Final report for GS18-182
Project Information
This project seeks to elucidate the multiple pathways by which wild bees may be impacted by neonicotinoid pesticide residues in soils that are used for nesting material. While significant progress has been made with regards to our understanding of the impact of neonicotinoid pesticides on honeybees and dietary exposure to residue in pollen and nectar, little is known about other exposure pathways which may exist for wild bees which use a variety of materials beyond pollen and nectar to complete their life cycles. Use of mud by mason bees (Osmia spp.) is thought to be a potential source of exposure to pesticide residue which has not been examined. Osmia species are an economically important group of pollinators which are commonly managed for orchard crops such as apple, cherry and blueberry (Bosch and Kemp 2000, Bosch et al. 2000, Bosch and Kemp 2002, Monzón et al. 2004, Sampson et al. 2004, Bosch et al. 2006). Imidacloprid residues can persist in soils for weeks or months after application, in concentrations ranging from 300 to 1200ppm within the immediate treatment areas (Knoepp et al. 2012, Fletcher et al. 2018). This project represents the first attempt to look at nesting materials as a potential exposure pathway for mason bees to neonicotinoid pesticides, utilizing the species Osmia lignaria Say.
Objective 1: To determine how adult female mason bee nest establishment is affected by imidacloprid soil drench treatments when treated mud is used for nesting.
Objective 2: To determine how offspring which develop within nests with treated soil are affected, in terms of survivorship and sublethal effects such as size, weight, and sex ratio.
Objective 3: To determine if female mason bees will detect or choose soils with lower residual concentrations of neonicotinoid.
Cooperators
Research
Objective 1
Eleven hundred Osmia lignaria (eastern subspecies) adult cocoons were obtained from a commercial source (Mason Bee Company, Deweyville, UT) in February 2019 and have been kept in cold storage at 4◦ Celsius until ready for emergence. Three hundred cocoons were emerged in the laboratory between March 11 and March 14, which included 120 females and 180 males. The females were individually marked and treated with contact exposure to soil for one hour in petri dishes at four different concentrations [< 1, 50, 390 and 780 parts per billion (ppb)], then released into six 6 x 6 x 2 m flight cages each enclosing 5 to 6 flowering rabbiteye blueberry shrubs at Durham horticulture research farm in Watkinsville, Georgia (Figure 1A). Each tent contained a Styrofoam nest block with 36 nesting holes filled with paper tubes (Figure 1B,C). Five females at each treatment level were released in each tent, for a total of 20 females per tent, and 30 untreated males were released per tent. Males were not treated to isolate the effects to females which contact the soil regularly for nest building. Bees were released in the flight cages on March 20, 2019 and behavior is being observed daily for 20 minutes per cage to ascertain effects on nesting behavior.
Cell production rate was measured by tracking dates of female nest initiation to completion. Nests were monitored daily to look for signs of females initiating nesting activity, including entering nests with pollen in the scopa and/or bringing mud to nest holes. A total of 20 hrs of formal observation was conducted over 14 days during the peak of blueberry bloom with each tent receiving 3 hrs and 20 mins of observation in 11 separate 20 min observation periods.
When nesting was completed, nest tubes were removed for examination. Reproductive output was measured by total number of cells produced per nest, numbers of pre-pupal cocoons per nest, weight of pre-pupal cocoons, and gender ratio of F1 generation adults. An otoscope was used to determine number of cells in each nest tube. Number of cells in combination with the amount of time from nest initiation to nest completion was used to assess nest cell production rate. During 2-5 June 2019, pre-pupal cocoons were removed from the nest tubes, number of pre-pupal cocoons per nest were counted, and cocoons were individually weighed to the nearest 0.0001 gm with a microbalance scale. F1 generation adults were cut from their cocoons in mid-April 2020 and sexed to determine gender ratio.
Another group of 180 adult male and female O. lignaria were exposed to treated soil in petri dishes at the same four concentrations (< 1, 50, 390, and 780 ppb), with three with replicates of each concentration at 20% soil moisture and three with replicates of each concentration at 40% soil moisture. Treated bees were released into small insect cages (60 x 30 x 30 cm) (Figure 1D). Seven females and eight males were released each cage, and mortality was being assessed daily for three days.
