Progress report for GNE24-324
Project Information
Poultry growers face a significant challenge posed by the resurgence of Infectious Coryza (IC), an infectious respiratory disease of chickens. The recent increase in the prevalence and severity of IC could be due to changes in pathogen genotype, virulence, and/or failure of the current vaccines. Inactivated vaccines against Avibacterium paragallinarum (AP) do not provide long-lasting and comprehensive protection from IC. Therefore, there is an urgent need to develop a safe, more effective, and cross-protective vaccine against the emerging AP strains. We have identified two AP strains, isolated from asymptomatic commercial flocks in Ohio, with defects in crucial virulence genes. These strains were non-pathogenic in specific-pathogen-free (SPF) chickens during a challenge trial. Based on these preliminary findings, we propose to evaluate the safety and efficacy of these strains as live vaccine candidates in commercial layers. The safety of the live vaccine candidates will be measured based on the clinical signs score and live body weight in challenged chickens. If deemed safe, the efficacy will be measured based on the vaccines’ ability to protect chickens against challenge from virulent field strain of AP. The proposed live vaccines could be effective alternatives to the currently available inactivated AP vaccines. Furthermore, the proposed vaccines, if successful will be more cost-effective in terms of production and administration, compared to current vaccines. The vaccines will reduce the economic burden faced by poultry growers due to persistent and increasing outbreaks of IC in commercial poultry flocks.
Objectives (400-word limit).
The project aims to mitigate the economic burden faced by poultry growers in the Northeastern United States, due to IC affecting poultry flocks, especially layer and breeder chickens. The goal of this proposed study is to provide poultry growers and the US poultry industry with a safe, effective, low-cost, and easy-to-administer, live attenuated vaccine against Avibacterium paragallinarum (AP). The proposed vaccine candidate can reduce the impact of IC in susceptible chickens. The specific objectives of the proposed NE-SARE project are as follows:
1. To determine the safety of the two live vaccine candidates in the commercial layers.
The safety of the two avirulent strains named AP36_OH_21 and AP37_OH_21 will be determined based on the clinical signs scores, and live body weight of the chickens after challenge inoculation with the vaccine candidates. We have already established that the vaccine candidates are not pathogenic in the SPF chickens. Based on the preliminary findings from the pathogenicity trial in SPF chickens and the observation of multiple defective virulence genes from the in-silico study, we hypothesize that the two vaccine candidates will be safe and produce no adverse effects in commercial layers as well.
2. To evaluate the efficacy of the live-vaccine candidates against the challenge from virulent field strain of Avibacterium paragallinarum in commercial layers.
The efficacy of the vaccine candidates AP36_OH_21 and AP37_OH_21 will be assessed based on the reduction in the clinical sign scores and the amount of bacterial shedding in the vaccinated groups compared to the unvaccinated control groups. We hypothesize that the two live vaccine candidates will elicit strong immunity which will reduce the bacterial colonization and severe clinical signs caused by virulent field strains of the AP in the commercial layers.
The purpose of this project is to develop a safe, effective, long-lasting, economical and easy-to-administer live-vaccine against Infectious Coryza infection in chickens. By advancing current knowledge in vaccine development and delivery, this project will contribute to sustainable agriculture by offering a more effective and sustainable solution for controlling Infectious Coryza. The proposed vaccines can also reduce the reliance on antimicrobials, mitigate economic losses, improve poultry welfare, and improve the sustainability of poultry production in northeastern USA and beyond. The development of effective vaccines as non-antibiotic control measures against Infectious Coryza will reduce the risk of the development and spread of antimicrobial resistance which has great implications on public health.
IC caused by Avibacterium paragallinarum (AP) imposes a significant burden on the health and productivity of laying and breeding chicken flocks. The infection causes substantial economic losses due to decreased egg production, increased culling in layers, and reduced growth rates in broilers (Blackall, 1999). Recently, the prevalence and severity of IC in chicken flocks has increased in the United States (Clothier et al., 2019; Crispo et al., 2019a). The bacterium long thought to have preferences towards warmer geographical regions, reflected by the persistent outbreaks in the southeastern states and California in the United States (Blackall, 1999; Deshmukh et al., 2015). However, multiple IC outbreaks are being reported from temperate Northeastern states such as Pennsylvania, Delaware, and Maryland, where it is considered a major emerging respiratory disease of poultry (Kuchipudi et al., 2021). The upsurge in prevalence, pathogenicity, and geographical spread of AP indicates potential changes in its virulence, transmission dynamics, and reduced efficacy of the current vaccines. Antibiotics fail to eliminate AP from the infected chickens, leading to chronic shedding in recovered chickens without clinical signs, making vaccination the best preventive option.
