Final report for GNE21-254
Project Information
Problem and objectives Modern agriculture often creates inhospitable environments for beneficial arthropods, including simplified habitats that lack essential resources such as food and shelter1. Pollinators and natural enemies of crop pests require plant-derived resources which are often limited in monoculture cropping systems and are only available for a finite period of time. Two on-farm practices that may be used to enhance floral resources include adding flower strips along the crop border and interplanting flowering plants (i.e., growing different plant species between cultivated crop rows) within the crop field. The benefits of both practices are widely recognized and have been shown to improve soil quality, suppress weeds, and enhance pollination and pest control services within a crop2–5.
Although floral additions may restore ecosystem services within a crop, little attention has been given to how these on-farm tactics affect ecosystem services in neighboring habitats (e.g., grasslands, forests). For example, studies have shown that flowering crops can dilute pollinator densities within neighboring grasslands, subsequently destabilizing pollinator communities and negatively impacting the reproductive success of concurrently flowering wild plants6–9. To date, little research has concomitantly studied the impact of interplanting crops with flowering plants and adding floral strips along crop borders, and fewer studies have investigated the broader impacts of floral additions to neighboring habitats. This proposal seeks to understand the effects of enhanced floral diversification on arthropod-mediated ecosystem services within the crop and surrounding habitat. Specifically, objectives of this project were to quantify effects of floral diversification on (i) beneficial arthropod abundance and diversity within the crop and neighboring plants; (ii) reproductive output of the crop and neighboring wild plants, and (iii) edamame fruit damage from insect pests.
Research findings Edamame (vegetable soybean; Glycine max (L.) Merr.) was our test crop. Edamame benefits from cross pollination and our recent work supports that these benefits include greater fruit weight and a higher proportion of commercial grade-A fruit among cross-pollinated fruits10. To evaluate the effect of floral supplementation on native plant communities in areas surrounding the crop fields, we selected two plant species native to Maryland to portray wild plants found in the neighboring habitat: partridge pea (Chamaecrista fasciculata) and Culver’s root (Veronicastrum virginicum). In relation to goal (i), we found that the presence of floral supplements significantly enhanced arthropod abundance and diversity in the supplemented edamame plots compared to the control plots. For example, we observed significant increases in biocontrol agents and native pollinators, particularly Bombus species, and higher parasitism rates of sentinel eggs masses. Bombus species were also among the most common visitors to the wild plants (C. fasciculata and V. virginicum). In relation to goal (ii), we found that fruits harvested from edamame plots interplanted with clover weighed as much as or less than fruits harvested from the control plots (edamame monoculture). This trend is likely due to plant-plant competition between edamame and clover, and the less than average precipitation early in the growing season (May-June). The presence of floral supplementation had no effect on V. virginicum seed weight. However, seed weight of C. fasciculata was significantly reduced in the presence of floral supplementation, patricianly at distances farthest away from the floral supplements (50m). We suspect that the clover interplantings and flower strips acted as a pollinator sink, concentrating pollinators of C. fasciculata within the floral supplements. In relation to goal (iii), we did not observe significant damage to the fruits in either treatment.
Impact on Agricultural community The knowledge gained from this project can be used to help edamame farmers better understand how to manage pests and improve yield, all while supporting biodiversity in their systems. Our work also provided support to the importance of considering the benefits and problems associated with the coordinated management of multiple ecosystem services rather than managing ecosystem services independently. For example, while the establishment and maintenance of pollinator habitats appeared to improve pest control agents and pollinator biodiversity, in some cases these habitats competed with the crop, adversely affecting crop yields and wild plant reproduction.
References
- Shackelford, G. et al. Comparison of pollinators and natural enemies: a meta-analysis of landscape and local effects on abundance and richness in crops. Biological Reviews 88, 1002–1021 (2013).
