Progress report for GNE24-317
Project Information
Insects and other arthropods are pivotal components of agricultural landscapes. They contribute to ecosystem services such as pollination, but can also cause devastating crop damage as pests. Impacting the effect of their interactions with crops, arthropod diversity is associated with many factors, among which are the local (e.g. plant, plot, field) and landscape (garden, surrounding ecosystems) habitats described by factors including plant diversity and land uses. Though a growing literature investigates the effects of local and landscape factors on the arthropod community, limited research has focused on these dynamics in urban agriculture, a particularly complex and heterogeneous landscape.
This study aims to better understand the effects of plant diversity and site characteristics at different spatial scales on the arthropod community composition in urban community gardens, which are increasingly recognized for their social and ecological benefits. To capture the diversity of agriculturally significant arthropod groups, we designed surveys of crop pests and herbivory and flower-visiting insects in gardens with different plant management intensities across spatial scales. We collected crop pests and herbivory during the harvest season, as well as local and landscape scale site characteristics in the community gardens, such as plant diversity, through on-site observation. We surveyed pollinators during the growing season and collected plant diversity and site characteristics using parallel methods. We will further collect garden sizes and the amount of green space in the neighborhood using remote sensing methods. By analyzing and determining crucial factors and scales that arthropod communities respond to, we aim to inform sustainable management practices in urban community gardens to optimize beneficial insects and reduce pest impacts while conserving the overall arthropod community through collaborations with urban gardeners, garden managers, and city planners.
Objective 1: To determine the role of plant diversity and site characteristics in affecting arthropod diversity in different functional groups (focusing on pests and pollinators) in urban community gardens. Using targeted sampling methods, we collected pests and other arthropods on crops in individual gardeners’ plots to assess arthropod species richness and abundance. We further collected plant diversity through plant censusing and site characteristics to describe individual gardeners’ plots and the gardens. In examining pest richness and abundance, we also included herbivory as an additional measure that directly reflects crop damage and influences gardeners’ management and crop yield. By connecting herbivory and pest communities, we aim to deepen our understanding of the impact on gardeners of potential response in pest communities to landscape factors. We collected pollinator richness and abundance along with plant diversity and site characteristics in the growing season in corresponding plots to understand the impact on pollinators and pollination services.
Objective 2: To investigate the effect of spatial scales of plant diversity and site characteristics on arthropod diversity (pests and pollinators). By using plant diversity collected at the plot level scale, and the garden plant diversity estimated from the samples of plot level diversity, we will compare the effect size and significance of the effect size of plant diversity at the two levels on arthropod richness and abundance. Site characteristics describing land uses and plant communities will be collected at the individual plot level scale, garden level scale, and neighborhood scale (from a buffer distance of the garden) and we will compare the effect size on the arthropod community.
Objective 3: To synthesize the effects of plant diversity and site characteristics on arthropod diversity in the urban community gardens for biodiversity support of different ecosystem functions, and to compare if and how pollinators and pests respond differently.
The purpose of this project is to better understand the plant diversity and site characteristics at different spatial scales that affect arthropod community composition in urban community gardens. This work will inform managers and gardeners of best practices to optimize the landscape for sustainable management of arthropods.
Insects and other arthropods are an important part of agricultural landscapes, serving essential roles in the success of crops, and other plants and animal communities in the agroecosystems (Pedigo et al., 2021; Weisser & Siemann, 2008). Pollination, for example, is a fundamental ecosystem service integral to the fruit set of many crops, valued at US $215 billion globally in 2005 (Gallai et al., 2009; Losey & Vaughan, 2006; Vanbergen & Initiative, 2013). Meanwhile, arthropod pests destroy an estimated 18 - 26% of annual crop production worldwide, with damages valued at US $470 billion (Culliney, 2014), making pest control an integral management aspect in crop production. Although insects and other arthropods can affect agricultural production, the reverse is also true. Agricultural intensification is a major driver that contributes to the declines in arthropod diversity and abundance worldwide, with most well-documented cases in Europe recording more than 75 percent decline over 27 years (Hallmann et al., 2017; van der Sluijs, 2020; Wagner, 2020). Land use changes associated with agricultural intensification in particular have led to drastic changes in arthropod community composition, through both direct losses of habitats and the effects of the spatial patterns in the newly created landscapes. (Crist et al., 2006; Outhwaite et al., 2022). In these landscapes, spatial heterogeneity created by different land use fragments provides suitable and unsuitable patches of habitats, and barriers and corridors that dictate insect population persistence and movement (De Carvalho Guimarães et al., 2014).
In response to the decline in arthropod biodiversity and abundance and the complex feedback between arthropods and agriculture, the management of the heterogeneous landscape around agriculture, namely the agricultural matrix, has become a new focus (Vandermeer & Perfecto, 2007). Plant diversity, land use fragment types, sizes, landscape configurations, and spatial connectivity can all impact arthropod community composition through the provision of resources and habitats (Vandermeer & Perfecto, 2007; e.g. Diekötter et al., 2008; Lemessa et al., 2015). Pollinator diversity, for example, benefits from local diverse floral resources and the amount of high-quality habitats on the landscape scale, while being less impacted by the configuration of habitats (Kennedy et al., 2013). The diversity and abundance of natural enemies that are associated with lower pest abundance (Letourneau et al., 2009) have often been found to respond positively to complex structures of vegetation locally, but may respond negatively to land cover diversity based on the insect groups and their use of resources and habitats (Holzschuh et al., 2010; Karp et al., 2018; Martin et al., 2016). Pest diversity is often correlated with local plant diversity due to the evolutionary history, but mixed effects have been seen with respect to pest abundance and further overall crop damage (Chaplin-Kramer et al., 2011; Dangles et al., 2009). These studies have provided a basis for understanding landscape management for insects and other arthropods in agriculture.