The commercially available Infectious Coryza vaccines are based on inactivated or killed whole-cell mixtures of different serovars of Avibacterium paragallinarum. Despite being relatively safe, the killed vaccines require multiple administrations, relatively higher dosages, and adjuvants, some of which may induce adverse effects. Furthermore, they induce limited cellular and mucosal immunity in poultry, both of which are crucial for broad and long-term immunity against AP infection. Administration of these killed vaccines also requires intra-muscular or subcutaneous injection in individual chickens, requiring huge effort and technical expertise. This complexity of vaccine administration has significant economic costs disincentivizing the regular AP vaccination. In addition, backyard flocks which often lack adequate veterinary care, vaccination, and regulatory oversight, carry the risk of being chronic carriers of AP for life. It magnifies the risk of the spread of IC from these carrier birds to susceptible chickens in nearby commercial poultry operations.
While the development of a live vaccine against Infectious Coryza has been one of the central topics among veterinarians, it has not been developed to this date. Nonetheless, natural AP infection was found to induce some amount of cross-protective immune response in the chickens (Rimler & Davis, 1977). Live vaccines, composed of attenuated or avirulent forms of the bacterium, mimic natural infection and induce robust immune responses, leading to higher levels of cellular and humoral immunity. Administration of the poultry live vaccines through the oral route also greatly reduces the complexity and cost of vaccine administration while inducing a higher level of mucosal immunity. We have identified two strains of AP that have been isolated from asymptomatic commercial poultry flocks. These strains did not induce any clinical signs of IC in SPF chickens and their genomes had multiple defective virulence genes including HMTp210, HctA, and FlfA. Based on these experimental and in-silico observations, these two strains of AP were deemed to have attributes of live vaccines and were thus proposed for further study and exploration.
Evaluation of vaccine safety and efficacy in commercial layers will provide the necessary validation that the vaccines will work in the targeted animal population. Commercial chickens differ from SPF chickens, which are commonly used in experimental studies, in terms of the microbiota present in the upper respiratory tract, known to influence post-vaccinal reaction and its severity. The development of a safe and effective live vaccine against Infectious Coryza will mitigate IC outbreaks due to emerging strains, and provide a wider spectrum of cellular, mucosal, and humoral immunity and long-term protection with fewer administrations, allowing easier administration via water or spray. These favorable attributes of the live vaccine will incentivize the vaccination of susceptible poultry flocks and lessen the economic impact of the disease. The findings from the study will help propel the vaccines toward commercialization.
Research
Update Jan 2026
Objective 1: To determine the safety of the two live vaccine candidates in the commercial layers.
Experimental Design
1.1 Growth of Avibacterium paragallinarum and preparation of the inoculum
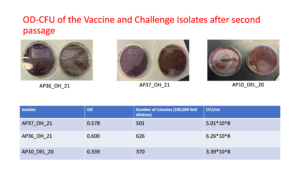
Frozen isolates of Avibacterium paragallinarum were streaked onto chocolate agar plates and incubated at 37 °C for 24–36 h under microaerophilic conditions. Characteristic dewdrop-like colonies were observed, and a second passage was performed by streaking a single colony onto fresh chocolate agar and incubating under the same conditions for 24–36 h. A single colony from the second passage was then inoculated into brain heart infusion (BHI) broth supplemented with 10% fetal bovine serum (FBS) and 0.0025% nicotinamide adenine dinucleotide (NAD) and incubated at 37 °C under microaerophilic conditions with constant shaking. After 16 h of incubation, the optical density of the culture was measured, followed by preparation of 10-fold serial dilutions in sterile BHI supplemented with 10% FBS and 0.0025% NAD. Aliquots (100 µL) of each dilution were spread onto fresh chocolate agar plates and incubated to determine colony-forming units, enabling correlation of optical density with viable counts for the vaccine strains and the positive control strain.
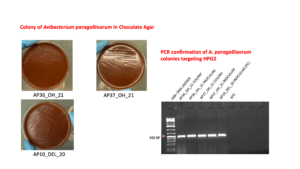
1.2. PCR confirmation of the A. paragallinarum Colony and Inoculum
Pure colonies were confirmed by PCR using DNA extracted by the boiling method. Briefly, a single colony was suspended in 100 µL of DNase-free water, while 10o µL of the bacterial inoculum grown in BHI broth was transferred to a microcentrifuge tubes. Samples were heated at 100 °C for 10 min to lyse bacterial cells, followed by centrifugation at 8,000 × g for 5 min to pellet cellular debris. The supernatant containing genomic DNA was collected and used as the PCR template.
