Progress report for GNE24-334
Project Information
Hydroponic farming continues to grow in popularity. Recent outbreaks linked to hydroponic produce highlight the need for improved understanding of how foodborne pathogens persist in these systems and provide a way to control them. The aim of this proposal is to (i) study the persistence of Salmonella, E. coli, and Listeria, in two types of hydroponic systems (deep water culture and Kratky) and to (ii) assess the efficacy of bacteriocins and bacteriophages as a pathogen control strategy. In the first objective of this project, pathogens will be inoculated into water to assess their persistence. In the second objective of this project, pathogen control interventions will be assessed to reduce food safety risk while maintaining, or promoting, plant health and productivity. I hypothesize that under conditions where biofilms do not form, the Salmonella and E. coli strains will persist for two weeks, while the Listeria strains will persist for one week as suggested by a previous study (Ilec et al., 2022). I also hypothesize that a treatment of bacteriocins and bacteriophages will reduce pathogen load. The findings from this project will help understand the microbial risks and how to manage them in hydroponic farms. Findings will be disseminated among Northeast hydroponic farmers and beyond through the Mid-Atlantic Fruit and Vegetable Convention.
Objective 1: Evaluate the persistence of Salmonella, E. coli and L. monocytogenes in hydroponic systems.
Expected outcome: Characterization of Salmonella, E. coli and Listeria persistence in hydroponic systems in a controlled laboratory setting.
Deliverables: Spreadsheet and graphs with concentrations of Salmonella, E. coli and L. monocytogenes in a nutrient solution over the growing period and the final harvested produce grown in the DWC and Kratky hydroponic systems.
Potential pitfalls and alternative approaches: We plan to inoculate leafy greens seeds with an inoculum at 108 CFU/ml of Salmonella, E. coli and L. monocytogenes. If this inoculum level leads to concentrations of pathogens that are below the limit of detection of our microbial detection methods, we will collect a larger volume of a sample and filter it in order to obtain quantities of pathogens.
Objective 2: Assess pathogen control steps aimed at reducing pathogens on seeds
Expected outcome: Developed and assessed pathogen control step for seeds that is accessible and feasible for hydroponic farmers to implement to reduce the risk of introducing Salmonella, E. coli and L. monocytogenes contamination in hydroponic systems.
Deliverables: A mitigation step made publicly available to hydroponic farmers in the NE region through a publication and communication at the Mid-Atlantic Fruit and Vegetable Convention.
Potential pitfalls and alternative approaches: If the mitigation step negatively impacts seed germination, plant growth or plant quality, we will evaluate alternative methods to control bacterial growth. Preliminary small-scale experiments will be conducted to assess these factors.
The purpose of this project is to investigate the persistence and control of bacterial foodborne pathogens in deep water culture (DWC) and Kratky hydroponic farming systems. Hydroponic farming is estimated to grow another 12.4% from 2024 to 2030 (Grand View Research.,2024). Hydroponic farming can reuse water and be done in urban areas where it is often more difficult to grow and access fresh produce (Zhen,2021). Hydroponic farming helps increase sustainability through water conservation, reduction of greenhouse emissions, and minimal use of chemicals (Pomoni et al.,2023). Despite these benefits, limited research has been conducted on how bacterial foodborne pathogens behave in these systems.
The DWC and Kratky systems consist of a water reservoir with a raft to support the plants during growth. The DWC has a pump to oxygenate the nutrient solution, while the Kratky system does not. The Kratky system is an easy adaptation from the DWC system allowing more data to be collected with no additional setup costs. BrightFarms, Gotham Greens, and Greens Do Good in the Northeast region the DWC technique.
There are gaps in hydroponic research. One study assessed the survival of Salmonella Typhimurium and Listeria monocytogenes in the nutrient film technique (NFT) hydroponic system (Ilic et al.,2022), while others assessed E. coli and Salmonella in a deep flow technique (DFT) system (Shaw et al.,2016; Xylia et al.,2022; Li et al.,2022). To the best of our knowledge, the persistence of foodborne pathogens has not yet been investigated in the Kratky or DWC system. Our proposed project therefore aims to fill this gap in knowledge by studying the persistence and control of Salmonella, E. coli, and Listeria monocytogenes in the DWC and Kratky systems.
As hydroponic farming continues to grow in popularity, we must understand the associated food safety risks. Foodborne pathogens Salmonella, E. coli, and L. monocytogenes have previously been found in produce grown in hydroponic farms (FDA; McClure et al.,2023; Wang et al.,2020). Furthermore, they have been shown to spread, form biofilms, and be taken up and attached to plants (Riggio et al.,2019; Hirneisen et al.,2012; Kyere et al.,2020). If hydroponic farming is to safely advance, we need to improve the understanding of how these bacteria behave in these hydroponic systems to develop effective control strategies.