However, studies establishing these trends have been conducted primarily in rural areas, while less is known in urban agriculture which has become increasingly important for addressing challenges in cities’ access to fresh food and nature (Kuddus et al., 2020). In urban ecosystems, insects and other arthropods face a unique landscape, often inhabiting the rare green spaces surrounded by urban environments. Urban community gardens as a major form of urban agriculture create a landscape where land use fragments of different quality and size may transition sharply at different spatial scales. From local plots that each gardener manages differently, to the gardens bordering roads and buildings, to neighborhoods where additional green spaces exist to provide additional habitats, arthropods of different groups may see and use the landscape differently, contributing to the complexity of management in urban community gardens (Barr et al., 2021; Ong et al., 2020).
In this study, we aim to understand the impacts of factors including local and landscape plant diversity, land use patches and configurations on arthropod diversity. Specifically, we focus on plant diversity and site characteristics that describe spatial patterns at different spatial scales in urban community gardens and investigate their effects on pollinators and pests, one beneficial and one detrimental group of agriculturally significant arthropods. This study will help inform sustainable management of the landscape to optimize agricultural productivity and biodiversity conservation.
References
Barr, A. E., L. J. A. van Dijk, K. Hylander, and A. J. M. Tack. 2021. Local habitat factors and spatial connectivity jointly shape an urban insect community. Landscape and Urban Planning 214:104177.
Chaplin-Kramer, R., M. E. O’Rourke, E. J. Blitzer, and C. Kremen. 2011. A meta-analysis of crop pest and natural enemy response to landscape complexity. Ecology Letters 14:922–932.
Crist, T. O., S. V. Pradhan-Devare, and K. S. Summerville. 2006. Spatial variation in insect community and species responses to habitat loss and plant community composition. Oecologia 147:510–521.
Culliney, T. W. 2014. Crop Losses to Arthropods. Pages 201–225 in D. Pimentel and R. Peshin, editors. Integrated Pest Management: Pesticide Problems, Vol.3. Springer Netherlands, Dordrecht.
Dangles, O., V. Mesías, V. Crespo-Perez, and J.-F. Silvain. 2009. Crop damage increases with pest species diversity: evidence from potato tuber moths in the tropical Andes. Journal of Applied Ecology 46:1115–1121.
De Carvalho Guimarães, C. D., J. P. R. Viana, and T. Cornelissen. 2014. A Meta-Analysis of the Effects of Fragmentation on Herbivorous Insects. Environmental Entomology 43:537–545.
Diekötter, T., R. Billeter, and T. O. Crist. 2008. Effects of landscape connectivity on the spatial distribution of insect diversity in agricultural mosaic landscapes. Basic and Applied Ecology 9:298–307.
Gallai, N., J.-M. Salles, J. Settele, and B. E. Vaissière. 2009. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecological Economics 68:810–821.
Hallmann, C. A., M. Sorg, E. Jongejans, H. Siepel, N. Hofland, H. Schwan, W. Stenmans, A. Müller, H. Sumser, T. Hörren, D. Goulson, and H. de Kroon. 2017. More than 75 percent decline over 27 years in total flying insect biomass in protected areas. PLOS ONE 12:e0185809.
Holzschuh, A., I. Steffan-Dewenter, and T. Tscharntke. 2010. How do landscape composition and configuration, organic farming and fallow strips affect the diversity of bees, wasps and their parasitoids? Journal of Animal Ecology 79:491–500.
Karp, D. S., R. Chaplin-Kramer, T. D. Meehan, E. A. Martin, F. DeClerck, H. Grab, C. Gratton, L. Hunt, A. E. Larsen, and A. Martínez-Salinas. 2018. Crop pests and predators exhibit inconsistent responses to surrounding landscape composition. Proceedings of the National Academy of Sciences 115:E7863–E7870.
Kennedy, C. M., E. Lonsdorf, M. C. Neel, N. M. Williams, T. H. Ricketts, R. Winfree, R. Bommarco, C. Brittain, A. L. Burley, D. Cariveau, L. G. Carvalheiro, N. P. Chacoff, S. A. Cunningham, B. N. Danforth, J.-H. Dudenhöffer, E. Elle, H. R. Gaines, L. A. Garibaldi, C. Gratton, A. Holzschuh, R. Isaacs, S. K. Javorek, S. Jha, A. M. Klein, K. Krewenka, Y. Mandelik, M. M. Mayfield, L. Morandin, L. A. Neame, M. Otieno, M. Park, S. G. Potts, M. Rundlöf, A. Saez, I. Steffan-Dewenter, H. Taki, B. F. Viana, C. Westphal, J. K. Wilson, S. S. Greenleaf, and C. Kremen. 2013. A global quantitative synthesis of local and landscape effects on wild bee pollinators in agroecosystems. Ecology Letters 16:584–599.
Kuddus, M. A., E. Tynan, and E. McBryde. 2020. Urbanization: a problem for the rich and the poor? Public Health Reviews 41:1.
Lemessa, D., P. A. Hambäck, and K. Hylander. 2015. The effect of local and landscape level land-use composition on predatory arthropods in a tropical agricultural landscape. Landscape Ecology 30:167–180.
Letourneau, D. K., J. A. Jedlicka, S. G. Bothwell, and C. R. Moreno. 2009. Effects of natural enemy biodiversity on the suppression of arthropod herbivores in terrestrial ecosystems. Annual Review of Ecology, Evolution and Systematics 40:573.
Losey, J. E., and M. Vaughan. 2006. The Economic Value of Ecological Services Provided by Insects. BioScience 56:311–323.
Martin, E. A., B. Seo, C.-R. Park, B. Reineking, and I. Steffan-Dewenter. 2016. Scale-dependent effects of landscape composition and configuration on natural enemy diversity, crop herbivory, and yields. Ecological Applications 26:448–462.
Ong, T. W., K. Li, A. Lucatero, D. Pak, L. Hawkes, M. Hunter, and J. Vandermeer. 2020. Taylor Made Landscapes: Using Taylor’s Law to Scale Between Metapopulations and Source-Sinks in Urban Garden Space. Frontiers in Sustainable Food Systems 4.