PCR reactions were prepared in a total volume of 25 µL, consisting of 12.5 µL of GoTaq® G2 Hot Start Green Master Mix, 1.25 µL each of HPG2 forward and reverse primers, 5 µL of DNase-free water, and 5 µL of template DNA. PCR was carried out with initial denaturation at 95°C for 2 minutes to activate Hotstart master-mix, followed by 25 cycles of denaturation, annealing and extension (94°C for 20 seconds, 63 °C for 30 seconds, and 72 °C for 40 seconds), and final extension with 72°C for 10 minutes, a protocol adapted from previous study (Chen et al., 1996). PCR amplicons were visualized on a 1% agarose gel with Ethidium bromide.
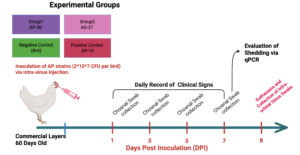
1.3 Animal Experiment
A safety trial was conducted in 60 day old chickens to reassess the pathogenicity of two live vaccine candidate strains and a virulent field strain of Avibacterium paragallinarum. Prior to inoculation, five birds were randomly selected and screened for A. paragallinarum by HPG2 PCR to confirm the absence of infection. Birds were then randomly assigned to four experimental groups, each containing five birds corresponding to two vaccine candidate strains (AP36_OH_21 and AP37_OH_21), one virulent field strain (AP10_DEL_20, and a negative control group. Bacterial inocula were prepared as described previously and standardized to the same concentration, after which 0.2 mL of the inoculum (10^8 CFU/ml) was administered into the right infraorbital sinus of each bird using an insulin syringe. After the challenge, the chickens were monitored daily for the clinical signs of IC, and clinical signs were scored and recorded at 4 different levels: 0 for no signs, 1 for those with mild facial edema and nasal discharge, 2 for those with moderate facial edema and nasal discharge, and 3 for those with abundant nasal discharge, severe facial edema, and lacrimation (Bragg, 2002). Choanal swabs were collected at 2, 3, 5, and 7 days post inoculation to assess bacterial shedding and persistence of the vaccine candidates and the field strain. All animal procedures were performed under approved IACUC protocol R-JUN 25-26.
2. Experimental Design for Objective 2: To evaluate the efficacy of the live-vaccine candidates against the challenge from virulent field strain of Avibacterium paragallinarum in commercial layers.
2.1. Rationale:
Commercial layers are routinely administered with live vaccines against various pathogens and get exposed to many other microbes, some of which may colonize the respiratory tract and influence the severity of Infectious Coryza. Thus, to assess the efficacy of the live vaccine at the field level, it is essential to conduct the efficacy test on the chickens raised in commercial raising conditions.
2.2. Experimental Design and Procedures:
To evaluate the efficacy of the live-vaccine candidates in commercial layers, another trial will be conducted in chickens. Commercial layers (6-weeks old, n=40) will be purchased and divided into four equal groups with 10 birds in each group: Group A, No Vaccine and No Challenge; Group B, No Vaccine and Challenge by Field Strain 1; Group C, Vaccine AP36_OH_21 and Challenge by Field Strain 1; Group D, Vaccine AP37_OH_21 and Challenge by Field Strain 1. The chickens in the vaccination groups will be immunized via intra-nasal instillation of 0.2 mL (1×10^7 CFU/ml) of live-vaccine candidates at 7 weeks of age. The chickens in the non-vaccination control groups will be inoculated with 0.2 ml of BHI into the nasal passage. The vaccinated birds will be challenged by intranasal inoculation of 0.2 ml (1×10^5 CFU/ml) of one virulent field strain related to the current outbreak of AP at week 11. The birds in each experimental group will be monitored for 7 days post-challenge (DPC) and the clinical sign score of each bird will be recorded. Similarly, choanal swabs will be collected from the birds on day-1, day-3, day-5, and day-7 post-challenge and the bacterial load will be calculated. The clinical sign score will be calculated based on the protocol defined above in the safety trial. On day 8 post-challenge, birds will be euthanized humanely using CO2. The efficacy of the vaccines will be evaluated based on their ability to reduce the clinical sign scores and bacterial load in the vaccinated chickens compared to the non-vaccinated ones.