Objective 2:
To determine effects on larval development, we utilized specialized well plates which were developed for conducting experiments on grafted Osmia embryos in laboratory settings (Boyle and Pitts-Singer 2018). The well plates were printed on a MakerBot 3-D printer with PLA filament (resin based), fused deposition modeling, 0.3 mm filament thickness (each layer). The well plates contain 24 wells measuring 8 mm in diameter and 1.5 cm depth (Fig 2A). Each well was filled with 0.25 g of damp soil placed in the bottom of the well, using the same soil treatment levels (0, 50, 390, and 780 ppb). Nine well plates were utilized for the experiment, each well plate containing six replicates of each treatment level, for a total of 54 replicates of each treatment level. Recently capped reed nests containing embryonic O. lignaria were obtained from commercial distributors in Florida and Washington state. Reeds were mailed by the distributors in batches as soon as they were capped by females in the field (capping the end of the reed with mud is an indication the female has completed the nest). Immediately upon receipt, reeds were opened (Fig 2B), and individual larvae were randomly selected and removed along with their native pollen provision. The pollen provisions were weighed, and both the pollen provision and larvae were grafted into the wells containing 0.25g of treated soil. Pollen provisions from larvae not selected for the experiment were analyzed for background levels of imidacloprid contamination by liquid chromatography. No background levels were detected in any of the samples.
Once grafting was complete, well plates were placed in an incubator set to 27◦C. Well plates were observed daily for progress, and dates of transition to 2nd instar, 5th instar, start of silk spinning, and completed cocoon were logged for each individual. Signs of life for each larvae were checked daily, including movement, eating, observation of pollen moving through guts, or movement of hemolymph under skin. If no signs of life noted and common signs of death appeared (change in color, darkening, dull appearance and no response to stimulus), then the larvae was marked as dead. Completed cocoons were removed from well plate after three weeks and weighed in the pre-pupal stage.
Objective 3:
Two laboratory choice experiments were designed to examine whether females avoid or select soils containing imidacloprid residue. For the first experiment, mated and fed four-day-old females were placed in choice arenas in the following soil treatments: choice of 0 and 50, 0 and 390, 0 and 780, 50 and 390, 50 and 780, 390 and 780 ppb, with a full spectrum light source over the choice arena. Eighteen females per choice treatment were be observed in choice chambers for five minutes. Number of entries into each chamber and time spent in each choice chamber were logged.
For the second experiment, female and male O. lignaria were emerged in small test cages, allowed to mate and feed on a sugar/water/pollen mixture for five days. At the end of the fifth day post-emergence, a well plate containing pollen provisions was added to the test cages (Figure 3A). Well plates utilized for this experiment were developed for conducting experiments on grafted Osmia embryos in laboratory settings (Boyle and Pitts-Singer 2018). Honeybee pollen from a local beekeeper in Athens, Georgia was crushed and mixed with a 50:50 sugar and water solution to make pollen provisions. Mated 5-day-old females (5 per test cage) were given a choice of two soil treatments. One soil treatment contained imidacloprid concentration of either 100 (N = 6) or 1,000 (N = 6) ppb, the other was uncontaminated (0 ppb). Each soil treatment initially weighed 25g (Figure 3B). Paper nest tubes were provided in the test cages. Females laid eggs on the pollen provisions within the wells and utilized soil to cap the wells (Figure 3C), some females also utilized soil to cap ends of paper tubes (Figure 3D), although tubes were otherwise empty. After a period of two weeks of female activity, the two treatments of soil were air dried overnight and weighed. The dry weight of the remaining treated and untreated soil in each replicate was used to determine which soil was more heavily selected by the females.
