standard hydroponic grade, so the presence of vigorous 24/7 solution oxygenation, a non-standard practice, may have effected the changes. Unfortunately, their presence confounds determining if there were any impacts from bacterial sourced PGRHs, which are reported to have similar albeit less severe effects.



Crop #2: Hot Peppers
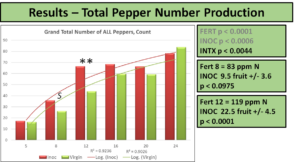
Nitrogen level was significant for all responses and interacted with some measures when indicated. If N level is not specifically mentioned, a response will be regarding pooled inoculation status. Hot pepper count yields saw an interaction between fertilization level and inoculation status, as seen in the following diagram. At 83 and 119 ppm N significant and near-significant differences between the count total of peppers produced were recorded, with more peppers in INOC plants
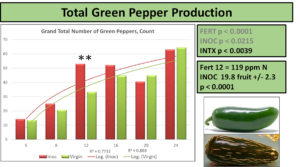
When partitioned by color of the pepper, the differences at 119 ppm are attributable to an increase in green fruit count production with no increase in red pepper numbers, as seen below.
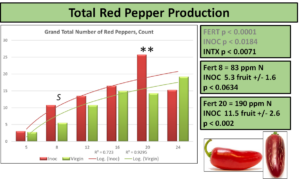
Interestingly, when comparing just peppers produced that were red, the differences at 83 ppm N are accounted for, but a new significant difference emerges at 190 ppm N. This level corresponds to the normal fertilization level recommended for hydroponic production, and demonstrates the greatest increase in pepper count production at any fertilization level, a 79% increase over NON. Red peppers are not rated any differently than green by USDA-AMS guidelines, but can be voluntarily reported as certain markets prefer coloration.
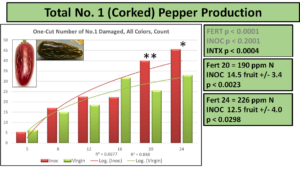
The USDA-AMS does grade hot peppers, and especially jalapenos, by external defects. One such defect, known as corking, is described by white miniature cracking lines formed on the exterior of the fruit. Although often sought after in foreign markets, the USDA considers these quality reducing imperfections and grades accordingly, as seen in the following diagram.
Grade A fancy typically demands the highest commodity pricing, with subsequent reductions for No 1. and No 2. In assessing bacterial impacts on fruit quality, a significantly higher counts of peppers were found to exhibit corking and thus would be graded as lower quality, as seen below.
At the recommended or enriched fertilization levels, more fruits from INOC plants exhibited damage and thus would be graded as No. 1 or No. 2. Under no categorical count response was the total dry matter of pepper production significant, meaning if a greater or lesser number of peppers were produced, those peppers were concomitantly smaller or larger as a counterbalance. Therefore an operator would have to decide between reduced count yields of hot peppers if he wanted green, Grade A fruits, or a large amount of corked and potentially red peppers if he wanted to maximize counts. In either situation, the biomass being marketed would not be impacted.
Total biomass accumulation in leaf/stem tissue and/or root tissue was not impacted by inoculation for hot pepper. Although samples were saved for vitamin, mineral, and secondary metabolite analysis, COVID-19 effected building and laboratory closures prevented the processing of those samples and data has yet to be collected. Should subsequent research on these sections be performed, SARE will be informed of the findings in an addendum report.
Crop #3: Tomatoes
Tomatoes did not exhibit any significant differences attributable to inoculation status; the only significant differences were from N fertilization level. Yields from both INOC and NON plants both generally increased over increasing N level, with no significant plateau observed even at the supra-optimal N level. Both bacterially treated and untreated plants exhibited high proportions (21.0-45.5%) of fruit with blossom end rot, a surprising finding as Ca was supplied well above established nutritional requirements. Otherwise the plants responded typically to N level deficiencies, and fruit formed was as described for this protected cultivar (5-6 oz average, singly per joint). Internal fruit defects were not noted.




