Final Report for GW16-068
Project Information
Alfalfa weevil is a pest of concern throughout the Western United States. Biological control of the weevil has been attempted through extensive releases of over 17 million parasitoid wasps of the alfalfa weevil in the 1950s and again in the 1980s. These parasitoid populations still persist in Wyoming, but alfalfa weevil outbreaks continue to be variable and severe. Conservation biological control, managing habitats to promote already existing natural enemies, is a promising avenue for improving control of alfalfa weevil. The overall goal of this project was to determine if alternative habitats at the landscape and local scale have the ability to enhance biological control of alfalfa weevil. At the local scale, a field study was conducted to compare visitation to annual and perennial flowering strips by parasitoids, other natural enemies, and pests. At the landscape scale, parasitism rates and in-field characteristics in producer fields across southeastern WY were measured and compared with landscape cover types. On-farm we found that annual flowering habitats supported both predators and a large number of herbivores, while perennial habitats contained increased predators but did not have increases in herbivores. Looking at the landscape scale, we found that the proportion of crops in the landscape was positively associated with pests and natural enemies at varying spatial scales. In particular, alfalfa weevil densities were positively associated with alfalfa at the 500m scale, and positively associated with natural and semi-natural habitats at the 2000m and 3000m scale. This information may be useful to producers interested in conserving natural enemies to reduce alfalfa pests.
Introduction
Intensification of cropland has lowered habitat diversity in agricultural landscapes leading to fewer alternative resources for natural enemies of pests. Alternative habitats near agricultural fields can provide overwintering habitat, refuge from disturbances, and additional food sources important for many natural enemies (1). Parasitoids are one type of natural enemy that kill their host in the process of parasitism, making them important biological control agents. Non-native parasitoid wasp species have been introduced for biological control of agricultural pests, including the alfalfa weevil (Hypera postica). Although there have been a number of studies that contribute to our knowledge of alfalfa weevil parasitism, it is still difficult to predict this parasitoid’s effectiveness for a given year or field. However, a still unexplored driver of parasitism is the effect of alternative habitats. Furthermore the effect of these alternative habitats can be assessed from the landscape perspective, where producers have no control over the habitat features surrounding their farm, or from an on-farm perspective, where producers can actively manage these habitats. The overall aim for identifying and understanding drivers of parasitism of alfalfa weevil is to improve biological control and reduce the severity of pest outbreaks.
The complexity of landscapes surrounding agricultural fields has been shown to influence the activity and abundance of natural enemies and pests in those fields (2). In particular, more complex landscapes (those with more natural and semi-natural habitat) have been shown to increase the activity of natural enemies in crop fields. In this study I aim to use landscape complexity and in-field characteristics to better predict the density of alfalfa pests and their natural enemies.
On the on-farm scale, incorporating flowering resources into agricultural landscapes can provide parasitoids access to alternative food sources (3) which promote parasitism through 1) increased parasitoid longevity and fecundity and 2) more time searching for hosts. Access to a carbohydrate food source has been shown to increase the longevity and eggs laid by the adult female of the most prevalent parasitoid in WY, Bathyplectes curculionis (4). We therefore hypothesize that providing parasitoids with floral resources near alfalfa crop fields can increase parasitism rates of alfalfa weevil. If this theory can be applied to pest control in intermountain west alfalfa fields, it would give producers another management tool for controlling a chronic and potentially devastating pest.
References:
- Landis, D. A., Wratten, S. D., & Gurr, G. M. “Habitat management to conserve natural enemies of Arthropod pests in agriculture.” Annual Review of Entomology, 45 (2000): 175–201.
- Chaplin-Kramer, R., O’Rourke, M.E., Blitzer, E.J., Kremen, C., 2011. A meta-analysis of crop pest and natural enemy response to landscape complexity. Ecol. Lett. 14, 922–932.
- The Xerces Society. Farming with Native Beneficial Insects: Ecological Pest Control Solutions. N.p.: Storey, 2014.
- Jacob, Helens S., & Edward W. Evans. “Influence of Carbohydrate Foods and Mating on Longevity of the Parasitoid Bathyplectes curculionis (Hymenoptera : Ichneumonidae).” 29.5 (2000): 1088–1095.
