Final report for GS16-159
Project Information
The hot and humid climate of the Southeast is a challenge for the successful production of squash due to numerous disease and insect pressures. Economically important pests and diseases of squash in the Southeastern United States include downy mildew (Pseudoperonospora cubensis), powdery mildew (Podosphaera xanthii), cucurbit yellow vine disease (Serratia marcescens), squash bugs (Anasa tristis), vine borers (Melittia cucurbitae), and pickleworm (Diaphani nitidalis). The organic market growers in the Southeast face additional challenges due to the lack of efficacious pesticides. Plant host resistance, if available, is the first line of defense when using organic methods of production. In previous studies, C. moschata cultivars have shown better adaptation to Southeastern conditions, and in this study, promising cultivars of C. moshcata as well as selected cultivars from C. pepo and C. maxima were tested in field trials at the University of Georgia for resistance to diseases and pests, as well as fruit quality and yield.
Another option for management is the use of bio control organisms to target pathogens and insect pests.
The first objective of this project was to determine appropriate varieties of winter squash for organic production in the Southeast using multi-year replicated field trials in which varieties were evaluated for disease resistance, insect feeding preferences and fruit quality parameters.
The second objective examines the use of bio control to reduce the impacts of plant diseases. Cucurbit yellow vine disease is a recently introduced bacterial disease transmitted by squash bugs. The disease results in plant death. Other strains of S. marcescens are known to be effective bio control and plant growth promoting organisms in other systems. This research looked at whether non-pathogenic endophytic strains of S. marcescens have disease-controlling abilities by optimizing the use of the squash bug vector to introduce not only the pathogen but also the bio control agents into young squash plants. Plants were then evaluated in the field for the development of downy and powdery mildew, and cucurbit yellow vine disease.
- Evaluation of varieties of winter squash for disease resistance, yield, fruit quality, and insect pest feeding preference.
- Development of a squash bug inoculation method to introduce potential bio-control organisms into squash plants.
Research
Objective 1
Experimental design

Three years (2015 to 2017) of field trials were carried out at the University of Georgia Durham Horticulture Farm in Watkinsville, GA, on USDA certified organic acreage using organic approved practices. A winter cover crop of crimson clover and rye was incorporated into the soil approximately three to four weeks prior to planting each year. Experimental beds were arranged in a randomized complete block design with four replicates of each cultivar (see Figure 1). One replicate consisted of five plants with 0.61 m in-row spacing. In-row spacing between replicates was 1.22 m in years one and two, and 3.05 m in year three. Bed centers were spaced 1.83 m apart in year one and was increased to 3.05 m in years two and three to accommodate rampant growth of winter squash vines.
Nature Safe 10:2:8 was broadcast by hand at a rate of 1,221 kg/ha (4.8-6.8 kg/row). The fertilizer was tilled in with a rototiller along bed centers one to two days before planting. For irrigation, a single line of Chapin drip tape was placed down the center of each bed and plants were irrigated for an hour in the morning three days a week until plants began vining, after which plants were watered four days per week for 2 h in the morning. Ground cloth was placed in the centers between rows. Seeds were directly sown on the following dates in the three seasons: May 21, 2015, May 20, 2016, and May 10, 2017. Following germination, a layer of wheat straw was placed around plants to conserve moisture and suppress weeds.
Seven cultivars were tested in years one and two (Table 1), and nine in year three. After the first year, four cultivars that did not perform well and showed little promise for commercial production were dropped and four additional promising cultivars were added. Three C. moschata cultivars were grown each of the three years: Waltham Butternut, Seminole and Thai Kang Kob. In total 11 different cultivars of winter squash were grown over the course of the trial. Fruit were harvested over a span of one week, starting five days after the latest maturation time provided by seed suppliers.