Objective 1: Adult acute exposure to treated soil
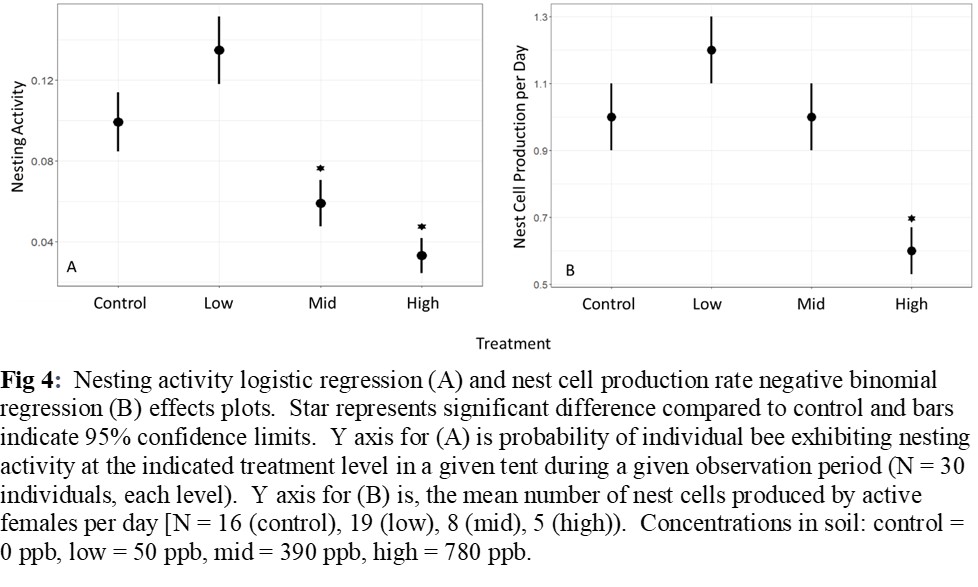
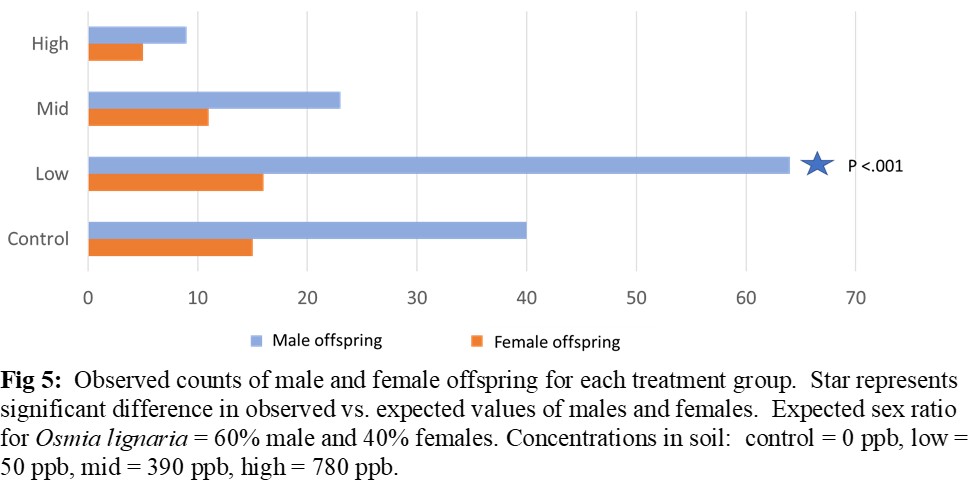
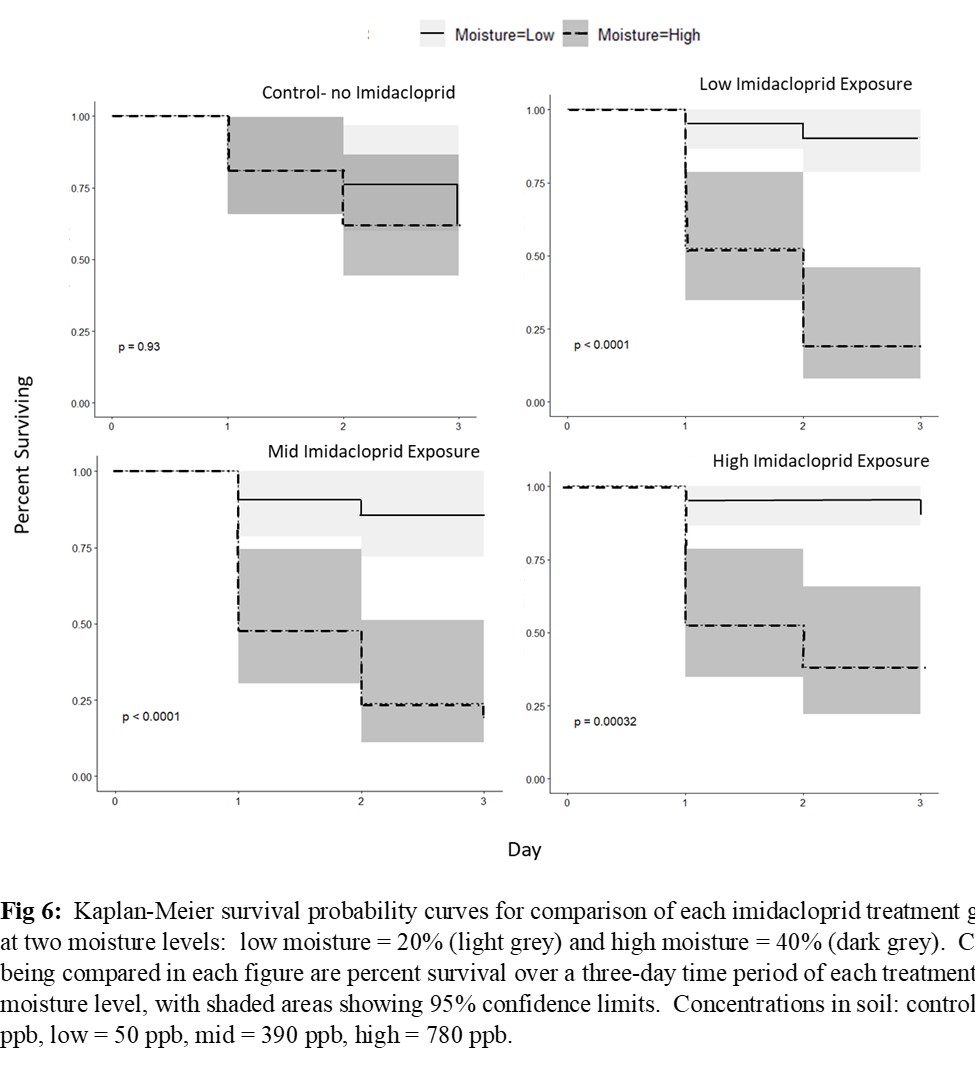
Nesting activity was reduced by 42% for females exposed at 390 ppb and by 66% for females exposed at 780 ppb (Figure 4A). Females treated at 780 ppb produced 40% fewer nest cells per day (Figure 4B). Sex ratios of F1 generation were skewed toward male in the 50 ppb treatment group with 50% fewer females (Figure 5). The number of cells and pre-pupae per nest, as well as the weight of pre-pupal cocoons did not vary among exposure levels. There were no mortality effects at 20% soil moisture for any level of imidacloprid, but at 40%, mortality of females was >50% at all levels of imidacloprid (Figure 6).
These results suggest that acute exposure to imidacloprid residue in soil can have negative impacts on soil-interacting bees, and the effects may be relative to the degree of soil moisture. The soil exposure pathway therefore deserves more attention in risk assessments and may need to be considered in best management practices for managing Osmia bees in orchard systems. Field-level risk assessments and mitigation tactics should consider the total area treated relative to the foraging area of bees in the landscape, the degree of soil moisture, whether the timing and interval of the treatment is concurrent with nesting bee activity, soil interacting bees are present within the immediate area, and soils are exposed, mulched, or covered with leaf litter after treatment, as mulch and leaf litter would limit contact by this group of bees.