Outhwaite, C. L., P. McCann, and T. Newbold. 2022. Agriculture and climate change are reshaping insect biodiversity worldwide. Nature 605:97–102.
Pedigo, L. P., M. E. Rice, and R. K. Krell. 2021. Entomology and Pest Management: Seventh Edition. Waveland Press.
van der Sluijs, J. P. 2020. Insect decline, an emerging global environmental risk. Current Opinion in Environmental Sustainability 46:39–42.
Vanbergen, A. J., and the I. P. Initiative. 2013. Threats to an ecosystem service: pressures on pollinators. Frontiers in Ecology and the Environment 11:251–259.
Vandermeer, J., and I. Perfecto. 2007. The Agricultural Matrix and a Future Paradigm for Conservation. Conservation Biology 21:274–277.
Wagner, D. L. 2020. Insect Declines in the Anthropocene. Annual Review of Entomology 65:457–480.
Weisser, W. W., and E. Siemann. 2008. The Various Effects of Insects on Ecosystem Functioning. Pages 3–24 in W. W. Weisser and E. Siemann, editors. Insects and Ecosystem Function. Springer, Berlin, Heidelberg.
Research
Study site
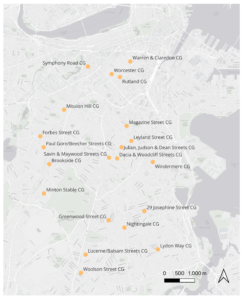
The study is conducted in urban community gardens in Boston managed by the Trustees of Reservation. Boston is a cultural and financial center in the Northeast, playing key roles historically and continuing to evolve as a modern metropolitan. The metropolitan area has varying levels of urban density. The Inner Core consists of high-density commercial districts with little vacant developable land, and a high density of multifamily housing in dense, walkable areas (Boston Region MPO, 2019). Some regional urban centers outside of the Inner Core feature multi-story buildings at each center, and moderately dense to lower-density, single-family residential development in the neighborhoods (Boston Region MPO, 2019). Boston is located on the coast of the North Atlantic, with a climate described either as a humid subtropical climate or a humid continental climate (Beck et al., 2018). Hot and humid summers are typical in these climates (Beck et al., 2018), supporting a productive growing season for various crops and flowers and the productivity of community gardens in the heart of the urban landscape.
The Trustees of Reservations is a non-profit organization managing 56 urban community gardens in Boston, across eight neighborhoods (The Trustees, 2023). The gardens are located in neighborhoods with varying landscapes from different urban densities and unique population demographics (Boston Planning and Development Agency, 2021). Gardeners are free to decide the crops or ornamental plants in the garden, resulting in a high variation in the plant community. All gardeners are asked to follow garden rules set by the Trustees (The Trustees, 2023) with possible additional agreements within each garden. In the community gardens, pesticide and chemical fertilizer use is prohibited, while natural fertilizer and pest controls are allowed.
Sample sizes and design
Twenty-two gardens in eight neighborhoods were selected as candidates for this project based on a pilot study of plant diversity in the Boston community gardens and the interests of The Trustees, including gardens with observed different management practices, such as crop selection and diversity, ornamental plant percentage, and levels of weeds. In each garden, five plots were randomly selected for arthropod sampling of flower-visiting insects and crop pests, prioritizing plots with vegetation data from prior years. In total, we aimed to sample 100 individual gardeners’ plots for arthropods. The sample size and design were adjusted in the field for meaningful collection and by the availability of appropriate sites. We initially proposed to sample pests and pollinators concurrently but prioritized pest sampling in fall 2024 harvest season to ensure high data quality in the limited time frame and ecological relevance based on the phenology of the different groups of arthropods. Flower-visiting insect collection were conducted in spring and summer 2025, following parallel designs. Additional information on the method change can be found in the “Explanation and justification of methods change” section.
Plant sampling
Plant communities were censused at the plot level to construct measures of plant diversity. At each plot, we sampled all plants present, including crops, herbs, ornamental plants, and weeds. We determined the unique species of plants and estimated the abundance of each species and its ground cover. We recorded the varieties of common crops when known to capture finer-scale plant diversity. We further estimated the total ground cover and weed cover at each plot.
At the plot scale, final metrics for quantifying plant diversity included plant richness at different taxonomic levels (species, genus, family, and order), total plant abundance, and Shannon’s diversity index. At the garden scale, we aggregated the data from the five sample plots to obtain garden level plant richness, and calculated mean abundance per plot as a representation of garden level plant abundance. Shannon’s diversity index of plants was calculated using the aggregated data from the five plots. Plots in the same garden share garden level diversity metrics.
Plant sampling was conducted at the same time of each arthropod sampling—repeated sampling was conducted for different rounds of arthropod sampling to ensure robust representation of the plant communities for the arthropods. A total of three rounds of plant samples were conducted in the designated sampling plots accompanying the arthropod sampling.
Site characteristics
Site characteristics describing the land use types and configurations were designed to be collected at the plot level, the garden level, and the neighborhood level. At the individual gardener’s plot level, we collected weed cover in percentage of the plot area, ground cover in percentage of the plot area. We obtained a bird’s eye view picture of each plot. At the garden and neighborhood level, site characteristics will be constructed with a combination of on-site observation and remote sensing satellite images. At the garden level, we will use previous records for the number of trees in the garden and the presence of communal pollinator plots or pollinator strips in the garden for pollinator habitats. We will obtain existing land use land cover GIS layers from Massachusetts and Boston government data sites, which are derived from a combination of USDA National Agricultural Imagery Program and WorldView satellite images (MassGIS Data, n.d.). With the land use data, we will obtain garden sizes and green cover and impervious surface cover at the neighborhood level. At the neighborhood level, a 250 m buffer will be established from the center of the garden to capture the neighborhood landscape for each garden without overlap.
