Final report for GS15-144
Project Information
Harlequin bug, Murgantia histrionica (Hahn), is an important pest of Brassica crops like collards, kale, mustard greens, and broccoli in the southern United States (Wallingford et al. 2011). Unlike the major lepidopteran pests and aphids that attack brassica crops, there are relatively few natural enemies of harlequin bug (HB) and, in general, only broadspectrum insecticides are effective for control. Use of these insecticides are not compatible with IPM programs. Thus, alternative management strategies for HB are needed for maintaining the sustainable balance of IPM in these important vegetable crop systems in the southern U.S. Pest populations of HB are highly variable and unpredictable from farm-to-farm, and therefore accurate monitoring of activity would greatly improve IPM decision-making and the timing of control tactics. To our knowledge, there is no monitoring device or proven trapping system for this pest. The recent identification and synthesis of the HB aggregation pheromone, mugantiol has led to the commercial availability of a potent attractant for this important pest. We are now exploring ways to utilize this new tool for pest management.
In a series of lab choice tests we determined a color preference of harlequin bug for darker colors (black and green) over lighter colors. After reconfirming harlequin bug color preference in the field, we looked at trap shape and circumference to construct a trap-prototype. Since our goal is to build a trap-and-kill device, we assayed whether a commercially-available long-lasting deltamethrin insecticide treated mesh would provide adequate contact mortality of harlequin bug and how quickly it would do so (LT50 bioassay). In conjuncture with our field studies we also investigated the additive and/or synergistic effects of combining the aggregation pheromone murgantiol with benzyl isothiocyanate (a mustard plant host volatile). Isothiocyanate is the chemical group –N=C=S, formed by substituting the oxygen in the isocyanate group with a sulfur. Many natural isothiocyanates from plants are produced by enzymatic conversion of glucosinolates.
Our field and lab behavioral studies determined that visual stimuli are synergistic to volatile attractants. Visual cues—such as color, surface area, and shape—are additive in attracting harlequin bug. Dark colors attracted more bugs than lighter colors. When comparing green and yellow traps baited with murgantiol we found that yellow traps were hazardous to beneficial predatory coccinellids, whereas dark green was not.
Harlequin bug response to the aggregation pheromone, murgantiol, was low during the summer growing season, even when combined with various visual stimuli. It was not until we combined trap-like devices with murgantiol and benzyl isothiocyanate that we saw significant response within dispersing on-farm populations. These are interesting findings as they suggest that murgantiol & benzyl isothiocyanate act synergistically to attract HB. The results of these studies should provide some important information for the development of an effective trap for HB that growers can use on their farms to detect invading populations and possibly to attract and kill them.
Objective 1. To determine visual cues such as color and trap-architecture that attract more harlequin bugs.
Obj. 1A. Assess HB preference for colors
Obj. 1B. Determine the efficacy of deltamethrin-treated mesh (long-lasting insecticidal mosquito nets) at killing HB - for use on traps
Obj. 1C: To evaluate the field response of HB and natural enemies to dark green versus yellow color in the presence of murgantiol pheromone
Obj. 2: To evaluate the efficacy of the HB aggregation pheromone, murgantiol with and without the addition of mustard oil volatiles
Cooperators
- (Researcher)
Research
Objective 1. Preference in harlequin bug for certain visual cues such as color and trap-architecture. (manuscript in press in Environmental Entomology 2017)
Obj. 1A. HB preference for colors
Materials and Methods
Insect cultures. Field-collected M. histrionica adults from Painter, VA (37o 35’04.61” N, 75o 49’15.64” W; elevation = 9 m) in mid-April 2015 initiated a colony for this study. Bugs were reared on potted collards (Brassica oleracea: VATES or Champion) in a greenhouse under summer-like conditions (16:8 L:D and 20 to 30oC). Adults and large nymph (4th through 5th instars) were sourced in our laboratory color choice experiments.