Disease quantification
Downy and Powdery Mildew
To evaluate downy and powdery mildew severity, 15 fully expanded, mature leaves were randomly selected from the fourth through seventh leaves on vines. Disease severity was determined based upon approximate percentage of leaf area covered by lesions. A scale of 0-5 was used with 0 representing no lesions, 1: 1 to 10%, 2: 11 to 24%, 3: 25 to 49%, 4: 50% to 99%, and 5: 100%. Plants were scouted three times a week for the first signs and symptoms of downy mildew and powdery mildew. Thereafter plants were rated weekly, starting when disease severity reached sufficient levels.
Cucurbit Yellow Vine Disease (CYVD)
Plants were scouted three or more times weekly for CYVD symptoms such as wilting or yellowing of the leaves. The phloem tissues in the lower stems of plants that had wilted and yellowed were observed for the browning characteristic of CYVD in the lower stem. A 7-8 cm stem piece from just above the soil line was surface sterilized and two 50 mg pieces of vascular tissue were aseptically extracted and macerated in phosphate buffered saline (PBS), and the suspension was streaked onto duplicate Luria-Bertani (LB) agar plates. Following incubation for three days at room temperature, bacterial colonies with characteristic morphology of S. marcescens were transferred to fresh LB plates. Bacterial colonies were boiled and the identity of CYVD-strains was confirmed using PCR and strain-specific primers.
Insect pest quantification
Numbers of adult squash bugs, nymphs, and egg clutches on stems, leaves, and on the ground beneath the canopy were counted weekly in the inner 1.5 m x 0.6 m area in the center of each replicate. In years one and two, observations were made weekly from two weeks after germination until the fourth week. In year three, squash bug counts started two days after germination. During the first three weeks, counts were performed three times a week. During the fourth week, counts were made twice a week. Cultivars were rated for pickleworm damage by determining incidence of holes from pickleworm burrowing on all viable fruits.
Yield and fruit quality
Harvested fruit were cured in a warm dry area for at least two weeks before processing. Up to three fruits per replicate, depending on availability, were evaluated for Brix and dry matter content in year two and two fruits per replicate were evaluated in year three. For Brix ratings, slices were frozen overnight, crushed, and the sugar content of the resulting juice was measured with an Atago PAL-1portable refractometer. To measure dry matter content, 10 to 15 g fruit pieces were weighed, baked at 105ºC for 8 h, and weighed again. In year two and three, selected cultivars were used in blind taste tests. In year two, 25 subjects rated four different cultivars on overall taste on a 1 to 5 scale, with 5 being of highest quality. In year three, 20 subjects evaluated fruits using the same criteria. For yield, harvested fruit from each replicate were weighed and an average replicate fruit weight was calculated for each cultivar.
Statistical analyses
All statistical analyses were run in R Statistical Software (R Foundation for Statistical Computing, Vienna, Austria). Scale numbers were transformed to midpoints of percentage ranges for each scale point prior to analyzing downy and powdery mildew severity. All analyses were run using a one way analysis of variance (ANOVA) accounting for blocking and an α=0.05. Multiple comparisons tests were performed using Tukey’s HSD. No block effect was observed in the trials.
Objective 2
Comparison of squash bug nymphs and needle inoculation

Squash bug eggs were collected from squash plants at the University of Georgia Horticulture Research Farm (Watkinsville, GA). Eggs attached to collected leaves were gently scraped off into two Magenta boxes containing a paper towel dampened with sterilized water. The Magenta boxes were maintained on a 12 h day/12 h night cycle under fluorescent lights and at temperatures between 25ºC and 27ºC until hatching, usually within four to five days. First instars were fed surface-sterilized squash fruit and leaves for three to four days until they reached the second instar stage. Food was removed and nymphs were starved for 24 h. A 1 x 108 CFU/mL bacterial suspension of CYVD strain P1 was prepared with sterilized water as described above. The bacterial suspension was placed in a Parafilm -covered 60 x 15 mm petri dish placed in a Magenta box. A separate group of nymphs was fed a sterilized water control prepared in a petri dish in the same manner. After exposure to the bacterium or water for 24 h, three nymphs were placed on ‘Costata Romanesca’ zucchini (C. pepo, High Mowing Seeds, VT) seedlings at the one to two true leaf stage for 72 h (Figure 2). Eight plants were exposed to the squash bugs for each treatment.