- Mallinger, R. E., Franco, J. G., Prischmann-Voldseth, D. A. & Prasifka, J. R. Annual cover crops for managed and wild bees: Optimal plant mixtures depend on pollinator enhancement goals. Agriculture, Ecosystems & Environment 273, 107–116 (2019).
- Snapp, S. S. et al. Evaluating Cover Crops for Benefits, Costs and Performance within Cropping System Niches. Agronomy Journal 97, 322–332 (2005).
- Bryan, C. J. et al. Efficacy of Cover Crops for Pollinator Habitat Provision and Weed Suppression. Environmental Entomology 50, 208–221 (2021).
- Kahl, H. M., Leslie, A. W. & Hooks, C. R. R. Effects of Red Clover Living Mulch on Arthropod Herbivores and Natural Enemies, and Cucumber Yield. Annals of the Entomological Society of America 112, 356–364 (2019).
- Gardner, E. et al. Field boundary features can stabilise bee populations and the pollination of mass-flowering crops in rotational systems. Journal of Applied Ecology 58, 2287–2304 (2021).
- Holzschuh, A., Dormann, C. F., Tscharntke, T. & Steffan-Dewenter, I. Expansion of mass-flowering crops leads to transient pollinator dilution and reduced wild plant pollination. Proc. R. Soc. B 278, 3444–3451 (2011).
- Montero-Castaño, A., Ortiz-Sánchez, F. J. & Vilà, M. Mass flowering crops in a patchy agricultural landscape can reduce bee abundance in adjacent shrublands. Agriculture, Ecosystems & Environment 223, 22–30 (2016).
- O’Connor, R. S. et al. Monitoring insect pollinators and flower visitation: the effectiveness and feasibility of different survey methods. (2019).
- Evans, K., El-Hifnawi, J., Hooks, C. & Espíndola, A. Benefits of Cross-Pollination in Vegetable Soybean Edamame: Cross-Pollination in Vegetable Soybean Edamame. Journal of Pollination Ecology 35, 329–340 (2023).
- Rand, T. A. & Louda, S. M. Exotic Weed Invasion Increases the Susceptibility of Native Plants to Attack by a Biocontrol Herbivore. Ecology 85, 1548–1554 (2004).
- IPBES. Assessment Report on Pollinators, Pollination and Food Production. IPBES http://ipbes.net/assessment-reports/pollinators (2016).
This proposal seeks to understand how floral diversification practices (i.e., the addition of wildflower strips and floral intercropping) within and bordering the crop, affect (1) the diversity and density of beneficial arthropod communities (natural enemies and pollinators), within the crop and in the surrounding habitat, and (2) pest control and the reproductive output of wild plant communities and crops. Specific objectives and sub-objectives are to:
Objective 1. Quantify the effect of floral additions (i.e., wildflower strips and floral intercropping) on the abundance and diversity of beneficial arthropods in designated croplands and in neighboring wild plant communities. I hypothesize that arthropod diversity and abundance will increase in plots with higher combined flower density (intercropping and wildflower strip).
Objective 2a. Assess how floral additions affect pest control and the reproductive output of crops and non-target (i.e., non-crop) wild plants in the neighboring landscape. I hypothesize that both pest control and the reproductive output of crops and wild plants will increase with proximity to floral additions.
Objective 2b. Evaluate the spatial scale (e.g. distance) at which the floral additions affect beneficial arthropod diversity and abundance, and translate to direct ecosystem services (i.e., pest control, plant reproduction) by measuring the limits of the spillover effect of floral additions on the crop and the natural habitat. I hypothesize that increases in the diversity and abundance of arthropods is local and will decrease with increasing distance from the wildflower strips. I also hypothesize that the addition of wildflower strips and floral intercropping will lead to increased competition for ecosystem services between plants in natural and managed lands, leading to a reduction of ecosystem services in those plants that are the furthest away from the wildflower strips.