Arthropod sampling methods
Crop pest collection
We initially proposed to randomly select leaves in any plants to inspect for potential crop pests for three minutes in each plot and collect any observed pests. To standardize the arthropod collection in the field by different field researchers and minimize variability introduced by random plant sampling, we adapted the methods to observe and collect arthropods from two focal crop types, tomato plants (Solanum lycopersicum) and pepper plants (Capsicum annuum and Capsicum chinense), which are common and important crops in the community gardens.
Focusing on the effects of plant diversity and site characteristics on pest communities in the focal crop types, we selected five individual gardeners’ plots per garden, prioritizing plots containing at least three plants of each focal crop. When fewer than five plots met the criteria, we selected from the remaining plots with the highest number of focal plants. In total, we sampled pests in 109 plots across the 22 gardens across eight neighborhoods (Figure 1) and on a total of 242 tomato plants and 225 pepper plants.
At each plot, we selected three tomato plants and three pepper plants, prioritizing non-adjacent individuals. Field researchers then observed and collected all arthropods from individual focal plants within a three-minute time frame, which was sufficient for examining the entire plant. Three field researchers concurrently manually collected all arthropods from the three selected plants of each focal crop type, using forceps, insect aspirators, nets, and by hand. The samples from each plant were preserved in individually labeled centrifuge tubes containing 70% ethanol for further processing. Larger arthropods were preserved in additional labeled vials. The arthropods collected from the plots were processed in the lab. All arthropods were classified by order, categorized into functional groups to separate herbivores and arthropods in other function groups (e.g., predators, parasitoid etc.). Each of the individual was assigned to a morphospecies.
We selected tomato and pepper plants as focal crops due to their popularity and value to the gardeners, and we additionally included herbivory levels and overall plant damage on the plants as a response variable to better inform management implications of the pest community results. We estimated the percent herbivory damage on the plants by examining all leaves on each plant and recording the percent area lost to herbivory (e.g. Figure 2). To reduce observational bias and assess the extent of damage to the plants, the herbivory estimates were categorized into predefined damage classes to standardize observations (Table 1).

Table 1. Crop damage in percentage and the corresponding damage classes.
|
Percent damage (%) |
Damage class |
|
< 5 |
1 |
|
6 – 15 |
2 |
|
16 – 50 |
3 |
|
> 50 |
4 |
We examined the effects of plant diversity and site characteristics on all collected arthropods in the sampling to assess any overall effect on arthropods collected through the manual collection method, and then examined the effects on pests (plant herbivores) specifically. Due to the high number of variables, I ran a correlation matrix to assess the collinearity of plant diversity and site characteristics to determine biologically relevant non-correlated variables to include in the next steps. To understand the joint effects of ecological processes and management on the arthropod groups, I ran generalized linear mixed models on both overall arthropod and herbivore richness and abundance, using variables representing plant richness, abundance, habitat size, management intensity, and additional appropriate variables. By using a model selection process, a final model with the lowest AIC was selected, and the effects of the included parameters were presented to show significant drivers for arthropods collected using the manual collection method.
Flower-visiting insects (pollinators)
Flower-visiting insects sampling methods were adapted from existing methods, through visual observation and quantification of insect visitation in the plot and on specific floral resources (O’Connor et al., 2019). I conducted two rounds of flower-visitor collection in the growing season in June and August using two separate methods. In the first round, in June, at each plot, I stood in front of the plot and observed a 1m*1m area for flower-visiting insects for five minutes (Figure 3), and assigned each insect to the following taxonomic groups: bumblebees, honeybees, solitary bees (including primitively eusocial species), hoverflies and other flies, and others. The total number of the morphospecies was recorded, along with the abundance of flower-visitors. We initially proposed to take a picture of each observed morphospecies, but due to the fast-flying nature of the organisms, we instead took notes and only took pictures when possible.
In the second round of the collection in August, we initially proposed to use a pepper plant to observe the flower-visitors that visited the flowers. However, after testing, we found that pepper flowers were relatively inconspicuous and did not attract sufficient visitors in the 5-min proposed observation time for comparison across sites. To address this, we replaced the focal plants with ornamental plants matching common colors used to attract flower-visitors: blue lobelia (Lobelia siphilitica), black-eyed susan (Rudbeckia hirta), and hairy mountain mint (Pycnanthemum vertacillatum var.pilosum) (Figure 4).


We initially explored the possibility of employing both pollinator sampling methods concurrently. However, due to resource constraints, we opted for a sequential sampling approach. The modification ensured that each method was applied consistently under comparable weather conditions and growing stages, thereby mitigating variability in flower-visitor communities caused by distinct environmental factors. Additionally, the sequential sampling allowed for an increased temporal coverage for flower-visitors. With the two flower-visitor methods, I sampled plant community for each round and will analyze the impact of plant diversity on flower visitors using these data separately and compare the effects.
Parallel to methods on the herbivore focused collection, I will run correlation matrix and select ecological relevant variables corresponding to the set of variables assessed for the other functional groups, with the addition of floral resource diversity and abundance.
Arthropod collection and processing materials
Camera, Ziploc bags, glass vials, 70% ethanol, insect aspirator, paper bags, centrifuge tubes, forceps, microscopes, arthropod guides, notebooks, and pencils.
Explanation and justification of method change
Due to the time constraints in the past field season and our aim for better data quality in the field collection, after careful consideration, we made adjustments to our methods while maintaining the integrity of our questions and objectives. We decided to separate the examination of the pest and pollinator communities before integrating the findings, and we adjusted the field methods for crop pest collection. This allows us to better control confounding variables and enhance data quality and ecological relevance.