Following feeding, nymphs were removed, surface sterilized in 70% Ethanol, washed in sterilized water, and macerated in tubes containing PBS buffer. The macerate was serially diluted and spread on LB plates in duplicate. Following incubation, bacterial colonies with characteristic morphology of S. marcescens were transferred to fresh LB plates. Single bacterial colonies were boiled in sterilized distilled water and identity of CYVD-strains was confirmed using PCR and strain-specific primers as previously described.
To compare with needle inoculation, eight plants each were needle inoculated with strain P1 or PBS. Inoculum was prepared from single colonies of S. marcescens strain P1 at a density of 1 x 108 CFU/mL in PBS. ‘Costata Romanesca’ zucchini (C. pepo, High Mowing Seeds, VT) seedlings at the expanded cotyledon stage were inoculated by pipetting a 20 µL drop of the bacterial suspension or PBS at the juncture of the apical meristem and cotyledons, pinching together and bending the cotyledons at a 45º angle from the stem, and stabbing 20 times through the cotyledons, the suspension, and the apical meristem with an inoculation fork consisting of five small gauge needles. The fork was sterilized between treatments.
Squash bug and needle inoculated plants were placed in the greenhouse for two days to recover from the inoculation before being planted in a certified organic plot at the University of Georgia Horticulture Research Farm in Watkinsville, GA. Plants were arranged in a completely randomized design with 46 cm in-row spacing and 3.0 m between rows. NatureSafe 10:2:8 fertilizer was incorporated at a rate of 6.8 kg per 55.7 m2 before planting. The plants were immediately covered with Agribon+ Ag-15 polypropylene floating row covers supported by 1.25 cm wide metal conduit hoops to exclude squash bugs. The row covers were sealed by covering the edges with soil and sand bags. The experiment was repeated three times in the summer of 2016 approximately two weeks apart.
Floating row covers were removed after six weeks, and stems and roots were removed from plants at eight to nine weeks after planting. Stem pieces from 1 cm below the cotyledons were sterilized and phloem tissue was examined for discoloration. Bacteria were extracted and identified as previously described.
Uptake of S. marcescens strain W11 by squash bug nymphs
Squash bug eggs were collected from a lab-maintained colony and were reared as previously described until they reached the second instar stage. Food was removed and nymphs were starved for 24 h. A 1 x 108 CFU/mL bacterial suspension in sterilized water of the endophytic, red pigmented S. marcescens strain W11 was prepared as previously described. Second instar squash bug nymphs that were hatched and reared in separate containers were fed either the bacterial suspension or sterilized water.
After exposure to the bacterium or water for 24 h, nymphs were exposed to surface sterilized squash fruit and leaves for three days. Single nymphs crushed in individual tubes. The macerate was serially diluted to and spread on LB plates in duplicate. Following incubation, pigmented S. marcescens colonies were enumerated and selected colonies were isolated onto fresh LB plates for identity confirmation as previously described.
Objective 1
Disease Quantification
Downy Mildew
In year one, the first symptoms of downy mildew were observed on July 10th. The disease progressed rapidly due to wet conditions and severity was determined at 14 days after first observed symptoms. Downy mildew severity was significantly higher (P=<0.001; Figure 2) in the highly susceptible ‘Waltham Butternut’ (x̅=84.59) and the powdery mildew resistant butternut cultivar ‘Metro P.M.R’ (x̅=71.50) when compared to all other cultivars.
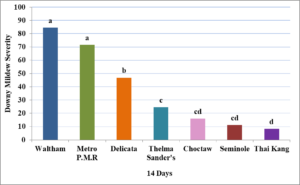
In year two, drier conditions led to a delay in development of downy mildew, with first symptoms appearing 15 days later than in year one on July 25th. Downy mildew severity was lower overall in year two for cultivars repeated from year one (Figure 3). All cultivars rated had significantly lower downy mildew severity at seven (P=0.01), 14 (P=<0.001), and 21 days (P=<0.001) than the highly susceptible ‘Waltham Butternut’.