The purpose of this project is to quantify the beneficial effects of enhanced floral diversification on ecosystem services within a crop and the surrounding habitat. The loss of natural and seminatural habitats associated with expanded agricultural acreage and an associated increase in monocultural plantings has resulted in reduced floral diversity in landscapes1, which has been shown to negatively affect beneficial arthropods2. Indeed, pollinators and natural enemies of crop pests require plant-derived resources which are often limited in monoculture cropping systems and may only be present for a finite period. Two practices that may be used to enhance these resources in agricultural fields include adding wildflower strips along the crop border and intercropping flowering plants within the crop field. These practices are known to help with weed suppression, and enhance biological control and pollination activity in the crop fields3,4. Flowering plants situated around the perimeter of a crop enhance and support natural enemies and pollinators by providing them food and refuge throughout the season2. Further, intercropping (i.e., growing different plant species between cultivated crop rows) has been shown to improve soil quality, suppress weeds and attract natural enemies4. Some of these intercropped plants (e.g., clover) can also attract pollinators and natural enemies, enhancing crop pollination, pest suppression, and crop yield3,5,6.
Although increasing floral resources could restore ecosystem services within a crop, little research attention has been directed at determining how these newly-added resources can affect ecosystem services in neighboring plant habitats. For example, pollinator visitation increases with increased floral density and diversity7,8. However, it is unclear as to what extent such an increase in pollinator visitation from the floral additions along crop borders and within crop rows will spillover into target crops and native plants in the surrounding habitat9.
Further, floral strips may also act as pollinator sinks, concentrating pollinators and pollination services from the surrounding habitats within the floral strip8–10. This could result in competition for pollinators between crops and wild plants, negatively impact the reproductive output of plant communities in neighboring habitats10, and indirectly affect food webs11. Ultimately, natural areas are sources of pollinators and biological control agents, and changes in resource availability and floral diversity could affect this spillover from natural to managed systems9. This, in turn, could have lasting implications on pest populations and ecosystem services in cropping systems. However, to date little research has been conducted on spillover effects in agroecosystems from managed to natural areas9.
Given the current pollinator crisis12, it is important that the effects of these practices are quantified beyond the crop-field. To date, virtually no research has been done to quantify the combined effects of using wildflower strips and intercropping to enhance natural enemy efficacy and pollination within crop fields and surrounding habitats. This project proposes to address these knowledge gaps and concomitantly develop pest suppression practices that are more congenial to pollinators and natural enemies (predators and parasitoids) within and outside the crop field, thus enhancing the sustainability of the entire agroecosystem.
Cooperators
- (Educator and Researcher)
Research
Project Objectives This proposal seeks to understand how floral diversification practices (i.e., the addition of wildflower strips and floral intercropping) within and bordering the crop, affect (1) the diversity and density of beneficial arthropod communities (natural enemies and pollinators), within the crop (edamame) and in the surrounding natural habitat, (2) pest populations and (3) the reproductive output of wild plant communities and crops.
Research and Results This was a two-year project. We originally proposed to collect data from 12 plots among four separate UMD research farms, but underestimated the labor intensive nature of the project and logistical challenges in coordinating research at multiple locations. We therefore decided to place all 12 plots at a single location at the University of Maryland Beltsville Agricultural Research Center. Data was collected biweekly, April-October, and included (1) hand-netting floral visitors of edamame, wildflower strip and wild plants, (2) sweep samples, (3) sticky cards, (4) passive trapping of potential pollinators with bee bowls, and (5) harvesting edamame pods and wild plant seeds to estimate plant reproductive output and yield. The field set-up consisted of two treatments (mowed strip without intercropping (control) or floral strip with floral intercropping (floral treatment)), each replicated six times (12 plots/year). In our study, the wild plant communities were represented by two species native to the Mid-Atlantic: Culver’s root (Veronicastrum virginicum) and partridge pea (Chamaecrista fasciculata). On either side of the (mowed/floral) strip, we placed each of these three plant species at three distances from the strip (10m, 30m, 50m). The floral supplements included a (1) wildflower strip composed of 17 locally-sourced flowering plants that are mostly native to the Mid-Atlantic region and (2) clover intercropping.