Sweet peppers benefit from inoculation by Gluconacetobacter diazotrophicus with a greater number of fruits and total biomass accumulation in fruit and aerial tissues. Incorporation into a production system may not yet be advised as significant numbers of fruits exhibited aberrant growth characteristics, but these were likely attributable to the cultivation techniques rather than bacterial-plant interactions. Further research at scale in situ may help determine if these defects are ameliorable, and if they are, this synergistic bacteria can significantly improve crop yields of sweet pepper.
Hot pepper production is impacted by G. diaz inoculation, but with results that are academic rather than applicable. Bacterial incorporation reduced overall fruit quality at production levels and only drove marketability-embracing improvements at levels of N fertilization that significantly reduced total yields. The trade-offs for producers are likely too great at current commodity prices, as even the increase in grading is not offset by yield losses. More research into secondary metabolite production may be warranted if marketability and profitability does not hinge on visual quality but on chemicals produced in planta.
Tomato production, contrary to emerging literature, was not impacted by inoculation. The techniques employed in this research, including aggressive 24/7 solution oxygenation may have altered plant growth response, despite being beneficial for bacterial proliferation. The research herein is likely then to only serve as an example of a DWC cultivation technique that should be avoided in future experiments.
Education & outreach activities and participation summary
Participation summary:
Two tours given to PSU student farm collaborators and student members, ~15 participants total, as part of agricultural recruitment and interest building demonstrations. Both Fall 2019.
Seminar 4/9/2021 presented via Zoom to unknown number of participants (Recorded, online via youtube)
Seminar from 2019 touching on preliminary pepper data
Upcoming journal articles:
Sebring, R.L., R.B. Bryant, J. Regan, J. Lambert, and S.W. Duiker. Biomass and Quality Impacts of Gluconacetobacter diazotrophicus in
Jalapeno Pepper, Sweet Pepper, and Greenhouse Tomato. (Manuscript in production)
Sebring, R.L., R.B. Bryant, and J. Lambert. Water soluble vitamin production in tomato inoculated with Gluconacetobacter diazotrophicus (Manuscript in production)
Sebring, R.L., R.B. Bryant, and J. Lambert. Tiny Bugs imparting a Hefty Bite: Jalapeno pepper inoculated with Gluconacetobacter diazotrophicus produces more piquant capsaicinoid compounds. (Manuscript in production)
Upcoming Dissertations:
Sebring, R.L., R.B. Bryant, R. Berghage, J. Lambert, J. Regan, and S.W. Duiker. Impacts Of The Bacterial Endophyte Gluconacetobacter
Diazotrophicus On Three Agriculturally Important Crops. PhD Dissertation. The Pennsylvania State University. 2011.
Project Outcomes
This project may form part of a foundation of exploration into the use of biological synergists in cropping systems. While the results were mixed or neutral, the ease of incorporation into existing practices lends merit to at least trying new approaches. Hurdles to adoption of new technology can be financial, physical, social, or even cultural. This research demonstrates how bacteria inside plants are not always a negative condition, and can even bring about some improvements in certain situations. What we found may not be impact wallets, but if we can erode negative perceptions of 'bugs' we can count that as a success, especially if further research into biologicals is to continue.
The primary knowledge gained outside of raw significance data was the difficulty in applying new 'sustainable' techniques to existing crop production methods. The literature on pepper production using this bacteria at the time of inception was non-existent, so we employed a tried and true method in DWC buckets but still had results that could not be fully explained. My advisor and I are both still optimistic when it comes to inclusion of biologicals into fertilization schemes, but the difficulty in replicating established yields, especially as they pertain to quality and not just quantity, is daunting. I especially learned that short duration, micro-experiments to test proof of concepts are invaluable before long-term whole plant lifecycle projects should be undertaken. The aeration scheme we employed may have been too aggressive, for example, and it would have been better to find that out after two weeks rather than 18.
I am looking to continue my research into biologicals as plant fertilization sources, as this SARE funded research culminated with my earning of a PhD in Horticulture. I will be looking at different avenues of G. diaz modulated improvements should I continue with this bacterium, as current literature is leaning more towards significant impacts it has on water use efficiency (instead of N fixation). The need for sustainable intensification still looms large on the agricultural horizon, so other technologies that demonstrate promise may also secure my attention, but sustainability will be central despite my chosen techniques.