During the summer of 2024 sampling, we focused on the effects of plant diversity and site characteristics on pest communities and adjusted the arthropod collection method to improve standardization and reduce confounding variables and sampling error. Initially, we proposed to randomly select leaves in any plants as researchers walk through the plot to inspect for crop pests for three minutes in each plot and collect any observed pests. Rather than a random inspection, we adjusted the methods to observe and collect arthropods from two focal crop types, standardizing the arthropod collection and minimizing variability introduced by random plant sampling. The plant community varies among plots, and a random selection of leaves from different plants introduces a significant confounding variable for the arthropod community, namely plant species identity. One group of pests, the specialist herbivores, often co-evolve with the plant species on which they feed, leading to a close association of these herbivores and specific plant identities (Leimu et al. 2012). When examining arthropod community composition, the strong relationship between specialist herbivores and specific plant identities can manifest as differences in arthropod community that are associated with species identity rather than the factors of primary interest, including surrounding plant community diversity (Dassou and Tixier 2016). By targeting a few focal plants, we eliminate the effect of variation in plant identity, thereby allowing for a more confident examination of how plant diversity and site characteristics affect arthropod communities.
Using focal plants also enabled us to enhance the data quality and data quantity of arthropod samples. Instead of randomly selecting leaves from different plants, we observed and collected all arthropods from individual focal plants within a three-minute time frame. The time frame was validated in the field as adequate for whole plant examination, leading to thorough sampling and reduced variation that would be introduced by different individuals using a random leaf sampling method. We managed to concurrently collect arthropods from three plants with three people in the field, resulting in a larger sample size of arthropods for each plot. Based on field validation, manual sampling on individual plants is effective for pest collection. In addition, we collected all arthropods observed on the focal plants during the time frame in the field and did not exclude non-pest arthropods, offering a comprehensive representation of the arthropod community that may utilize or land on the focal plants in the plot, providing supporting data to address our first two objectives.
We selected tomato plants (Solanum lycopersicum) and pepper plants (Capsicum annuum and Capsicum chinense) as focal plants. Both belong to the family Solanaceae. Tomato and pepper plants are the most common crops by the number of gardeners that have them in their plots in the Boston community gardens, based on previous crop surveys. They are not only prevalent but are also found in plots with varying crop diversity and management, making them appropriate choices for the study. Using focal plants tomatoes and peppers enables us to maintain control over plant identity where the arthropods were collected while capturing high variation in plant diversity and site characteristics at both the plot and garden levels.
Given the prevalence of the focal crops (tomatoes and peppers), and the high value of their fruits to gardeners, we also decided to include herbivory as an additional response variable. If plant diversity and site characteristics impact pest richness and abundance, it is crucial to determine whether these changes lead to differences in crop damage and yield. This is particularly relevant because we do not know if pest richness and abundance correlate linearly with herbivory in urban gardens due to complex herbivore-natural predator interactions (Dassou and Tixier 2016). By examining herbivory, we can provide actionable recommendations for optimizing plant diversity while balancing pest suppression, and shed light on the effects of the pertinent factors at specific spatial scales.
Pest data were collected at the plot level, while plant diversity and site characteristics were collected at both plot and garden levels, to investigate the effects of spatial scales of plant diversity and site characteristics on arthropod diversity. Plot- and garden-level plant diversities (richness, abundance, and ground cover) were collected in the field, while additional site characteristics at the garden level (e.g., ground cover and neighborhood greenness covers) are being quantified using remote sensing and existing land-use data as originally proposed. This allows us to construct statistical analyses to examine the local (plot-level) and landscape (garden-level) effects of plant diversity and site characteristics on the arthropod community in each plot, offering insights into management at the gardener, garden manager, and city scale.
The second part of the field study now examines the effect of plant diversity and site characteristics on pollinator diversity. The field study will take place from late May to June 2025, focusing on pollinators during peak pollination season. Due to time constraints during the harvest season in the past summer, we prioritized pest sampling because of its ecological relevance in the season. For the pollinator sampling, we will observe and collect data on pollinator diversity at the plot level as proposed, selecting plots and gardens matching previous pest collection. Originally, a simultaneous sample of pests and pollinators was proposed, which would have allowed for direct comparisons of the impacts of plant and site factors on these two groups. Although we would not be able to directly compare the impacts with the separation of sampling, we will still be able to integrate the data to assess the effects. Pests and pollinators are functionally distinct arthropod groups, each with unique ecological roles, phenological differences in active periods, and different mechanisms by which plants may drive their community composition. For instance, pests are primarily influenced by host plant availability, while pollinator activity is often driven by floral resources, plant structural complexity, and seasonality (Ratnadass et al. 2012, Bennett and Lovell 2019, Pardee et al. 2023). By separating their sampling periods, we can gather more targeted and ecologically relevant data, ensuring data quality while more effectively addressing each group’s unique responses to plant diversity and site characteristics.
After completing pollinator sampling, we will integrate the responses of pollinators and pests to gain a comprehensive understanding of the effects of plant diversity and site characteristics on the arthropod community. Since pests and pollinators do not directly interact, the effects of plant and site factors are likely to be additive. For example, one possible outcome could be that increased plant diversity at the garden level leads to higher pollinator diversity while having no effect on pest diversity or levels of herbivory. This will lead to the recommendation for increasing garden-level plant diversity. We will analyze both datasets within the same spatial framework to ensure a holistic understanding of how arthropods respond to plant diversity and site characteristics.
Although separating pest and pollinator sampling and focusing on specific plants required method adjustments, these refinements reduce confounding variables, improve data quality, and enhance ecological relevance while still aligning with the original research objectives. The trade-offs have been carefully considered, ensuring that the study remains robust and relevant in terms of its scientific and management implications.