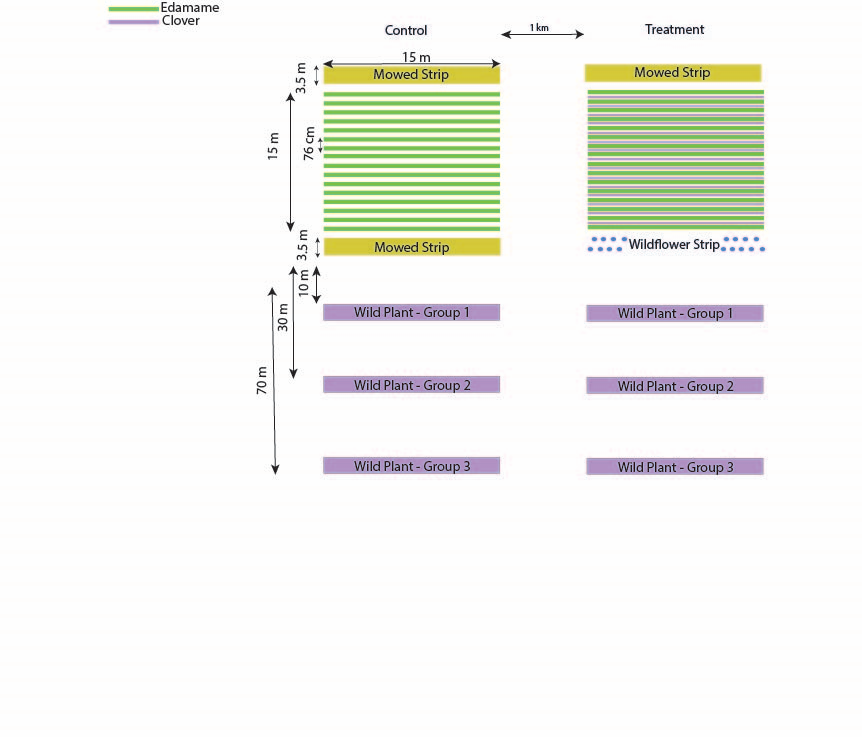
On the farm, each treatment will be separated by a minimum of 500m.
Edamame benefits from cross pollination, and our recent work supports that these benefits include greater fruit weight and a higher proportion of commercial grade-A fruit among cross-pollinated fruits. We found that the presence of floral supplements significantly enhanced arthropod abundance and diversity within the supplemented edamame plots compared to the control plots. For example, we observed significant increases in biocontrol agents and native pollinators, particularly Bombus species, and higher parasitism rates of sentinel eggs masses. Bombus species were also among the most common visitors to the wild plants (C. fasciculata and V. virginicum). In relation to edamame yields, we found that fruits harvested from edamame plots interplanted with clover weighed as much as or less than fruits harvested from the control plots (edamame monoculture). This trend is likely due to plant-plant competition between edamame and clover, and the less than average precipitation early in the growing season (May-June). The presence of floral supplementation had no effect on V. virginicum seed weight. However, seed weight of C. fasciculata was significantly reduced in the presence of floral supplementation, patricianly at distances farthest away from the floral supplements (50m). We suscept that the clover interplantings and flower strips acted as a pollinator sink, concentrating pollinators of C. fasciculata within the floral supplements. Regarding crop damage, we did not observe significant damage to edamame fruits in either treatment.
Goals (i) and (iii):
Our results suggest that combining flower strips and floral interplantings attracts a greater abundance and diversity of pollinators and parasitoids compared to crops planted as monocultures. The three most common visitors to edamame flowers included Bombus, Apis mellifera and Popillia japonica, comprising over 75% of edamame flower visitors. Visitation in the supplemented plot were 2.5 times greater than in the control plots. Bombus species were also among the most common visitors to the wild plants (C. fasciculata and V. virginicum) and to the clover interplantings. Given the reductions in seed weights for C. fasciculata, we hypothesize that the clover interplantings acted as a pollinator sink, reducing floral visitations by Bombus. However, more data on pollinator visitation rates is needed to confirm our hypotheses.