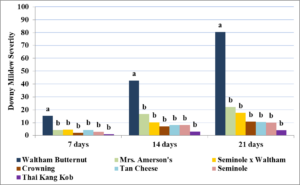
In year three, weather conditions were again wet and downy mildew was first observed earlier than in previous seasons, with initial symptoms seen on June 23rd. Disease severity increased slowly (Figure 4), leading to a delay in the first rating date. Only one replicate of ‘Thai Kang Kob’ and ‘North Georgia Candy Roaster’ survived to the first rating date. ‘Thai Kang Kob’ plants failed to establish and died early in the planting season while ‘North Georgia Candy Roaster’ succumbed to fusarium crown rot (Fusarium solani f.sp. cucurbitae). CYVD killed three or more plants in two or more replicates in ‘Waltham Butternut’ and ‘Delicata Zeppelin’ and few plants survived long enough to be affected by downy mildew.
Powdery Mildew
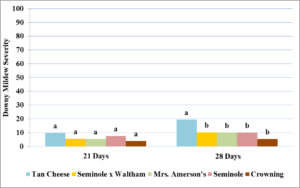
In year one, a rainy season inhibited development of powdery mildew, and no signs were seen on any cultivar throughout the season. In year two, powdery mildew was first observed on July 25th, with severity increasing on all cultivars over the 21 day rating period (Table 2). By the 14 day rating, ‘Waltham Butternut’ was defoliated by downy mildew and did not have enough mature foliage to rate for powdery mildew. No significant differences in powdery mildew severity were seen at 7 days (P= 0.08), 14 days (P=0.27), or 21 days (P=0.12) and severity was low in all cultivars.

In year three, signs of powdery mildew developed on June 23rd, with severity increasing slowly over the next 28 days. Ratings could not be carried out on most replicates of ‘Waltham Butternut’, ‘Delicata Zeppelin’, ‘North Georgia Candy Roaster’, or ‘Thai Kang Kob’ due to either poor establishment or death from other diseases, and these cultivars were not included in statistical analyses. A significant difference among cultivars in powdery mildew severity (Table 3) was seen at 21 days (P= 0.01) and 28 days (P=<0.001).

Cucurbit Yellow Vine Disease
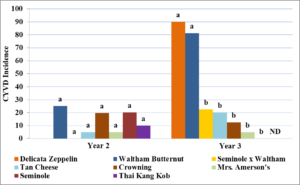
In year one squash bug numbers at the beginning of the season were very low and no plants developed symptoms associated with CYVD. In year two, squash bug populations increased over the season and at least some plants within each cultivar, except for ‘Waltham x Seminole F5’, developed CYVD with an average incidence within cultivars of 0 to 25% (Figure 5). No significant difference was seen among cultivars (P= 0.51). In year three, there was a significantly higher incidence of CYVD in ‘Delicata Zeppelin’ (see Figure 5) and ‘Waltham Butternut’ compared to other cultivars (P=<0.001).
Insect pest quantification
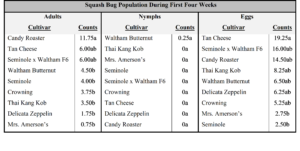
In year two, no difference in cultivar feeding preference was seen among adult, nymph, or squash bug eggs (Table 4).
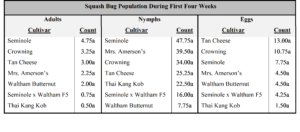
In year three, adults showed a preference for feeding on ‘North Georgia Candy Roaster’ over several other cultivars (P=<0.001; Table 5). Nymphs were only seen on ‘Waltham Butternut’ during the first four weeks, but not a significantly higher numbers (P=0.46). Significantly more eggs (P=0.01) were counted on ‘Tan Cheese’ (x̅=19.25) than ‘Mrs. Amerson’s’ (x̅=2.75) or ‘Seminole’ (x̅=2.50).