Laboratory Color Choice Assay. Corrugated polypropylene (Coroplast, Inteplast Group, Vanceburg, KY) panels (dimensions at 0.6m × 0.6m × 4mm thickness) were assembled with clear packing tape into a hexagonal choice-test arena with in an interior diameter = 1.42 m interior diameter. Reflectance spectra data was collected using a DH-2000-BAL light source (Ocean Optics Inc., Dunedin, FL), Ocean Optics SpectraSuite software (Ocean Optics Inc., Dunedin, FL) and a USB4000 Spectrometer (Ocean Optics Inc., Dunedin, FL) calibrated with a Labsphere Reflectance Standard AS-01158-060 USRS-99-010 BZ37A (Labsphere, Inc, North Sutton, NH). The arena was designed for a 6-way color choice test including these specific colors: green (510nm), red (624nm), black (no spectral peaks), white (420 - 880nm), yellow (568nm), and purple (440nm, 677nm). The hexagonal arena was illuminated under “cool-white” (4100K) fluorescent lighting (Philips Lighting Corporation, Somerset, NJ) with ambient temperatures ranging from 20 – 23oC, and rotated in a six times in order to eliminate panel location bias.
Randomly-selected harlequin bugs were grouped by sex and life stage and starved 12 to 16 h before conducting experiments. Grouped bugs were organized into units of five individuals in a 200ml centrifuge tube and immobilized on ice for three minutes before gently introducing them into the center of the arena. Five minutes were allotted for all bugs to disperse and select a color panel once one of the five bugs regained motion. Color selection “choice” was determined if an individual bug made contact anteriorly or oriented within 2 cm of the colored panel.
With three replicates of the experiment, a total of 89 females, 86 males and 91 late-instar nymphs were assayed. Bugs tested had no prior experience to the arena and were not reused in subsequent assays.
Field experiment. A six-way color choice experiment using corrugated polypropylene, with the same dimension and colors listed in our lab color choice assay, was conducted at Kentland Farm in Whitethorne, VA (37o 12’06.73”N, 80o 33’52.27”W; elevation = 517 m) in early autumn 2015. Color panels were deployed as traps in a bare tilled field without host plants to intercept local populations of harlequin bugs on September 25, 2015. Trap surfaces were treated with a pyrethroid insecticide bifenthrin (Bifenture 2E, United Phosphorous Corporation, King of Prussia, PA), at 0.2 g per liter applied with a manual pressurized backpack sprayer (Solo Sprayers, Newport News, VA) to knock down intercepted bugs. Naturally dispersing populations were assumed to be highest near a 2015 spring-and-summer mustard and collard field, and therefore we situated the trap transect 6 m distant from the edge of, and parallel to, the east-west axis of the field with the faces of the square traps exposed north and south.
Six weeks before the field color choice experiment, the collard and mustard field was mowed (leaving ca. 0.5 m tall residue) and seeded with rye (Secale cereale L.) cover crop to encourage a slow dispersal of harlequin bugs out of the field. The color choice test transect was a randomized block design of five replicate series with traps spaced 3.66 m on center. One week before deploying the traps, the infested field was mowed low (leaving ca. 0.15 m host residue, at this point fairly desiccated) to encourage dispersal of harlequin bugs. Dead harlequin bugs were collected within a 0.2 m radius of each panel trap centroid 48 h after setting out traps, and every 72 h for 9 days.
Statistical Analysis. All data were analyzed in JMP 11.0.0 (SAS Institute, Cary, NC). Residuals conformed to normality, therefore one-way and two-way ANOVA were used to test for statistical difference in color response from lab and field experiments, and Fisher’s exact test for determining group differences in lab assay. Data reported were non-transformed with α set at =0.05. Mean numbers of bugs responding were separated with a post-hoc Tukey’s HSD test.
Obj. 1B: Lethal time on commercial deltamethrin-treated screens (long-lasting insecticidal mosquito nets)
In order to determine the feasibility of using deltamethrin-treated screens to kill harlequin bugs in a future trap device, we needed to first determine if the treated screens were effective at immobilizing and/or killing harlequin bugs.