The most common flower visitors in the flower strip were Scoliidae and Halictidae, comprising over 50% of floral visitors. Scoliid wasps are important biocontrol agents, as many of the beetles they parasitize are pests, including the Japanese beetle, Popillia japonica, which were common in our plots. We also observed that Scelionidae japonicus was the most common parasitoid of the brown marmorated sentinel eggs, with the majority of parasitism occurring in the floral supplemented plots. For both the floral supplemented and control plots, there was little damage to the fruits.
Overall, our results support the notion that increases in plant biodiversity in the Mid-Atlantic support a variety of beneficial arthropods including pollinators and natural enemies that in turn, benefit crops. However, care should be taken as to which species to interplant to minimize the competition between species.
Goal (ii):
To evaluate the effects of floral supplementation on edamame reproductive outputs, we first characterized the reproductive strategy of vegetable soybean (edamame; Glycine max). Our results indicate that although edamame can self-pollinate, it benefits from cross-pollination and likely from floral visitation in measurable and market-relevant ways1. These benefits include greater fruit weight (Figure 1) and a higher proportion of commercial grade-A fruit among cross-pollinated fruits, with some of these effects accentuated with proximity to the flower strip (Figure). Despite these positive effects of the flower strip, we did not see the same trend in edamame plots interplanted with clover. Fruits harvested from edamame plots interplanted with clover weighed as much as or less than fruits harvested from the control plots (edamame monoculture). This trend is likely due to plant-plant competition between edamame and clover, and the less than average precipitation in the Mid-Atlantic early in the growing season (May-June) during the years of the implementation of this project.
In relation to the effect of floral supplementation on native plant communities in areas surrounding the crop fields, the presence of floral supplementation had no effect on V. virginicum seed weight. However, seed weight of C. fasciculata was significantly reduced in the presence of floral supplementation, especially at distances farthest away from the floral supplements (50m). Specifically, the reduced reproductive output of C. fasciculata may be the result of the flower strip and clover interplantings acting as a biodiversity sink, concentrating pollinators and their services within these floral supplements.
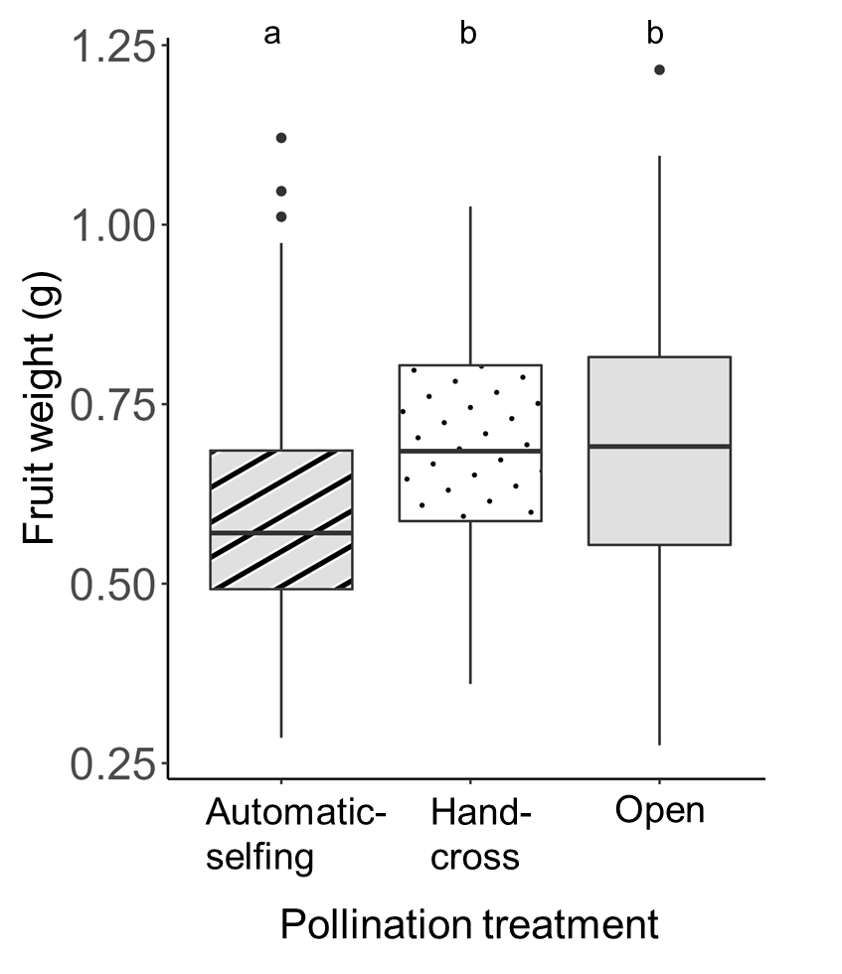
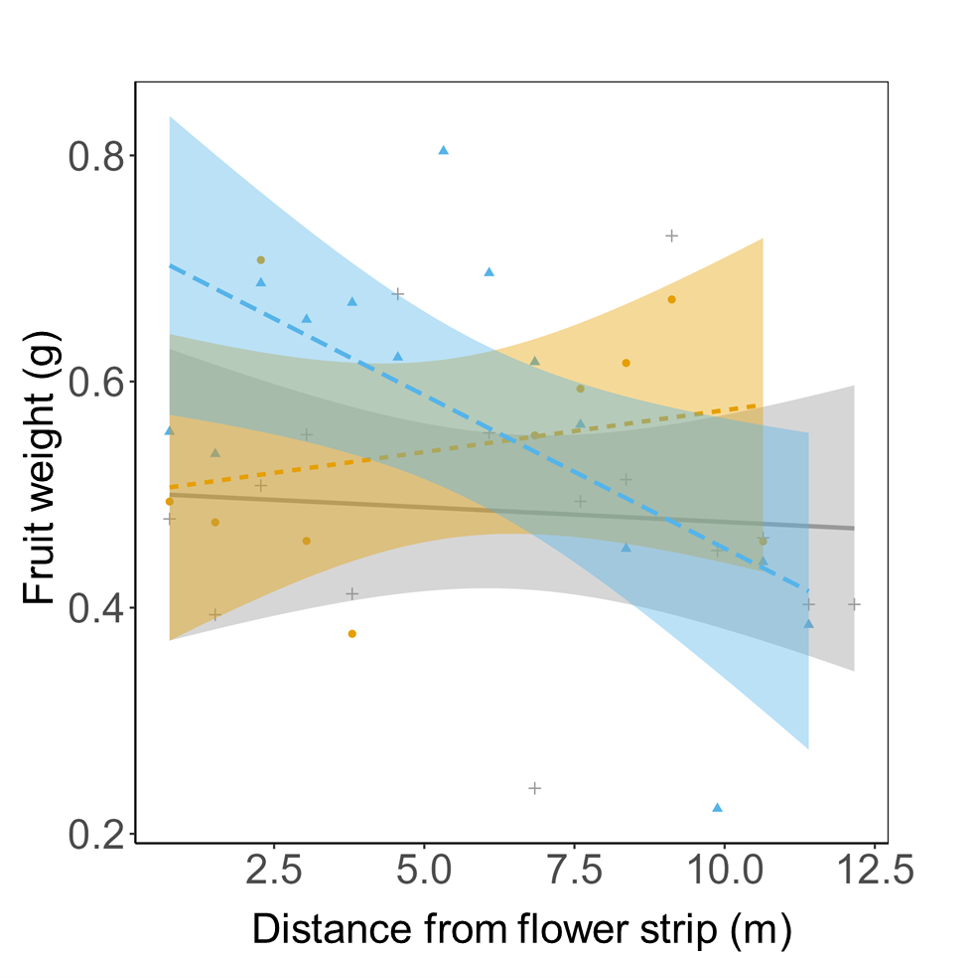
Reference
1. Evans, K., El-Hifnawi, J., Hooks, C. & Espíndola, A. Benefits of Cross-Pollination in Vegetable Soybean Edamame: Cross-Pollination in Vegetable Soybean Edamame. Journal of Pollination Ecology 35, 329–340 (2023).