The aim of this study was to examine drivers of parasitism of alfalfa weevil by B. curculionis at a landscape and local scale. At the landscape scale we focus on comparing parasitism rates between multiple field sites with different landscape characteristics and addressing these objectives:
Objective 1: Determine if landscape composition (percentage of crop and non-crop habitat) affects parasitism rates, and densities of pests and natural enemies in producer fields.
Objective 2: Evaluate whether flowering weeds and/or aphid populations in alfalfa fields could be providing parasitoids with in-field resources.
At the local scale we focus on how parasitoids and natural enemies visit flowering alternative habitats, addressing the following objective:
Objective 3: Assess how abundance and community composition of arthropod functional groups (herbivores, predators, parasitoids, and pollinators) differ between different habitat types (annual flowers, perennial native flowers, alfalfa, control of fescue grass).
Cooperators
Research
Objectives 1 & 2
Sampling was conducted in 20 irrigated alfalfa fields in southeastern Wyoming, each separated by 4km to prevent spatial autocorrelation. At each field a sweep net was used to sample alfalfa weevil larvae, sweeping before the first cutting and insecticide applications. Samples were collected from all fields within 2 weeks to limit differences in parasitism due to timing and consisted of six, 50-sweep samples from each field, each separated by 10m. Weevils, common predators and pests were counted to determine densities of these groups in each field. Parasitism rate of alfalfa weevil was determined by rearing 50 weevil larvae from each field to adulthood over a 3 week period.
The following habitat characteristics were measured to account for variability between producer fields, each measured within a quadrat at the beginning of the 6 sweep transects: vegetation height, percent alfalfa cover, and alfalfa damage (scored on a scale from 0 to 5, with 0 indicating no damage and 5 complete defoliation). In-field floral resources (the number of open blooms and the plant species in bloom) for each field were recorded along two, 75m transects, each 50m apart.
Associations between landscape composition and parasitism rates, density of alfalfa weevil, predators, and pests, were determined using the National Agriculture Statistics Service cropland data layer in ArcMap. Cover types surrounding field sites were grouped into three land cover categories which included natural/semi-natural habitat, alfalfa, and other crops. The percentage of the landscape in these cover types was assessed at 4 diameters around field sites: 500m, 1000m, 2000m, and 3000m. Due to strong correlations between land cover types, a principal components analysis was used for these variables to create principal components that were independent of one another (1). Stepwise multiple linear regressions were run in JMP 12 (SAS Institute Inc., 2015) to characterize how landscape factors and field characteristics were related to the abundance of aphids, H. postica larvae, Coccinellidae, and Nabidae at each landscape scale.
Objective 3
In 2014, a field experiment at the Sustainable Agriculture Research and Extension Center (SAREC) near Lingle, WY was established to test whether floral resources are attractive to insect natural enemies and pests in our region. Specifically, floral strips (3 ft x 25 ft) were planted adjacent to 25 ft x 25 ft alfalfa plots (as shown in Figure 1). Treatments were a 1) a regionally-adapted perennial flower mix of 9 species, 2) an annual flower mix of 9 commonly used annuals for supporting natural enemies, and 3) a fescue grass control. Each treatment was replicated 5 times using a randomized block design to account for variability in soil and irrigation direction at the site. A 25ft grass buffer surrounds each plot.
Arthropod communities in each habitat type were sampled every 2 weeks during the growing season at the field experiment at SAREC, for a total of 7 sampling dates. Flower strips, control strips and alfalfa plots were sampled using a reversed leaf blower with fine mesh bag secured to the intake to prevent damaging fragile flower habitats (2, Figure 2). Sampling was conducted for 30 seconds, focusing on flowering parts of the plant when available. Samples were transported back to the lab, where bag contents will be put in ethanol until processing could occur. In addition to vaccum sampling, the vegetation height and the number of open blooms in each habitat type were also quantified. In the early season before the first cutting, alfalfa plots were sampled for alfalfa weevil larvae using the same methods as in producer fields, scaled down to account for our smaller plot sizes.