|
Experimental Groups |
Immunization Strain (1 × 107 CFU/ml) 7 WOA |
Challenge Strain (1 × 105 CFU/ml) 11 WOA |
Challenge age in weeks |
Collection of choanal swabs (DPC) |
Clinical sign Scoring (DPC) |
No. of chickens |
|
A |
- |
- |
11 |
1,3,5,7 |
0,1,2,3,4,5,6,7 |
10 |
|
B |
- |
Filed Strain-1 |
11 |
1,3,5,7 |
0,1,2,3,4,5,6,7 |
10 |
|
C |
AP36_OH_21 |
Filed Strain-1 |
11 |
1,3,5,7 |
0,1,2,3,4,5,6,7 |
10 |
|
D |
AP37_OH_21 |
Filed Strain-1 |
11 |
1,3,5,7 |
0,1,2,3,4,5,6,7 |
10 |
|
Total |
40 |
|||||
| WOA= weeks of age DPC=days post-challenge |
|
|||||
3. Avibacterium paragallinarum isolation and quantification
Choanal swab, collected in 1.5 ml of BHI. Isolation will be attempted according to the methods described previously (Dufour-Zavala, 2008). The remaining media will be centrifuged to yield a pellet. DNA will be extracted from the pellet using spin-column-based commercial extraction kits following the manufacturer’s instructions. For the quantification of the Avibacterium, qPCR will be conducted to amplify the RecN gene using SYBR Green Master Mix (Thermo Fisher Scientific, USA) (Kuchipudi et al., 2021). The mean clinical sign score and mean bacterial quantity from four experimental groups will be tested for statistical significance. In this way, the efficacy of the live-vaccine candidates in commercial layers will be evaluated.
4. Estimation of Sample Size and Statistical Analysis
The primary statistical outcome from the safety and efficacy trial will be the mean clinical score from different treatment groups. We assume that the birds in the AP challenge groups will show a mean clinical sign score of 1.5 with a standard deviation of 0.58. Assuming the non-challenged birds will not show any clinical signs of IC, a sample size of 10 in each experimental group was determined to have sufficient statistical power to find a significant (α=0.05) difference between groups. Two-tailed T-test will be conducted to perform the test of significance between experimental groups and the p-value < 0.05 will be considered significant. All the animal experiments will be conducted in duplicates to confirm our findings after obtaining extramural funding.
Update Jan 2026
Experimental Modifications
[In an initial, (unreported) safety experiment, each experimental group consisted of 10 birds, of which five were challenged via the natural route of infection (intranasal and ocular) and five were challenged by infraorbital sinus inoculation at a dose of 1 × 10⁷ CFU per bird. No clinical signs were observed in the vaccinated groups, and only mild, non-scorable signs (lethargy) were noted in the positive control group challenged with strain MEL-AP10. These observations indicated that the challenge dose and route were sub-optimal to reliably induce measurable disease, and that uniform challenge via infraorbital sinus inoculation was required. Based on these findings, the study design was revised to administer the inoculum exclusively via the infraorbital sinus at an increased dose of 0.2 mL containing 1 × 10⁸ CFU/mL, representing a twofold increase relative to the initial challenge dose.
Based on the results of the initial challenge trail, we carried out slight modifications. Under this revised design, it was anticipated that birds in the vaccine and negative control groups would no clinical signs (expected mean clinical score ≈ 0), whereas birds in the positive control group would develop some measurable disease (expected mean clinical score ≈ 1.5), and an estimated variance of 0.58, based on the observations and using the predefined clinical scoring system. Given these expected effect sizes and the results of the preliminary safety experiment, the safety evaluation was conducted using five birds per experimental group, which was sufficient for statistical validation.
Update Jan 2026
We obtained the necessary IACUC approval to carry out the animal experiment , and the protocol number is R-JUN-25-26. We were unable to procure 6 weeks old commercially raised layers because of the lack of suitable vendor and the number of chickens required for the experiment. We carried out the safety experiment in the non SPF 60 days old Dekalb White layers, which were procured from the commercial vendor (Hendrix Genetics, EPhrata, PA) vaccinated at hatch against a panel of bacterial and viral disease of the layers and were raised until the age of challenge with appropriate housing, feed, and water required.
Growth of Avibacterium paragallinarum and Molecular confirmation
After incubation of the Avibacterium paragallinarum strains under microaerophilic conditions at 37 °C for 36 h, colonies were readily visible on chocolate agar. The vaccine strains (AP36_OH_21 and AP37_OH_21) produced relatively larger, fewer, and rougher colonies on the first passage, whereas the virulent strain AP10_DEL_20 formed numerous minute, dewdrop-like colonies characteristic of A. paragallinarum. For each strain, the relationship between optical density and viable counts was determined using spectrophotometric measurements and corresponding CFU enumeration. The identity of both the recovered colonies and the prepared inocula was confirmed as A. paragallinarum by PCR amplification of nearly 500 bp HPG2 gene.