Education & outreach activities and participation summary
Participation summary:
We presented our research at the Entomological Society of America Eastern Branch in March 2022. We also presented aspects of our work at a MOFFA (Maryland Organic Food and Farming Association) farm tour on Maryland's Eastern Shore in summer 2022 and at the UMD farm tour at Upper Marlboro in August 2023. We published our work in the Roots in Research Newsletter (Flower Power: Floral Diversity Attracts Beneficial Arthropods in an Edamame Agroecosystem), UMD Extension Maryland Grows Blog (Floral supplements support ecosystem services in our green spaces), and will be presenting parts of our research at a Master Gardener meeting in Montgomery County, MD in May 2024. In summer 2023, I created a short outreach video for the College of Mathematical and Natural Sciences about this research (https://www.instagram.com/reel/CwiWSJMNPY2/?utm_source=ig_embed&ig_rid=46cc8664-e171-4a69-b368-16d415cbd29b) and in December 2023, a press release was published about our peer-reviewed article regarding the benefits of cross pollination in edamame: Pollinators make a big impact of edamame Marketability. Through this project, we also provided ample research opportunities in the field and lab for undergraduate students (twelve students total).
Project Outcomes
Our results support the notion that increased plant biodiversity in the Mid-Atlantic support a variety of beneficial arthropods, including pollinators and natural enemies, which can benefit crops and farmers. In the case of edamame, we show that such increases can lead to higher marketability of the product, and a potential for increased net-returns for farmers. We believe that this benefit can be extended to grain soybean, given that grain soybean and edamame are the same species. In terms of the effect of these practices on semi-natural habitats surrounding the crops, our results indicate that although these practices tend to benefit biodiversity in crops, their effect is mixed for wild plants present in the agroecosystem. This is something that should be explored more in the future, since this could affect the “global” sustainability of floral supplementation and, potentially, the long term maintenance of ecosystem functioning in the agroecosystem.
Our strategy of combining living mulches with floral strips also yielded mixed results, with reductions in yields from potential competition between the clover and the crop. If our results are confirmed, this would indicate that although successful at increasing biodiversity and likely controlling weeds, the practice should be at least modified to reduce such competition. Options could be the choice of different living mulches that will compete less than clover, changes in the spacing and density of clover planting, and other containment measures such as mowing at certain times of the season.
Finally, the choice of annual (native) plant species for the flower strip can also be beneficial to farmers. In contrast to the use of perennial floral mixes, our use of annual species makes the establishment of the strip faster, which can provide services in the first growing season. This also allows rotating the strips annually and in tandem with crop rotations. This is often the case in small vegetable farms, where crop rotation is common and there is little space and time for perennial mixes to establish.
Throughout the course of this project, I have gained valuable knowledge about sustainable agriculture. For example, I gained practical experience on the implementation of different practices that are usually recommended in the framework of sustainability. This experience allowed me to better understand challenges and solutions for their use, and gave me greater insight into how to better promote them among future users.
My future research will integrate plant-animal interactions with sustainable pest management tactics in human managed ecosystems. This project has expanded my research portfolio and broadened my knowledge of sustainable management practices, including pest control and pollinator enhancement. This grant has for this reason allowed me to explore and continue what I believe will be a career in agroecosystem studies.
The project has already had several outputs, which include one peer-reviewed publication in the Journal of Pollination Ecology and two more in preparation, and one blog published on the UMD Extension Maryland Grows Blog.