For pickleworm ratings, only a small sample size was available for ‘Crowning’, ‘Thai Kang Kob’, and ‘Tan Cheese’ in year two due to availability of few mature fruits. The least pickleworm damage was observed in the cultivars ‘Mrs. Amerson’s’, ‘Waltham Butternut’, and ‘Waltham x Seminole F5’. More fruits were evaluated for pickleworm damage in year three, and pickleworm was lower on average for all cultivars. The least number of damaged fruits was seen in ‘Mrs. Amerson’s’, ‘Seminole x Waltham F6’, and ‘Seminole’. Insufficient fruits were available of ‘Thai Kang Kob’, ‘Waltham Butternut’, ‘North Georgia Candy Roaster’, and ‘Delicata Zeppelin’ for pickleworm counts.
Yield and fruit quality
In year one, yield could not be assessed due to uncontrollable growth of the vines. Yield was high but most fruit rotted in the field due to the wet conditions. In year two, with the exception of ‘Waltham Butternut’, fruit did not reach full maturity before plants collapsed and no yield data was taken. Fruit of the appropriate size, weight, and color to indicate maturity was only available for testing for some cultivars. Number of fruit tested for each cultivar ranged from one to twelve depending on availability of fruit. Only ‘Waltham Butternut’ met the 10% standard for dry matter and was the only cultivar rated highly for taste (Table 7). In year three, yield was high at above 10 fruit/replicate for all cultivars except ‘Mrs. Amerson’s’ which only had an average of 7.5 fruit/replicate (Table 6). Average fruit weight was slightly below expected based upon cultivar descriptions provided by seed sources, with the exception of ‘Tan Cheese’.
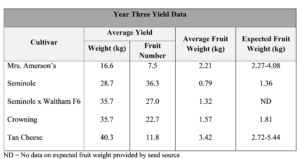
In year three, dry matter content was above 10% for all cultivars except ‘Tan Cheese’ (Table 7). Brix was above 10°Bx on average for ‘Seminole x Waltham F6’ and ‘Crowning’, and was very low in ‘Tan Cheese’ and ‘Seminole’.

Discussion
In this study, greater disease resistance appears to be present in C. moschata cultivars compared to C. pepo, with the exception of the C. moschata butternut cultivars ‘Waltham’ and ‘Metro P.M.R’. Several cultivars were found to be well-adapted to Southeastern conditions and offered improved control of downy mildew and CYVD compared to the commonly grown cultivar ‘Waltham Butternut’.
Downy mildew was the most damaging foliar disease evaluated during the three years of the study, and had completely or nearly defoliated susceptible cultivars such as ‘Waltham Butternut’, ‘Metro P.M.R’, and C. pepo ‘Delicata Zeppelin’ within three weeks after symptoms were first seen. Several of the C. moschata cultivars evaluated, such as ‘Thai Kang Kob’, ‘Crowning’, and ‘Seminole’, were developed in tropical or subtropical climates where downy and powdery mildew are present for much or all of the year, and appear to have the highest levels of resistance to these pathogens.
'Crowning (Chinese Tropical Pumpkin)', 'Seminole x Waltham' (South Anna Butternut), 'Seminole', 'Tan Cheese', 'Thai Kang Kob'. and 'Mrs. Amerson' showed moderate to good resistance to downy mildew, powdery mildew, and CYVD compared to the susceptible butternut 'Waltham'. No significant preference was seen among cultivars for insect feeding, although some more vigorous cultivars such as 'Tan Cheese' and 'North Georgia Candy Roaster' were slightly more preferred by squash bugs in the first few weeks after planting and may be good candidates for future trap cropping studies.