Vacuum samples were sorted for predators, parasitoids, and pests. All Hemipterans and Hymenopterans were identified to family and classified as herbivores, predators, parasitoids, or other. In addition, I counted other groups of interest including alfalfa weevil (Hypera postica), flea beetles (Alticini), big-eyed bugs (Geocoris), fleahoppers (Halticus), Orthopterans, lady beetles (Coccinellidae), green lacewings (Chrysopidae), and spiders (Araneae) because these herbivores and natural enemies are commonly found in Wyoming alfalfa fields. To analyze vacuum data a repeated measures mixed model (PROCMIXED, SAS Institute Inc., 1996) was used, with block as a random factor, treatment or habitat as fixed factors, and date as the repeated factor. Tukey’s tests were used to separate means for significant factors and interactions (p-value less than 0.05).
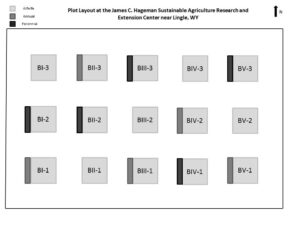 Figure 1. Layout of Plots at the SAREC field site. The area between and around plots was a buffer zone planted to tall fescue. Sprinkler irrigation runs west to east.
Figure 1. Layout of Plots at the SAREC field site. The area between and around plots was a buffer zone planted to tall fescue. Sprinkler irrigation runs west to east.
 Figure 2. A leaf blower put in reverse with a mesh bag secured over the intake to collect insects. Here used to vacuum sample an alfalfa plot.
Figure 2. A leaf blower put in reverse with a mesh bag secured over the intake to collect insects. Here used to vacuum sample an alfalfa plot.
Objective 1
Landscape composition was associated with the densities of alfalfa weevil, aphids, nabids, and coccinellids, but not with parasitism rates. The most predictive scale varied depending on insect. Alfalfa weevil were positively associated with the percent of alfalfa crops in the landscape at the 500m scale, and positively associated with natural and semi-natural habitat at the 2000m and 3000m scales. Aphids were positively associated with the amount of other crops in the landscape at the 3000m scale, and negatively associated with natural and semi-natural habitat. Nabids were also positively associated with other crops and negatively associated with natural and semi-natural habitat at the 3000m scale. Lastly, coccinelids were positively associated with the amount of alfalfa in the landscape at the 1000m scale. In general, our findings on the most predictive scales for determining abundance of different arthropod groups were aligned with previous research. Previous work (1) has found that generalist pests and enemies are better predicted at larger spatial scales, while specialists are better predicted at smaller scales.
Objective 2
Parasitism rates were not associated with in-field blooming weeds or with aphid densities as we had hypothesized. The only significant predictor of parasitism rates in alfalfa fields was host density (p = 0.0113, Figure 3). Parasitism rates were higher in fields with lower alfalfa weevil densities. Rand (2) also found that host density was a significant driver of parasitism rates, although the relationship differed over the two years of her study. In year 1, percent parasitism was positively correlated with alfalfa weevil density and in year 2, percent parasitism was negatively correlated with alfalfa weevil density.
Objective 3
In general, functional group habitat preference changed throughout the growing season (Figure 4). For example, in the early season, herbivores showed no preference between annual, perennial, or control-fescue habitats, while predators were most abundant in the control-fescue habitats. The lack of response by herbivores to a particular habitat in the early season may be because all habitats were relatively vegetative (with few open blooms) and several of the herbivores collected in this study fed on the flowering or seed structures of plants. Predators and parasitoids, however, were more abundant in the control-fescue, which is supported by previous work finding that grassy field margins may be important for natural enemies in the early season (3,4). In the late season, both herbivores and predators were most abundant in the annual habitats. This corresponds to when the annual habitats had the greatest amount of open blooms (Figure 5). Also shown in Figure 5, is that perennial habitats had a longer bloom period across the season because these plants were established in 2014. The annual flower species, however, were planted after the last frost date for Lingle, WY on June 1, 2015, causing a lag in bloom for the annual habitats. This lag can also be seen in Figure 6, where the perennial habitats are further along than the annuals on the same date. We think that this lag in bloom would be an important consideration for producers interested in supporting natural enemies of early season pests, like the alfalfa weevil.
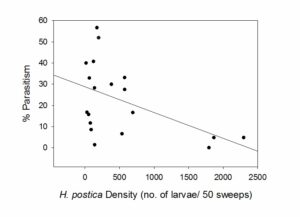 Figure 3. Relationship between alfalfa weevil density and parasitism by Bathyplectes curculionis.