Reference
Beck, H. E., Zimmermann, N. E., McVicar, T. R., Vergopolan, N., Berg, A., & Wood, E. F. (2018). Present and future Köppen-Geiger climate classification maps at 1-km resolution. Scientific Data, 5, 180214. https://doi.org/10.1038/sdata.2018.214
Bennett, A. B., & Lovell, S. (2019). Landscape and local site variables differentially influence pollinators and pollination services in urban agricultural sites. PLOS ONE, 14(2), e0212034. https://doi.org/10.1371/journal.pone.0212034
Boston Planning and Development Agency. (2021). Neighborhood Demographics—Boston Neighborhood Demographics, 2015-2019 American Community Survey—Analyze Boston. https://data.boston.gov/dataset/neighborhood-demographics/resource/d8c23c6a-b868-4ba4-8a3b-b9615a21be07
Boston Region MPO. (2019). Chapter 2—Land Use in the Boston Region MPO Area. In Needs assessment of the long-range transportation plan of the Boston region metropolitan planning organization. https://www.ctps.org/data/html/plans/LRTP/charting/Charting_Progress_2040_Chapter2_final.html
Dassou, A. G., and P. Tixier. 2016. Response of pest control by generalist predators to local-scale plant diversity: a meta-analysis. Ecology and Evolution 6:1143–1153.
Leimu, R., A. Muola, L. Laukkanen, A. Kalske, N. Prill, and P. Mutikainen. 2012. Plant-herbivore coevolution in a changing world. Entomologia Experimentalis et Applicata 144:3–13.
MassGIS Data: 2016 Land Cover/Land Use | Mass.gov. (n.d.). Retrieved November 2, 2023, from https://www.mass.gov/info-details/massgis-data-2016-land-coverland-use
O’Connor, R. S., Kunin, W. E., Garratt, M. P. D., Potts, S. G., Roy, H. E., Andrews, C., Jones, C. M., Peyton, J. M., Savage, J., Harvey, M. C., Morris, R. K. A., Roberts, S. P. M., Wright, I., Vanbergen, A. J., & Carvell, C. (2019). Monitoring insect pollinators and flower visitation: The effectiveness and feasibility of different survey methods. Methods in Ecology and Evolution, 10(12), 2129–2140. https://doi.org/10.1111/2041-210X.13292
Pardee, G. L., K. M. Ballare, J. L. Neff, L. Q. Do, D. Ojeda, E. J. Bienenstock, B. J. Brosi, T. H. Grubesic, J. A. Miller, D. Tong, and S. Jha. 2023. Local and Landscape Factors Influence Plant-Pollinator Networks and Bee Foraging Behavior across an Urban Corridor. Land 12:362.
Ratnadass, A., P. Fernandes, J. Avelino, and R. Habib. 2012. Plant species diversity for sustainable management of crop pests and diseases in agroecosystems: a review. Agronomy for Sustainable Development 32:273–303.
The Trustees. (2023). Boston Community Gardens. The Trustees of Reservations. https://thetrustees.org/place/boston-community-gardens/
Herbivore targeted sampling (2024)
In our summer 2024 sample, through manual collection of arthropods from tomato and pepper plants, we collected a total of 2469 individual arthropods, belonging to 13 arthropod orders, identified to 268 morphospecies. These arthropods were from six functional groups, with herbivores being the most abundant, consisting of 74.5% of all arthropods (Figure 5), followed by omnivores, decomposers, predators, parasitoids, and a flower visitor. Mean herbivore abundance per plot was 17 individuals.
Plant community varied among plots and gardens, showing a median in richness of 18 species per plot, ranging from 5 to 43 species. This variability reflects different crop selection preferences and management styles of individual gardeners.
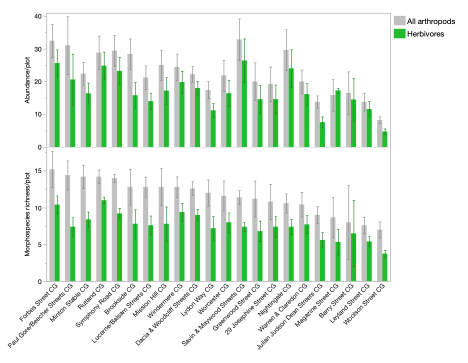
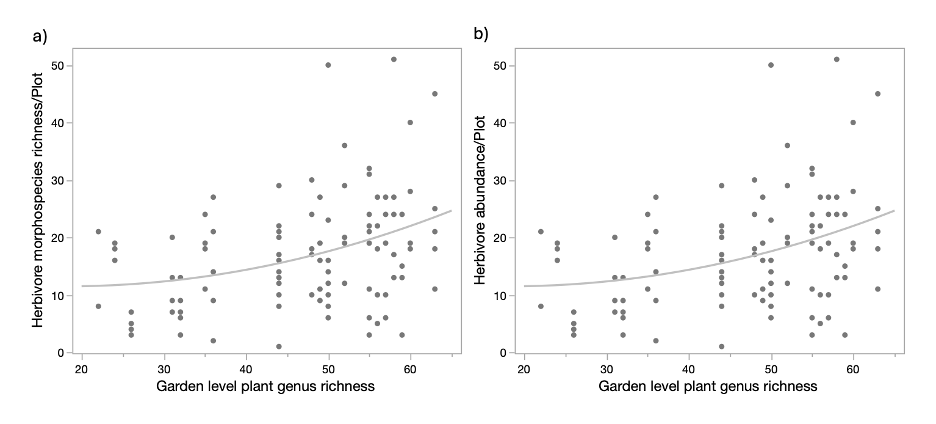
Among all variables of interest, including plant richness at different taxonomic levels at different spatial scales, plant counts and ground cover, weed cover, plant diversity, we used correlation and model comparison and selected plant genus richness at both the local and garden level, ground cover, and weed cover as parameters for the model to understand overall arthropod response (all functional groups included). Garden level plant genus richness had a significant positive effect on both arthropod morphospecies richness (Effect size = 0.0116, F= 9.477, P = 0.003) and abundance (Effect size = 0.0196, F = 17.3, P = 0.0002) in the plots, while no other effects were detected.
Herbivore morphospecies richness and abundance were also both significantly positively affected by garden level plant genus richness (Richness: effect size = 0.0126, F = 8.35, P = 0.006; Abundance: effect size = 0.0192, F = 9.92, P = 0.003). Similar to the overall arthropod community responses, local plant richness, overall abundance, and abundance of focal crops (tomato and pepper plants) did not have a significant effect on the herbivores.