Objective 2: Larval exposure to treated soil in nest material
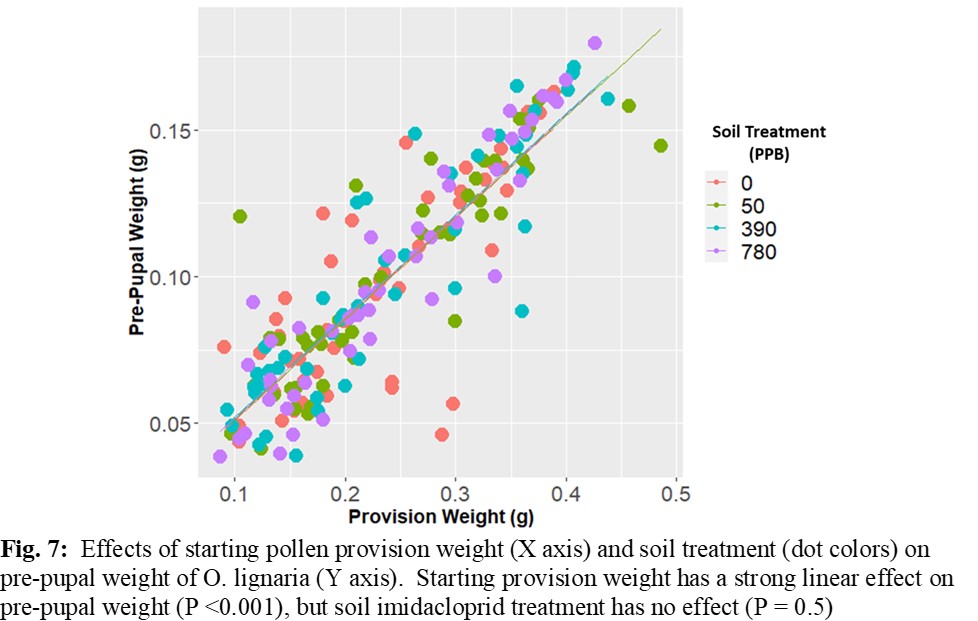
There were no differences in larval mortality among any of the treatment groups. All groups experienced between 15 and 20% mortality, which is likely representative of background mortality levels. There were likewise no differences in larval development time, with all groups completing the development cycle from second instar to cocoon in 18 to 24 days. Pre-pupal weight did not differ between treatment groups, and was highly related to starting provision size (Figure 7).
Results of this experiment indicate that soil residues of imidacloprid in nesting material up to concentrations of 780 ppb do not pose a significant threat to larval development or mortality. In past studies, Osmia larvae have shown some resistance to neonicotinoids in food provisions (Nicholls et al. 2017). The current study seems to further confirm that the larval stage has a level of resistance to the chemical. While this is an encouraging result, further testing of higher contamination levels and higher moisture levels in soil may be appropriate. The route of exposure for larvae in the current study would be a combination of contact exposure to the soil partition, and potentially some oral ingestion if residues migrated into the pollen provision, however we were not able to assess if such a migration occurred as the larvae consumed all or most of their pollen provisions. These results suggest that soil residues of imidacloprid used in nesting material have minimal impact on developing larvae.
Objective 3: Female soil choice
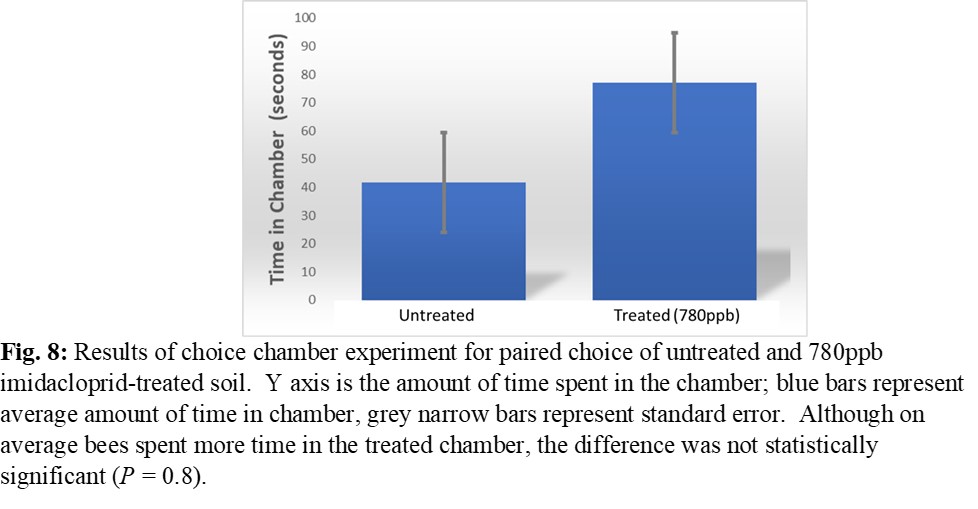
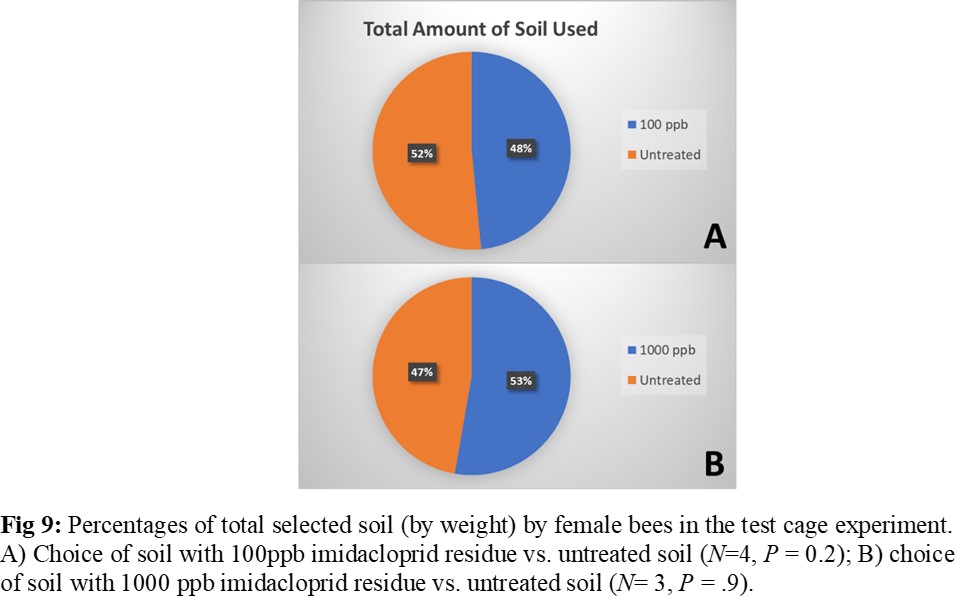
The choice arena trials did not reveal a clear discernment between imidacloprid treated and untreated soils by adult female bees in terms of number of entries and time spent in chamber for all choice combinations (Figure 8). Results of the test cage experiments indicated even selection of treated and untreated soil by female bees when selecting the soil for nesting material, even at levels as high as 1,000 ppb (Figure 9). Females in seven out of the 12 test cages utilized soil during the trial, including four of the test cages containing 100 ppb treatment soil and three of the test cages containing 1,000 ppb treatment soil. In the remaining five test cages, the females exhibited no nesting behavior of any kind, and thus those replicates were eliminated from analysis.