Dates: June 12 – 14, 2015
Treatments:
Black DeTerrance™ mesh from Vestergaard frandsen - cut 120mm circles + excised collard leaf - Exposure times: 90, 45, 30, 15, 5, 1 minutes
Control no-mesh (collard leaf)
Design:
Four replicates of deltamethrin-treated mesh (DeTerrance™) were cut into 120mm circles to fit snug into respective Petri dishes. Field collected harlequin bug late instars (3rd to 4th) from Beltsville, MD were separated into random groups of 10 bugs. Each group of nymphs were gently placed ventral side down on the mesh/Petri dish combo, with the lid fit snug to limit upward movement onto untreated surfaces of the Petri dish. Exposure times followed at 90, 45, 30, 15, 5, & 1 minutes by removing the mesh from respective Petri dishes. Immediately after timed exposure bugs were asses for mortality, moribundity, and no visible effects on motor response. Bugs were retained in respective petri dishes for post exposure effects (moribund, mortality & no-effects) at 24, 32, & 56 hrs. Data were analyzed using Simple Probit Analysis (JMP Software, SAS Institute: SimpleProbit.jmpaddin). Time in minutes was transformed logarithmically (alpha set at 0.05) to predict LT50, LT75, LT90 and LT95.
Obj. 1C: To evaluate the field response of HB to dark green versus yellow color in the presence of murgantiol pheromone
Dates: Sept.8.2015 – Sept.11.2015
Treatments:
Design:
Green versus Yellow Corrugated plastic panels (each baited with a single murgantiol lure) were wrapped in a single layer of deltamethrin treated mesh. For green panels we used black mesh since we did not have a commercially available green insecticide treated mesh. As for the yellow panels, we used yellow insecticide treated mesh.


Green and yellow panels were grouped in a two-way choice test blocked in four replicates around overwintered collard plot. Treatments were spaced 10m apart and replicates spaced 33m apart. The overgrown, weedy, collard plot was inspected for harlequin bugs prior to setting up the experiment. Panels were centered in 1.0m2 of bare soil, to limit inference from plant visual and volatile cues. Additionally, the trap was situated flush to the soil surface to ensure bugs were able to make an unobstructed contact with the trap. With panels arranged the plot was high mowed (image #) to disperse resident insects. At 48hrs, dead harlequin bugs and coccinellid species (determined with field ID markers) were counted within 30cm of the trap centroid.
Obj. 2: To evaluate the efficacy of the harlequin bug aggregation pheromone, murgantiol with and without the addition of mustard oil volatiles.
Dates: Sept.8.2015 – Sept.11.2015
Field experiment was conducted at Kentland Farm in Whitethorne, VA to determine effective shape of dark-green trap devices, and determine significance of benzyl isothiocynate at high and low emissions as a synergist to murgantiol.
Treatments:
Square vs. Pyramid vs. Ramp trap combined with either murgantiol, murgantiol l+ low benzyl isothiocynate, or murgantiol + high benzyl isothiocynate



Design: Randomized complete block design. A pyramid trap, used by Leskey et al. (2015) in brown marmorated stinkbug trapping traps, ramp trap used in trapping banana weevil and square panel used earlier in lab color choice test, were evenly coated with dark green spray paint (Rust-Oleum, Gloss Hunter Green, UltraCover Paint+Primer, Vermon Hills, IL). Traps are spaced nine feet apart from their centroid along the east-west transect of a field margin south of two fields with summer cole crops; the most adjacent were collards (var. champion) neighbored by a mustard trap crop experiment.
One week prior to establishing treatments host residue in neighboring plots was mowed low to encourage harlequin bug dispersal. Five days later cabbage seedlings were planted in-between trap locations, at three to a row by three rows.
Nine treatments in total for each of the four blocks, which were each baited with a single murgantiol septa (loaded at ##) with and without either low or high doses of benzyl isothiocynate (technical grade, 94%, Sigma Aldrich). Low and high volatile emissions rates were dispensed with one and two, respectively, 75mm capillary tubes with _#_ interior diameter (Fischer Scientific Co., Pittsburgh, Pa.; cat. no. 02-668-68). Once in the field capillary tube(s) were pierced through Teflon seal in a 1ml gc-vial () loaded with 200ul of BITC. Individual vials and septum were affixed 15 – 25 cm from the soil surface to traps with a small green zip-tie, and stapled as necessary.