Using ordinal logistic regression, I assessed how herbivore richness and abundance, along with plant richness and abundance at different spatial scales, affect herbivory observed in the focal plant tomatoes and peppers. For tomato plants, there were no significant effects from any of the factors examined. On the contrary, pepper herbivory was significantly affected by plant variables, but not by pepper herbivore richness or herbivore abundance. Total ground cover at the plot level, garden level plant genus richness, and number of pepper varieties all had a significant positive relationship with pepper herbivory, while the ratio of weed cover to ground cover had a significant negative relationship with pepper herbivory.
The results suggest that plant richness has an effect on arthropod richness, where higher plant richness at the garden level is associated with higher richness and abundance of both herbivores and overall arthropods. However, its further relationship with herbivory was not consistent between the two focal crops. Herbivore richness and abundance did not predict herbivory on either plant species, while plant communities had a significant effect on herbivory in pepper plants. This could be due to the different defense mechanisms between plants, where the trichomes in tomatoes may prevent chewing insect herbivory more effectively than the smooth leaves of pepper crops.
Flower-visitor targeted sampling
In the spring/summer 2025 sample, we conducted two rounds of sampling for flower visitors, and data analyses are in progress. Through the first round of sampling in a set observation area (1m*1m) in each plot, we observed a total of 726 individual flower visitors in 9 categories, including honey bees, bumble bees, solitary bees, wasps, hoverflies, other flies, moths and butterflies, ants, and beetles. The median of flower visitor richness in each plot was 4, with a range from 0 to 10, while the mean abundance of flower-visitors observed was 6.67 in each plot, ranging from 0 to 23.
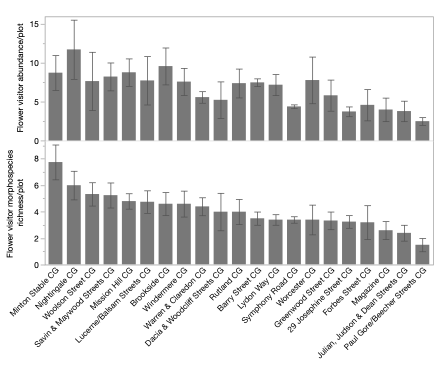
In the Initial model, I examined the effect of local plot level plant richness, abundance, ground cover, weed cover, and floral abundance on flower visitor richness and abundance. Flower visitor morphospecies richness did not show responses to any of the predictor variables. However, floral abundance in the plot had a significant positive effect on flower visitor abundance (effect size = 0.0015, F = 9.7, p = 0.003, Figure 8). In addition, plant richness at the local scale also had a marginally significant effect on flower visitor abundance (effect size = 0.02, F = 3.8, p = 0.055, Figure 8).
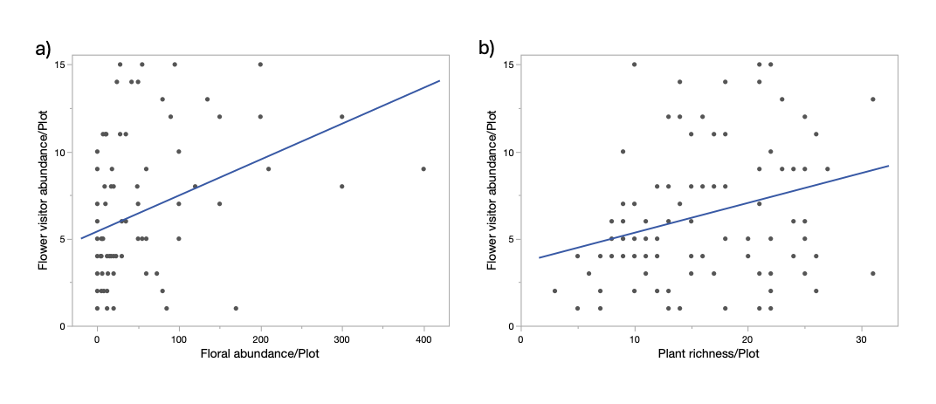
With our initial analysis, we found that the richness of flower visitors in the urban community gardens did not respond to local level plant factors, suggesting a potential filtering effect at a larger spatial scale, where certain flower visitors were more likely to be present in these gardens. Meanwhile, flower visitor abundance was significantly affected by floral abundance, agreeing with previous studies, and was marginally significantly predicted by plant richness. Further analysis on the landscape scale effects would be important to understand additional drivers of flower visitor richness and abundance.
From our analyses thus far, we are observing a positive relationship between plant richness at different spatial scales and arthropod richness in different functional groups, suggesting a positive biodiversity association across trophic levels. Plant biodiversity increases arthropod richness or abundance regardless of the functional group, but seems to support pollination services by increasing the abundance of flower visitors. Though an increase in pest abundance and richness was also observed with increasing plant richness, we did not observe a consistent direct effect on plant damage as measured by herbivory. This suggests that there may be a limit to herbivory or a balancing effect that impacts herbivory in these common crops, such as management, or support for natural predators that control the plant damage.
Education & outreach activities and participation summary
Participation summary:
We collaborate closely with The Trustees to enhance the understanding of arthropod communities and landscape characteristics in their managed gardens. Sharing the observations and findings periodically with The Trustees is crucial to inform future management strategies effectively. Specifically, we have employed both formal and informal channels of communication, informally through direct communications in writing and in meetings biannually or when significant findings arise, and formally through reports of the results. In fall 2024, we shared preliminary results from the summer sampling and a comprehensive summary of past findings from our lab. These were presented to The Trustees Boston Community Garden Team through a Zoom meeting and a visual report. The report focuses on key topics, including plant community composition, gardeners’ planting preferences, and previous findings on arthropod communities. We also created a simple infographic on plants and arthropods in the gardens and are editing the infographic for distribution through social media. In summer 2025, we shared ongoing analysis results through informal in-person meetings during the field season, and continued communication through email. Based on the study outcomes, we will work closely with The Trustees to identify potential areas of improvement to promote sustainable insect management practices, such as incorporating flower strips or promoting or reducing plant growth in peripheral lands in the gardens directly managed by The Trustees’ staff. Through The Trustees, we hope to also reach city planners to identify context-specific measures that can create a supportive landscape for both gardeners and residents, such as the improvement of urban green space and tree planting in city planning. Through the process, we hope to strengthen collaboration, build trust, and enhance our collective understanding of urban community gardens and their roles in the city landscapes.