The results of these studies do not provide evidence of an avoidance response to treated soil nor a preference for untreated soil when selecting nest material. This demonstrates a potential for O. lignaria females to interact with treated soil while foraging for soil nesting resources, and that female O. lignaria may be prone to selecting treated soil for use in the nest. The results of the test cage experiment also reveal that female O. lignaria are capable of some degree of nesting plasticity, and can nest in a single-cell foundation as opposed to deep cavities. This experiment utilizing single-cell foundation well plates represents a new lab-rearing methodology, which has the potential to be utilized broadly as an experimental setup for answering many different questions about female choice, fecundity, longevity and nesting plasticity in Osmia and potentially in other cavity nesting bees. The results of the test cage experiment are preliminary due to small sample size, but the methods designed in this experiment can be further refined and replicated.
Bosch, J., and W. P. Kemp. 2000. Development and emergence of the orchard pollinator Osmia lignaria (Hymenoptera: Megachilidae). Environ. Entomol. 29: 8-13.
Bosch, J., and W. P. Kemp. 2002. Developing and establishing bee species as crop pollinators: The example of Osmia spp. (Hymenoptera: Megachilidae) and fruit trees. Bull. Entomol. Res. 92: 3-16.
Bosch, J., W. P. Kemp, and S. S. Peterson. 2000. Management of Osmia lignaria (Hymenoptera: Megachilidae) populations for almond pollination methods to advance bee emergence. Environ. Entomol 29: 874.
Bosch, J., W. P. Kemp, and G. E. Trostle. 2006. Bee population returns and cherry yields in an orchard pollinated with Osmia lignaria (Hymenoptera: Megachilidae). J. Econ. Entomol. 99: 408-413.
Boyle, N., and T. Pitts-Singer. 2018. In vitro larval rearing of Osmia lignaria (Hymenoptera: Megachilidae) for diet manipulation studies., Entomological Society of America, Vancouver, BC, Canada.