It's important to understand how hydroponic systems can be contaminated. Peat moss plugs, used for germination, have been a hot spot for bacterial growth in hydroponic systems (Dankwa et al.,2019). The seed germination environment has high water and nutrient retention providing an environment conducive to bacterial growth. If contaminated seeds enter the facility, the contaminants can multiply in the germination area and spread rapidly in the enclosed hydroponic system (Saldinger et al.,2023).
We propose to use bacteriocins and bacteriophages as a potential intervention. Hydroponic farming reduces the need for harsh chemicals, an aspect we tried to preserve for this proposal.
We will set up a hydroponic system inside a biosafety level-2 food safety pilot plant, a state-of-the-art facility at Penn State that is available for controlled food safety experiments. Our experimental design will include six replicate hydroponic units with a flexible design that can be used for DWC and Kratky setups. Leafy green seeds will be inoculated with a bacterial cocktail of E. coli, Salmonella, and Listeria. The contaminated seeds will be germinated and planted into all six hydroponic system replicates. Three of the systems will be used for an assessment of the interventions at the germination stage, while the other three serve as positive controls. One type of hydroponic system at a time will be assessed. The ambient temperature and humidity will be controlled to mimic commercial indoor growing facilities.
After planting, the nutrient solution, plants, and roots be sampled. E. coli, Salmonella, and Listeria will be enumerated on the appropriate selective media. In addition to microbial testing, plant material will also be tested to evaluate the leafy green quality upon harvest. All collected data will be analyzed using a repeated measures ANOVA in RStudio. This information is expected to lead to the development of food safety practices that can help Northeastern farmers produce safe food, improve the region’s economic viability, and advance sustainable growing practices.
Research
Hydroponic system setup. A hydroponic system was built inside a BSL-2 food safety pilot plant at The Department of Food Science at The Pennsylvania State University. A total of six replicate systems were constructed with bins, rafts, and artificial lighting. Three of the six replicates will be used for growing plants using untreated seeds and three will be used for growing plants using seeds that had been treated to reduce the load of foodborne pathogens (Salmonella, E. coli, L. monocytogenes) inoculated onto seeds. This setup will allow us to investigate pathogen persistence with and without seed treatment. The same experiment will be conducted using the DWC and Kratky hydroponic systems. For the Kratky setup, we will use an air pump, which will allow us to alternate between DWC and Kratky systems. Two independent experiments, each with three replicates of treatments, will be run as a DWC setup and two will be run as the Kratky setup. The pH and electric conductivity of the systems and the temperature of the room will be continuously monitored and adjusted as needed.
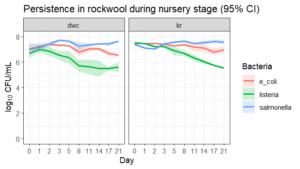
Seed inoculation and germination. A batch of butterhead lettuce was purchased. This variety was selected based on what we commonly see NE hydroponic farmers growing. We inoculated Salmonella Javiana and Thompson, Escherichia coli STEC O179:H8 and EPEC O13:H6, and two strains of Listeria monocytogenes at ~6 log10 CFU/mL in reverse osmosis water and applied it onto rockwool containing the lettuce seeds (28 cubes per 3 system replicates). Next, the inoculated seeds were germinated for 3 weeks in rockwool cubes until they were ready to be transplanted (Objective 1). This design will help determine the persistence of these pathogens (using an untreated control; Objective 1). After the seeds are germinated and established, the plants will be transferred to the hydroponic system.
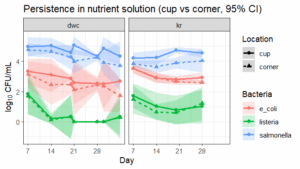
Rockwool, nutrient solution, and leafy greens testing. Pathogen persistence was assessed during the 3-week nursery stage and the subsequent 4-week plant growth stage in deep water culture (DWC) and Kratky (KR) hydroponic systems (Objectives 1 and 2). Pathogens were quantified in nutrient solution collected near plant roots and from system corners throughout the plant growth stage, as well as in rockwool cubes and on plant root surfaces at harvest. At each sampling point, 10 mL of nutrient solution was collected from each system, and the volume was replenished with stock nutrient solution as needed.
All samples were serially 10-fold diluted and spread plated onto selective media for pathogen detection: xylose lysine deoxycholate agar for Salmonella, eosin-methylene blue agar for Escherichia coli, and Agar Listeria according to Ottaviani and Agosti for Listeria monocytogenes. Plates were incubated at 37 °C for 24, 24, and 48 hours, respectively.