We aim to engage gardeners within the community gardens that we sample. We aim to raise awareness among gardeners about the insects and other arthropods in their plots and the potential impact of their management choices. For the past two years, we actively engaged with gardeners during the sampling period and through in-person presentations on arthropods in the gardens at the garden coordinators’ meeting. In October 2024, we attended the gardener coordinators’ meeting with over 25 garden coordinators and 6 Trustee's team members, and presented preliminary results from this year’s sampling and past findings on plant and arthropod communities in the gardens. Gardeners expressed interest in our research findings and shared personal anecdotes about encounters with common insects highlighted in the presentation. During both field seasons, we proactively communicated sampling plans with The Trustees and created short project introductions that were shared with the garden coordinators. During fieldwork, we engaged in informal conversations with the gardeners, learning about their experiences with gardening and arthropods while sharing information about our research and ways to engage in citizen science research. These exchanges helped connect our research to gardeners’ experiences and inspired more questions and discussions.
We will present arthropod groups including herbivores and flower visitors identified during the study as well as other garden characteristics in spring 2026 to gardeners. Taking into consideration the multi-lingual nature of the gardeners, we will translate the infographics into other commonly used languages, including Chinese, Spanish, and Portuguese. These infographics will be distributed in print to post in gardens, and digitally through gardener newsletters, fostering a greater understanding of the garden ecosystem and sparking curiosity among gardeners. We will continue to actively participate in gardeners’ meetings to present the preliminary findings of the work and will suggest strategies for sustainable management of insects and other arthropods within the gardens if appropriate, including the annual gardeners’ gathering. Through ongoing collaboration and community engagement, we hope to continue connecting our work with gardeners and garden managers.
We extended the impact of the research by reaching a broader audience including urban gardeners and the academic community. In collaboration with the Trustees, we will leverage online social media platforms (Instagram) to share the findings of our work with the general public, with an emphasis on urban gardeners, in the hope of raising awareness about the often-overlooked or misunderstood arthropod communities within urban landscapes. By stressing the impact of personally managed land in urban areas compared to the surrounding city landscapes, we aim to inspire a greater appreciation for the unique biodiversity in urban gardens and the importance of city landscape management. We started adding observations during the field sample and encouraged the use of iNaturalist to share observations of pollinators and other arthropods to encourage gardeners in Boston and beyond to contribute to everyone’s understanding of the garden arthropod community. To reach the academic community, we presented the study at the Ecological Society of America annual conference to share the results and invite feedback. We plan to publish our findings in a scientific journal to engage other academic researchers in the field. We hope to draw attention to Boston’s community gardens and urban community gardens as valuable subjects for further studies. Ultimately, we aspire to motivate more research on the topic, which will not only support urban gardeners but also inform city planning for the establishment of new urban community gardens.
Project Outcomes
This study examines plant-arthropod diversity and interactions within the urban landscape using Boston as a case study. The project’s findings will establish a foundational knowledge of the frequently overlooked and underappreciated diverse plant and arthropod communities in urban landscapes for Boston and cities beyond in the Northeast. Furthermore, the outcome of the project will provide opportunities for developing management recommendations that can better support urban agriculture while maintaining its critical ecological roles in contributing to biodiversity in urban landscapes.
By deepening our understanding of how plant community composition affects pest communities, we aim to provide insights that support more diverse and resilient plant and arthropod communities that also reduce management efforts. In Boston community gardens, a variety of natural pest control methods are employed, while pesticide use is prohibited. Our research provides input into cultural management of pests and support for beneficial arthropods. From our study, we found that high plant diversity supports a diverse arthropod community without necessarily increasing herbivory levels. This allows us to further promote the planting of diverse crops, contributing to positive ecosystem services and biodiversity conservation in the urban landscape.
The flower-visiting insect study aims to provide management recommendations while raising awareness of beneficial arthropods in the garden landscape. From the study, we will identify site characteristics and practices that promote pollinator abundance and diversity, which enhance ecosystem services for gardeners, including increased plant pollination and yield. Integrating with results from the pest work, we will provide specific recommendations that maximize pollinators and minimize pests at the plot scale. We also hope to engage gardeners by highlighting pollinators as beneficial arthropods and fostering curiosity about diverse arthropods in urban gardens through presentations, workshops, and citizen science projects.
Through this study, we built upon our understanding of cultivated crops in community gardens and gained new insights into the significant role of ornamental plants and weeds in contributing to plant richness and diversity. Through the analysis, we are seeing the important effect of plant diversity on arthropods, while gaining a deeper appreciation of the complexity of the plant-arthropod interactions in the urban landscape.
Through informal conversations with gardeners, we also became aware of their concerns regarding different crop diseases, which could be unfamiliar to gardeners and often lack clear and accessible treatment options. These discussions presented the potential for future research and collaboration with gardeners and crop scientists, to develop practical and applicable research that benefits the gardening community in urban community gardens.
For our project, we separated pest and pollinator sampling to obtain more ecologically and socially relevant results based on the phenology of organisms and planting seasonality. This gives us more directly actionable results for the management of respective arthropod groups in the gardens while allowing for integration by using parallel methods. However, a further study using a simultaneous collection could provide more direct information on the responses of different arthropod groups to plant diversity and site characteristics, which can complement the results from this study.