In addition to disease resistance, fruit quality and yield are important considerations for grower profitability. Among the tropical and heirloom cultivars, ‘Tan Cheese’, while not exceptional in taste, yields well for a large-fruited variety and is an exceptionally vigorous plant with good downy mildew resistance and is a good cultivar for pies. The smaller fruiting ‘Seminole’ yields well and has exceptional resistance to downy and powdery mildew. ‘Crowning’ yielded well with resistance to downy and powdery mildew, although this cultivar may be more susceptible to pickleworm damage. ‘Mrs. Amerson’s’ has excellent disease resistance, but the fruit and seed cavity are large and the flesh has a low dry matter content resulting in a watery texture. ‘Mrs. Amerson’s’ may be better suited for soups and sweetened pies. Low seed germination was also common with this cultivar so more seeds are needed to achieve a good stand.
The remaining cultivars were not as attractive for fresh market production but may be good for home consumption. ‘Choctaw Sweet Potato’ performed very well as far as yield and downy mildew resistance, but fruit size was too large to be readily marketable. ‘Thai Kang Kob’, though exceptionally disease resistant with flavorful and unusual fruits, was very low yielding, and may be better for breeding improved resistant lines than production. The C. pepo cultivars ‘Thelma Sander’s Sweet Potato’ and ‘Delicata Zeppelin’ did not perform well in this study but may produce well in drier years.
C. moschata winter squash cultivars appear to be better adapted to the semi-tropical conditions of the Southeast and several are good candidates for organic production in Georgia. The exception is ‘Waltham Butternut’, a C. moschata cultivar which may have some C. maxima background. ‘Waltham Butternut’ may perform well in a dry year in the Southeast when disease and pest pressures are lower, but this cultivar is highly susceptible to multiple diseases in a wet year.
In this study, early defoliation from downy mildew and death from CYVD resulted in only a few low quality fruits in year three. During selection for fruit quality, shorter time to maturity, and smaller plants, ‘Waltham Butternut’ and ‘Metro P.M.R.’ became more susceptible to downy mildew and perhaps also CYVD. To meet the demand for butternuts in the market, ‘Seminole x Waltham’ may be a good replacement. The cross was made in Virginia under organic growing conditions and this cultivar has the productivity, plant vigor, and disease resistance of ‘Seminole’, with equal to or better taste than ‘Waltham Butternut’. The fruits are somewhat larger than a traditional butternut, which may not appeal to all consumers, although further selection may reduce fruit size. Additional testing may be needed to more precisely determine levels of resistance to insect pests and the squash bug-vectored CYVD as well as provide further validation of resistance to downy and powdery mildew in cultivars only trialed for two of the three years.
Objective 2
Comparison of squash bug nymphs and needle inoculation
Several squash bug nymphs died during each inoculation attempt, although usually not until the final 24 h of the 72 h inoculation period. Following inoculation, 60.9 to 75% of nymphs were found to carry S. marcescens CYVD strains.
Upon removal of the row covers from the first set of inoculated plants, all plants died within a few days due to high heat and heavy squash bug feeding, and no data was collected. Most plants from the second and third set survived, although plant health was negatively affected by downy mildew and hot conditions. Fruit production initiated within a week after removal of row covers. Wilting began at eight weeks after planting in the majority of plants including those not carrying the pathogen.
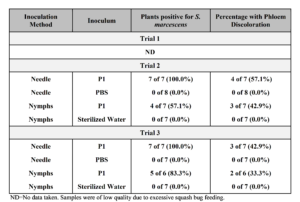
All of the plants needle-inoculated with P1 were found to carry CYVD strains of S. marcescens compared to 57.1% and 83.3% of plants inoculated by nymphs in the second and third trial, respectively (Table 8). Phloem browning was observed in 57.1% and 42.9% of the needle-inoculated plants in the second and third trial, respectively, and 42.9% and 33.3% of plants inoculated by nymphs, respectively. No correlation was seen between number of nymphs carrying the bacterium during feeding and development of CYVD symptoms.
Ability of squash bug nymphs to uptake endophytic S. marcescens strain W11
Pigmented S. marcescens colonies were recovered from 100% (n=14), 92% (n=25), and 72.7% (n=22) of nymphs in the three replications of the experiment at concentrations ranging from 1 x 103 CFU/mg to 4.81 x 105 CFU/mg tissue (Table 9). No contamination with pigmented S. marcescens was seen in nymphs fed only sterilized water. No difference in mortality was seen between bugs that took up W11 or only sterilized water.