Trap surfaces were treated with a pyrethroid (Bifenthrin, United Phosphorous Corporation, at 3.2ml/ga + surfactant mixed in a backpack sprayer) to knockdown intercepting bugs for more accurate counts. Dead harlequin bugs were collected within 20cm radius of trap centroid 72hrs after setting out the traps. Since the bugs were so numerous (several thousand individuals), counts were tallied later in the laboratory, accounting for sex and life stage (adult versus nymph).
Results
Laboratory Color Choice Assay
Approximately 92.5% of the released M. histrionica (246 of the 266 bugs tested) dispersed in the lab color choice arena and made a color choice according to the choice criterion. However, adults were significantly more likely (Fisher’s exact test, P = 0.0035) to respond than nymphs with 7 of the 175 (4%) adults and 13 of 91(14.3%) nymphs unresponsive (i.e. did not disperse) in this assay. Color discrimination among red, yellow, green, purple, black and white in the lab was statistically significant for females, males and nymphs (Two-way ANOVA: F = 21.93; df = 5, 306; p < 0.0001). A two-way ANOVA indicated a significant interaction (F = 2.04; df = 10, 306; p = 0.0296) between color panel preference and responsive life stages (i.e. males, females, and nymphs). A post-hoc Tukey’s HSD test revealed that red and green were equally attractive colors for females in the lab experiment. Green alone was the most attractive color for females, nymphs and males; females and males were least likely to respond to white and yellow, and nymphs were similarly least responsive (see Table 1 for complete post-hoc analysis).
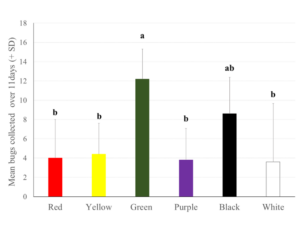
Mean harlequin bugs collected at insecticide treated traps during a field experiment occurring over consecutive 72hr collection periods over the course of 11days in early-October 2015 located in Whitethorn, VA . Six colors in a randomized complete block design with a five replicate series intercepted naturally dispersing bugs. Bars with the same letter are not significantly different (Tukey’s HSD test p < 0.05).
Field experiment
There was a significant effect of color treatment on numbers of M. histrionica arriving to insecticide-treated panels in the field (F = 4.28 df = 5, 24; p = 0.0063). Nymphs were overall more numerous relative to adults in this field study, with 38 adults and 145 nymphs collected at traps. Total bugs intercepted by traps in the field mirrored lab choice responses with green panels attracting significantly more harlequin bugs than the other five colors tested; black panels ranked second (Fig. 1). Red was among the least preferred colors along with yellow, purple and white.
Table 1
|
Mean Harlequin Bug Response in Lab Color Choice Test |
||||||||
|
Females |
Males |
Nymphs |
||||||
|
x̅ |
s |
x̅ |
s |
x̅ |
s |
|||
|
Red |
1.56a |
1.20 |
0.89abcd |
0.96 |
0.67abcd |
0.69 |
||
|
Yellow |
0.17d |
0.38 |
0.17d |
0.38 |
0.22cd |
0.43 |
||
|
Green* |
1.44a |
1.15 |
1.39a |
1.24 |
1.50a |
1.04 |
||
|
Purple |
0.72abcd |
0.83 |
0.78abcd |
0.88 |
0.39bcd |
0.50 |
||
|
Black |
0.78abcd |
0.55 |
1.33ab |
1.14 |
1.17abc |
0.92 |
||
|
White |
0.17d |
0.38 |
0.00d |
0.00 |
0.39bcd |
0.50 |
||
|
Mean harlequin bugs (x̅) and corresponding standard deviation (s). Numbers with a letter in common are not significantly different according to a Tukey’s HSD, P < 0.05
* indicates most preferred color shared by all groups tested |
||||||||
Discussion
Our experiments were conducted in the absence of added olfactory stimuli. Stenberg and Ericson (2007) suggested that there may be greater sensitivity to visual over olfactory cues when specialist herbivores are starved than when satiated. Bugs were starved prior to our lab color choice test experiments and may have been more sensitive to visual cues and lab conditions than under natural field conditions. Regardless, our field and lab experiments yield consistent results with darker colors, specifically green, showing the most promise for trap construction materials for managing and monitoring harlequin bug populations.