Fletcher, E., K. T. Morgan, J. A. Qureshi, J. A. Leiva, and P. Nkedi-Kizza. 2018. Imidacloprid soil movement under micro-sprinkler irrigation and soil-drench applications to control asian citrus psyllid (acp) and citrus leafminer (clm). PloS one 13: e0192668-e0192668.
Knoepp, J. D., J. M. Vose, J. L. Michael, and B. C. Reynolds. 2012. Imidacloprid movement in soils and impacts on soil microarthropods in southern appalachian eastern hemlock stands. J. Environ. Qual. 41: 469-478.
Monzón, V. H., J. Bosch, and J. Retana. 2004. Foraging behavior and pollinating effectiveness of Osmia cornuta (Hymenoptera: Megachilidae) and Apis mellifera (Hymenoptera: Apidae) on “comice” pear. Apidologie 35: 575-585.
Nicholls, E., R. Fowler, J. E. Niven, J. D. Gilbert, and D. Goulson. 2017. Larval exposure to field-realistic concentrations of clothianidin has no effect on development rate, over-winter survival or adult metabolic rate in a solitary bee, Osmia bicornis. PeerJ 5: e3417.
Sampson, B. J., S. J. Stringer, J. H. Cane, and J. M. Spiers. 2004. Screenhouse evaluations of a mason bee Osmia ribifloris (Hymenoptera: Megachilidae) as a pollinator for blueberries in the southeastern united states. Small Fruits Review 3: 381-392.
Educational & Outreach Activities
Participation summary:
Curriculum and workshop was developed to teach groups how to recognize, promote, protect and keep wild bees, including mason bees, leafcutter bees and bumble bees, and have conducted outreach to the following groups:
- Audubon Society Meeting, Clarke County, GA, March 2018: approximately 50 citizens
- Metro Atlanta Beekeepers Association, May 2018: approximately 150 beekeepers
- University of Georgia, Young Harris Beekeeping Institute, May 2018: approximately 100 beekeepers.
- Gwinnett County Beekeepers Association, August 2018: approximately 35 beekeepers
- Lee Arrendale State Prison, beekeeping class, September 2018: approximately 15 inmates/beekeepers
- Tri-county Beekeepers Association, October 2018: approximately 20 beekeepers
- Northeast Georgia Mountain Beekeepers Association, Jan 10, 2019: approximately 30 beekeepers
- Athens Academy High School Honors and AP Biology classes, April 2019: approximately 120 students
- Trees Atlanta, July 2019: approximately 15 citizens
Additional publications and presentations:
Publication in international peer-reviewed journal: Fortuin, C. C. et al. (2021). Acute contact with imidacloprid in soil affects the nesting and survival success of a solitary wild bee, Osmia lignaria (Hymenoptera: Megachilidae). Chemosphere, 264 (128572). https://doi.org/10.1016/j.chemosphere.2020.128572
Presentation at the annual Entomological Society of America meeting, Nov 2019, "Effects of imidacloprid soil drench applications on nesting Blue Orchard Mason Bees (Osmia lignaria).": https://esa.confex.com/esa/2019/meetingapp.cgi/Paper/145674
Article in the Orchard Bee Association quarterly newsletter, Orchard Buzz: “Assessing Pesticide Exposure Risks to Mason Bees (Osmia).”, Jan 2020.
Poster presentation at the annual Entomological Society of America meeting, Nov 2020: "Mason bees do not detect imidacloprid in soil: A new methodology for assessing Osmia lignaria (Hymenoptera: Megachilidae) soil choice in controlled laboratory environment."
Project Outcomes
In the United States, it is estimated that pollinators contribute to $14.2 – 23.8 billion in agricultural production (Chopra et al. 2015). Wild bees such as Osmia spp., Megachile spp. (leaf cutter bees) Nomia spp. (alkali bees), and Bombus spp. (bumble bees), contribute a substantive fraction of that total, an estimated $3.44 billion in the U.S. (Calderone 2012). This contribution is largely in support of minor fruit crops such as cherry, apple, blueberry, tomato and squash, and seed crops such as alfalfa. In addition, many systems have shown substantive improvement in pollination outcomes when wild bees are utilized in lieu of honeybees or as a supplement to honeybee pollination (Garibaldi et al. 2013).