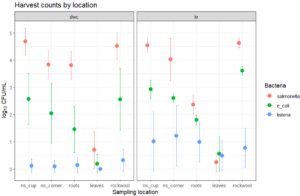
At the end of the growing period, 25 g of the outer portion of harvested leafy greens were aseptically collected and homogenized by stomaching in 225 mL of phosphate-buffered solution. Plant roots were also aseptically collected, and phosphate-buffered solution was added based on root weight prior to homogenization by stomaching. Produce and root homogenates were independently diluted, plated, and analyzed using the same methods as nutrient solution samples. Pathogen counts were recorded and used for statistical analyses to evaluate pathogen persistence and/or die-off across system type, location, and growth stage (Objectives 1 and 2).
Produce quality assessment. Under the guidance of Dr. DiGioia, I will assess the quality of the harvested produce. I will measure the total weight of the grown plant. I will also place a place in an oven to remove moisture so the total dry matter can be measured. Finally, a fluorometer will be used to measure chlorophyll levels. This will help assess the impact of the PAA measure on produce quality.
All data will be analyzed in RStudio using a repeated measures ANOVA to quantify the changes in the concentration of target pathogens over the growing period, to assess the effect of the seed treatment on pathogen concentrations, and to compare the produce quality findings. X The significance value will be set at P < 0.05. The Kratky and DWC systems will be evaluated separately as well as compared to each other to assess the differences between the two system types in terms of pathogen persistence and/or die-off. The change in pathogen concentrations will be plotted to visualize the differences over time and between systems. The highly controlled laboratory and system environments will help reduce random environmental effects and improve the statistical power to detect significant effects of the treatments if they exist.
2025 Update
E. coli and Listeria concentrations declined in the nursery and plant growth stage (p < 0.001). Salmonella counts increased during the nursery stage (p = 0.029; 0.01 log units/day) but did not change during the plant growth stage in the systems (p = 0.35). E. coli and Salmonella concentrations were lower at system corners than near the plant roots during the plant growth stage (linear mixed model, p = 0.047 and p = 7.14 × 10⁻⁹, respectively). Pathogen concentrations did not differ between the DWC and KR systems (p = 0.7). At the end of the growth period, all three pathogens were detected on roots (Salmonella at 3.1 ± 0.4, E. coli at 1.6 ± 0.6, Listeria at 0.6 ± 0.4 CFU/g) and rockwool (Salmonella at 4.6 ± 0.3, E. coli at 3.1 ± 0.9, Listeria at 0.6 ± 0.3 CFU/g ).
2024 Update
I spent last semester ordering supplies and setting up my hydroponic system in the BSL2 lab. The lights and sensors are set up and it has been confirmed that the tubs have no leaks. I have also tested one of my treatments on butterleaf lettuce seeds to ensure it does not negatively impact germination rates. All of this work will allow me to start my first replicate of this study in the Spring semester.
Salmonella concentration increased during the nursery stage and persisted through plant growth in the system. E. coli and Salmonella concentrations were consistently higher near plant roots than at system corners, highlighting root-associated microenvironments as key hotspots for pathogen persistence independent of hydroponic system design. We sought to establish persistence of these three pathogens in two hydroponic systems, which we did. Moving forward, we will focus on intervention steps.
Education & outreach activities and participation summary
Participation summary:
I presented this research at a Penn State Legislator research tour. Around 50 legislators were reached and the importance of food safety in hydroponic farming systems was highlighted. I have also submitted an abstract to present a talk at the 2025 International Associated of Food Protection Conference this summer in Louisiana.
I will present my findings at the Mid-Atlantic Fruit and Vegetable Convention in 2026. My collaborator, Dr. Francesco Di Gioia will serve as the chair of the hydroponic session at the Convention, providing an excellent opportunity for me to present findings from the proposed project in a talk or a poster. Presentation of my research findings through outreach at the Convention will help me disseminate my research findings directly to growers, which are the primary stakeholders of my proposed project. I am also planning on reporting results of this project in a peer-reviewed scientific publication that can help guide future hydroponic farming food safety research and regulations. This project is expected to generate new information that can be used to improve microbial food safety of hydroponically grown produce without compromising produce quality. In addition to formal presentations and research papers, I am also excited to share the findings of this project with the NE hydroponic farmers I have connected with. During my site visits they have expressed great interest in further research and collaboration. I am confident the findings of this study will help meet those needs, foster collaboration, and lead to future research and breakthroughs.
Project Outcomes
The goal of my project is to find sustainable and efficient ways to improve the safety of hydroponic systems.
I learned that my initial plan to inoculate the seeds didn't result in a high enough inoculation level for testing. Instead, I inoculated the rockwool after the seeds were placed inside. This allowed sufficient starting inoculation levels for the experiment. I also learned that PAA negatively impacts seedling germination and health. Due to this, I found bacteriocins and bacteriophages to test instead.
I have gained a substantial amount of knowledge building my hydronic system. I learned how to approach my research question while considering different aspects, like plant science and engineering. I know am more confident in how hydroponic systems work and what they need to be successful. I also learned a lot more about the energy requirements from grow lights and alternative options that are more sustainable. I'm excited to continue this experiment and see what data is generated.