A positive attraction to green in harlequin bug may be associated with their attraction to Brassica host plants of high nutritional quality (Prokopy and Owens 1983, Loader and Damman 1991). The addition of red color components to a green or black trap could perhaps enhance attraction to M. histrionica females, but this idea still needs testing to confirm. Species-specific visual stimuli increase trap captures when added to a monochromatic in other insect pest systems. For example, Domingue et al. (2015) showed that 3-D printed emerald ash borer, Agrilus planipennis, (Coleoptera: Buprestidae) decoy baited branch traps with a (Z)-3-hexen-1-ol lure captured significantly more A. subcinctus than without the visual-decoy. They also observed sex bias for the visual decoys with males becoming ensnared closer to decoy than females (Domingue et al. 2015).
The interaction of color with other factors such as trap shape, background contrasts, and olfactory stimuli, needs to be evaluated before determining the best application of our results to an optimal trap design for monitoring and managing harlequin bug populations. Mixed-isomer syntheses of 10,11-epoxy-1-bisabolen-3-ol (i.e. murgantiol) (Khrimian et al. 2014), are powerful attractants for harlequin bugs in the field, especially in combination with a host plant. Weber et al. (2014) reported a subtle but significant female preference for natural murgantiol isomer ratios over more economically viable crude isomer mixtures with 1:1 (SSRS: SSRR) isomer ratios. Selection of optimal color stimuli would be expected to enhance female attraction to a trap baited with natural or crude mixtures of murgantiol isomers. Future studies should evaluate the combination of both olfactory and visual stimuli of a trap for M. histrionica.
Results - toxicity of the deltamethrin-incorporated mesh to HB: The calculated LT50 was 18.69 min of exposure to the screen (slope = 1.403692; 95%CI = 16.28 and 21.10. Thus, deltamethrin-treated mesh (DeTerrance™) was an effective killing agent for HB and was subsequently used for future experiments of trap development.
Results of murgantiol-baited yellow versus dark green panels in the field:
Murgantiol baited green traps were significantly more attractive than murgantiol baited yellow traps (t-test t(26.95709) = -3.76, p = 0.0008). In addition, yellow was significantly more attractive (t-test t(29.496) =2.14, p = 0.0412) to coccinellid species (cumulatively: Harmonia spp., C. maculata, & Coccinella septempunctata).
Thus, not only was dark green the better color to use for attracting more HB, but it killed fewer ladybeetles than yellow panels.
Results - field evaluation of different trap types and lures: Several significant results were found relative to traps for HB (Table 1), including: 1) low dose among all traps captures more HB than no ITC on traps; 2) High dose among all traps captures more HB than no ITC on traps; 3) Ramp traps with murgantiol and low dose of ITC attract more HB than with no ITC; 4) Square traps with murgantiol and low dose of ITC attract more HB than with no ITC; 5) Square traps with murgantiol and high dose of ITC attract more HB than with no ITC; 6) Ramp traps with murgantiol and high dose of ITC attract more HB than with no ITC; 7) Pyramid traps with murgantiol and low dose of ITC attract more HB than with no ITC. Based on these findings, the fundamentals of an effective trap design would be a low dose (single capillary tube) emitting benzyl isothiocynate combined with murgantiol on a pyramidal shaped device.
Table 1. Ranked p-values of all significant interactions. Since p-value approaches 0.05 in the bottom row it is likely that if the alternative is valid.