Safety of the Live Vaccine Candidates
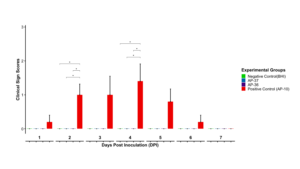
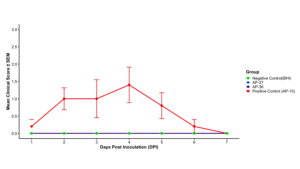
The majority of birds challenged with the virulent field strain AP10_DEL_20 developed clear clinical signs of infectious coryza, with clinical scores increasing from day 2 post-inoculation, peaking between days 3 and 4, and subsequently declining, consistent with the known course of infection. In contrast, birds inoculated with the live vaccine candidates (AP-36 and AP-37), as well as those in the negative control group, exhibited no detectable clinical signs throughout the 7-day observation period, with clinical scores remaining at zero. Clinical scores in the AP10_DEL_20 group were significantly higher than those in the vaccine and negative control groups at days 2 and 4 post-infection; however, scores declined rapidly after day 4, and no significant differences were observed among groups thereafter. By day 7 post-infection, clinical disease had resolved, and no clinical signs were observed in any experimental group (Figure 1A, Figure 1B). The results indicate that the live vaccine candidate do not induce clinical signs of disease in the evaluated chicken and are promising candidate as live vaccine candidates.
An important consideration from this work is the need to distinguish clinical safety from colonization dynamics. Although no clinical signs were observed in vaccine groups, persistence and shedding of A. paragallinarum may still occur at subclinical levels. To evaluate this, we are evaluating the persistence and shedding of the bacterium using a recN-based qPCR assay on choanal swab samples. These data will determine whether differences in colonization or shedding exist among the vaccine candidates and the virulent strain, providing critical insight into transmission risk.
Update Jan 2026
Conclusions
Following IACUC approval (protocol R-JUN-25-26), safety trials were conducted in non-SPF, 60-day-old Dekalb White layers raised mimicking commercial conditions. Birds challenged with the virulent field strain AP10_DEL_20 developed clinical signs consistent with infectious coryza, confirming the robustness of the challenge model, whereas birds inoculated with the live vaccine candidates AP-36 and AP-37, as well as negative controls, showed no detectable clinical signs throughout the observation period. These results demonstrate that the candidate live vaccines do not induce disease under the conditions tested and meet the primary safety objective of the study. Ongoing recN-based quantitative PCR analyses of choanal swabs aimed at evaluating bacterial shedding helps to determine whether subclinical colonization is present, or if there is any risk of potential persistence and transmission of these different strains.
References
- Chen, X., Miflin, J. K., Zhang, P., & Blackall, P. J. (1996). Development and application of DNA probes and PCR tests for Haemophilus paragallinarum. Avian diseases, 398-407.
- Bragg, R. R. (2002). Virulence of South African isolates of Haemophilus paragallinarum. Part 1: NAD-dependent field isolates.
Education & outreach activities and participation summary
Participation summary:
Update (August, 2024- January, 2025)
In a seminar program organized by the Center of Excellence in Microbiome Sciences at the University of Maryland, I did a presentation on Avibacterium paragallinarum (AP) infection in poultry and and various strategies to combat it. I put into light the impact of AP infection in the laying and meat-producing chickens in the United States, particularly in the Northeast region. In my presentation, I provided an brief overview of infectious coryza, including its pathogenesis, impact, and potential mitigation strategies such as the use of a live vaccine. I also highlighted live vaccines for mitigating infectious coryza as a key research direction, alongside the use of probiotics, which I am implementing in our laboratory, separately, among several poultry disease researchers, professors, UMD extension personnel, graduate students working on animal health, and food safety.
Update January, 2026
On April 26, 2025, during Maryland Ag Day, we participated as volunteers and exhibitors representing the Department of Veterinary Medicine and hosted an educational booth featuring infographics on animal and zoonotic diseases. Among the displayed materials was a poster on infectious coryza that highlighted its economic impact on poultry production and current control strategies. The exhibit attracted strong engagement from students, members of the general public, and approximately ten individuals involved in backyard poultry operations. Through interactive discussions, we shared information about infectious coryza and discussed ongoing research efforts supported by NE-SARE, fostering awareness of the disease and its management among diverse audiences.