Discussion
The use of second instar squash bug nymphs fed a bacterial solution instead of adult squash bugs fed squash cubes was an improved method of inoculating plants with CYVD strains of S. marcescens with a transmission efficiency of between 33 and 42%. The highest level reported in previous studies was 10% for nymphs and 17% for adults (Bruton et al. 2003; Pair et al. 2004; Wayadande et al. 2005). In this study, the level of transmission was similar to that achieved with needle inoculations although full symptom expression with either method was not achieved in the greenhouse or field.
Squash bugs readily acquired S. marcescens strain W11, a pigmented watermelon endophyte shown to be antagonistic against CYVD strains, with 72.7 to 100% of nymphs taking up the bacterium. Interestingly, this is higher than the 60.9 to 75% uptake observed with CYVD strain P1. The efficient uptake by squash bugs of strain W11, a potential biocontrol agent against CYVD strains, could be used to deliver W11 into plants or to suppress the CYVD bacterium within the squash bug. However, in preliminary greenhouse studies in which plants were fed upon by squash bugs carrying W11, nymphs failed to transmit the endophytic bacterium into plants. Other methods of introducing the bacterium into the plants may be more more efficient and effective.
Overall, inoculating with squash bug nymphs provides a new method of transmitting CYVD strains into plants. The nymphs are easier to rear and manipulate than adults, and the bacterial solution is an improvement over vacuum-infiltrated squash cubes. While the efficiency and disease expression could be improved, this method shows potential as a more reliable pathogenicity test which would facilitate future studies on the etiology and epidemiology of CYVD as well as introduction of biological control agents.
Educational & Outreach Activities
Participation summary:
Project Outcomes
This project built upon work in Virginia to show that winter squash can be successfully produced in the Southeast. There are several cultivars with good resistance to several of the more common diseases in the Southeast which also have good yield and fruit quality. These cultivars, such as 'Seminole' and 'Seminole x Waltham (South Anna Butternut)', would offer more consistent yields of high quality fruit in the Southeast than many of the more commonly used cultivars such as 'Waltham' and other commercial butternuts. These higher yields, coupled with greater disease resistance, mean a need for less plants and acreage to produce the same crop, improving both economic sustainability as well as the efficient use of sometimes scarce arable land. While these more durable cultivars are readily available, experimental testing and dissemination of the results to growers, as accomplished in the study, will allow growers to better assess their choices and risks. While impact cannot be estimated at this point, seed producers of some of the varieties tested in these studies have seen an increase in sales for those cultivars, showing interest among Southeastern growers.
This project provided me with the opportunity to hone my research skills and methodology and also allowed for me to interact with Georgia's as well as other Southeastern organic growers through travel funded by SARE. Through these interactions, I have been able to better gauge the interests and concerns of these growers, as well as learn more about how grower's select their varieties and produce their winter squash. During my research, I was able to coordinate and participate in most aspects of growing a crop from start to finish, from deciding when to mow and till cover crops through final cleanup of crop residue. From this experience, I now have a solid understanding of the challenges and opportunities associated with the sustainable production of vegetables in the southeast, as well as the requirements associated with organic certification.
Additional testing is needed to determine whether there are differences in susceptibility among squash cultivars to cucurbit yellow vine disease. Resistance would be a valuable contribution to disease prevention tools as methods of controlling the disease are currently limited. Examination of squash bug numbers in the field also showed a slight preference for feeding on some varieties over others. These more preferred varieties may be good candidates for future trap cropping studies. Most vegetable cultivars available for organic use have been tested in other parts of the country and organic growers in the southeast need better access to cultivars that perform well under the unique conditions of the southeast. Therefore, further testing of cultivars of various crops under organic conditions in the southeast would be very valuable, as well as work on producing organic seed for targeted crops under these conditions. Growers participatory trials on cultivar and seed selection would also be valuable.