|
Alternative Hypothesis |
Interaction |
p value (a = 0.05) |
F |
F - Crit |
|
Low dose among all traps captures more HB than no ITC on traps |
Trap + Murgantiol vs. Trap + Murgantiol & L ITC |
0.00011 |
22.22315 |
4.30095 |
|
High dose among all traps captures more HB than no ITC on traps |
Trap + Murgantiol vs. Trap + Murgantiol & H ITC |
0.00061 |
15.95932 |
4.30095 |
|
Ramp traps with murgantiol and low dose of ITC attract more HB than with no ITC. |
R + Murgantiol vs. R + Murgantiol & L ITC |
0.00455 |
19.39007 |
5.98738 |
|
Square traps with murgantiol and low dose of ITC attract more HB than with no ITC. |
S + Murgantiol vs. S + Murgantiol & L ITC |
0.00705 |
16.06809 |
5.98738 |
|
Square traps with murgantiol and high dose of ITC attract more HB than with no ITC. |
S + Murgantiol vs. S + Murgantiol & H ITC |
0.00711 |
16.00739 |
5.98738 |
|
Ramp traps with murgantiol and high dose of ITC attract more HB than with no ITC. |
R + Murgantiol vs. R + Murgantiol & H ITC |
0.02420 |
8.96255 |
5.98738 |
|
Pyramid traps with murgantiol and low dose of ITC attract more HB than with no ITC. |
P + Murgantiol vs. P + Murgantiol & L ITC |
0.03442 |
7.42529 |
5.98738 |
|
Pyramids traps with murgantiol and high dose of ITC attract more HB than with no ITC. |
P + Murgantiol vs. P + Murgantiol & H ITC |
0.05801 |
5.46607 |
5.98738 |
Educational & Outreach Activities
Participation summary:
Dimeglio, A., T. P. Kuhar, and D. C. Weber. 2017. Color Preference of Harlequin Bug, Murgantia histrionica (Heteroptera: Pentatomidae). Journal of Economic Entomology in press
DiMeglio A.S. (2016) “Lethal Exposure Time of Harlequin Bug Nymphs on ZeroFly® (a long-lasting insecticidal net)”: Published in Kuhar T.P., Schultz P.B., Doughty H., “Arthropod Pest Management Research on Vegetables in Virginia (2015)”, 64 -67.
Northeastern Plant, Pest, and Soils Conference., “Developing an effective trap-and-kill device for harlequin bug: importance of trap color and semiochemical attractants” Philadelphia, PA; January 5, 2016
2015 ESA Annual Meeting with the American Society of Agronomy, the Crop Science Society of America, and the Soil Science Society of America., 3minute Student Oral Competition "Harlequin bug response to color: Its relation to attract-and-kill systems" Minneapolis, MN; November 16, 2015
Project Outcomes
Summary of Results of Project:
Currently, there is no available trap for harlequin bug, a damaging pest of brassica vegetables in the south. A trap could help alert growers to pest problems and could perhaps be used to attract and kill bugs for management purposes. In a series of lab choice tests we determined a color preference of harlequin bug for darker colors (black and green) over lighter colors. After reconfirming harlequin bug color preference in the field, we looked at trap shape to construct a trap-prototype (larger structures attracted more bugs). Since our goal is to build a trap-and-kill device, we assayed whether a commercially-available long-lasting deltamethrin insecticide treated mesh would provide adequate contact mortality of harlequin bug and how quickly it would do so (LT50 bioassay). The deltamethin-incorporated mesh was an effective killing agent for HB. The aggregation pheromone murgantiol was attaractive to HB, but in conjuncture with our field studies we also investigated the additive and/or synergistic effects of combining murgantiol with benzyl isothiocyanate (a host VOC). Isothiocyanate is the chemical group –N=C=S, formed by substituting the oxygen in the isocyanate group with a sulfur. Many natural isothiocyanates from plants are produced by enzymatic conversion of glucosinolates. Isothyocyanates (mustard oils) were highly and synergistically attractive to HB.
Our knowledge of sustainable agriculture were enhanced by this project as we familiarized ourselves with the current literature on IPM practices for harlequin bug. Also, conducting the experiments enhanced our knowledge of developing better IPM approaches for this pest.
The interaction of color with other factors such as trap shape, background contrasts, and olfactory stimuli are all important factors in developing an optimal trap design for monitoring and managing harlequin bug populations. We demonstrated that HB prefer dark colors and utilized dark green for field experiments. Dark green also caused less impact on lady beetle species. Olfactory attractants are also important for HB. Mixed-isomer syntheses of 10,11-epoxy-1-bisabolen-3-ol (i.e. murgantiol) are powerful attractants for harlequin bugs in the field, especially in combination with host plant volatiles such as mustard oil. Selection of optimal color stimuli would be expected to enhance female attraction to a trap baited with natural or crude mixtures of murgantiol isomers.