The studies presented here are the first of their kind in terms of exploring the soil-based exposure pathway in wild solitary bees. Given the majority of wild bees in the U.S. utilize soil either for a nesting substrate or as nesting material, the soil exposure pathway has broad implications for wild pollinator management and protection. Wild bees such as Osmia becoming increasingly popular as managed pollinators in many systems, as there is growing concern that honeybees may not be able to continue to meet the increasing demands of agricultural pollination if these trends continue (Aizen and Harder 2009). These studies justify the need to include the soil pathway as a consideration in risk assessments for imidacloprid and other neonicotinoids which are applied directly to soil and will help contribute to effective integrated pest and pollinator management strategies in orchard systems. While more work is needed to determine the most effective risk management strategies, variables to consider would include the degree of soil moisture, whether the timing and interval of the treatment is concurrent with nesting bee activity, whether soil interacting bees are present within the immediate area, and whether soils are exposed, mulched, or covered with leaf litter after treatment, as mulch and leaf litter would limit contact by Osmia as well as other soil nesting species.
Aizen, M. A., and L. D. Harder. 2009. The global stock of domesticated honey bees is growing slower than agricultural demand for pollination. Curr. Biol. 19: 915-918.
Calderone, N. W. 2012. Insect pollinated crops, insect pollinators and us agriculture: Trend analysis of aggregate data for the period 1992–2009. PLOS ONE 7: e37235.
Chopra, S. S., B. R. Bakshi, and V. Khanna. 2015. Economic dependence of u.S. Industrial sectors on animal-mediated pollination service. Environ. Sci. Technol. 49: 14441-14451.
Garibaldi, L. A., I. Steffan-Dewenter, R. Winfree, M. A. Aizen, R. Bommarco, S. A. Cunningham, C. Kremen, L. G. Carvalheiro, L. D. Harder, O. Afik, I. Bartomeus, F. Benjamin, V. Boreux, D. Cariveau, N. P. Chacoff, J. H. Dudenhoffer, B. M. Freitas, J. Ghazoul, S. Greenleaf, J. Hipolito, A. Holzschuh, B. Howlett, R. Isaacs, S. K. Javorek, C. M. Kennedy, K. M. Krewenka, S. Krishnan, Y. Mandelik, M. M. Mayfield, I. Motzke, T. Munyuli, B. A. Nault, M. Otieno, J. Petersen, G. Pisanty, S. G. Potts, R. Rader, T. H. Ricketts, M. Rundlof, C. L. Seymour, C. Schuepp, H. Szentgyorgyi, H. Taki, T. Tscharntke, C. H. Vergara, B. F. Viana, T. C. Wanger, C. Westphal, N. Williams, and A. M. Klein. 2013. Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 339: 1608-1611.
The Osmia genus represents over 120 species in the U.S. and 30 species of bees east of the Mississippi, and several Osmia species are now commercially available and easy to manage using block nests. As many growers seek alternatives to honey bees as pollinators, Osmia and other managed wild bee groups are presenting opportunities for supplemental or primary pollination in crops such as blueberries and apples in the southern region. However, it is increasingly recognized that contact with nest substrates and nesting materials create a unique risk framework for wild bees which cannot be adequately addressed by honeybee studies. These alternative exposure pathways are not considered in the risk assessment framework for pesticide registration in the U.S., and constitute a substantial gap in our understanding of risk to wild bee groups. Through this project we gained an improved understanding of the risk pathways which need to be considered when integrating Osmia into a pollination strategy for orchard fruit crops.
Future research in the area of imidacloprid exposure to wild bees through soil should focus on replicated field-level studies. Results from laboratory or cage toxicity studies, such as the studies presented here, do not always translate to field-level risk. Thus, integrating laboratory and field studies can be important for risk assessments and development of field management strategies. As soil is not the only media used for nesting by wild bee species, exposure to other environmental media in such as detritus, leaves, and resin may be important for understanding risks to other groups of wild bees. In addition, multiple chemical stressors can be present in agroecosystems, understanding the effects of multiple chemical exposure to soil nesting bees and investigating potential synergistic effects will be an important topic for further research.