Figure 3. Relationship between alfalfa weevil density and parasitism by Bathyplectes curculionis. 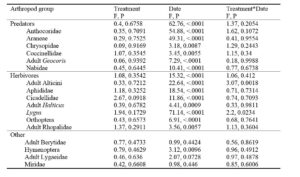 Figure 4. Partial ANOVA table for analyzed arthropod groups in alfalfa. F and P values are listed for treatment, date, and treatment*date interactions for the 2015 growing season.
Figure 4. Partial ANOVA table for analyzed arthropod groups in alfalfa. F and P values are listed for treatment, date, and treatment*date interactions for the 2015 growing season.  Figure 5. 2015 Bloom calendar for annual and perennial flower species, where light green represent the species in bloom and dark green represent peak bloom weeks of highest open bloom abundance. Note that coreopsis had two peak blooms of equal bloom abundance
Figure 5. 2015 Bloom calendar for annual and perennial flower species, where light green represent the species in bloom and dark green represent peak bloom weeks of highest open bloom abundance. Note that coreopsis had two peak blooms of equal bloom abundance  Figure 6. Photos of annual (a.) and perennial (b.) floral habitats on July 9, 2015.
Figure 6. Photos of annual (a.) and perennial (b.) floral habitats on July 9, 2015.
References:
- Chaplin-Kramer, R., O’Rourke, M.E., Blitzer, E.J., Kremen, C., 2011. A meta-analysis of crop pest and natural enemy response to landscape complexity. Ecol. Lett. 14, 922–932.
- Rand, T.A., 2013. Host density drives spatial variation in parasitism of the alfalfa weevil , Hypera postica , across dryland and irrigated alfalfa cropping systems. Environ. Entomol. 42, 116–122.
- Balzan, M. V., Moonen, A.-C., 2014. Field margin vegetation enhances biological control and crop damage suppression from multiple pests in organic tomato fields. Entomol. Exp. Appl. 150, 45–65.
- van Rijn, P., Alebeek, V.F., den Belder, E., Wäckers, F., Buurma, J., Willemse, J., van Gurp, H., 2008. Functional agro biodiversity in Dutch arable farming: results of a three year pilot. IOBC / WPRS Bull. 34, 125–128.
Research outcomes
Education and Outreach
Participation summary:
Multiple strategies were used for outreach to alfalfa growers in Wyoming and the broader community, including fact sheets, field days, presentations, blog posts, and Instagram.
Fact Sheets
Jones, A., M. Pellissier, R. Jabbour. 2015. Insects in Wyoming Alfalfa. University of Wyoming Extension B-1275. (http://www.wyoextension.org/agpubs/pubs/B-1275.pdf).
Pellissier, M., R. Jabbour. 2016. Flowering Plants to Support Beneficial Insects. (https://insectag.files.wordpress.com/2016/09/flower-fact-sheet3.pdf).
Pellissier, M., A. Jones, R. Jabbour. 2016. Aphids in Alfalfa. University of Wyoming Extension B-1287. (http://www.wyoextension.org/agpubs/pubs/B-1287.pdf).
Social Media
Pellissier, M. 2015. Blog Post: What about Aphids in Alfalfa? (https://insectag.com/2015/07/21/what-about-aphids-in-alfalfa-2/).
Pellissier, M. 2016. Blog Post: Flower Power. (https://insectag.com/2016/01/21/flower-power/).
Pellissier, M. 2016. Blog Post: Alfalfa Weevil Infestations from a Landscape Perspective. (https://insectag.com/2016/07/27/alfalfa-weevil-infestations-from-a-landscape-perspective/).
UWYO Insect Agroecology, @weevilwarriors. Instagram account with 218 followers, where lab and research updates are shared with a diverse audience. (https://www.instagram.com/weevilwarriors/).
Presentations
Benander, M. R. Jabbour. 2015. Landscape and local factors affecting parasitism of alfalfa weevil. Department of Plant Sciences Seminar. (presentation)
Benander, M., R. Jabbour. 2015. Multifunctional flowering habitats – insights from field research. Sheridan Agricultural Experiment Station Field Day. (presentation)
Benander, M., R. Jabbour. 2015. Local and landscape effects on parasitism of alfalfa weevil by a specialist parasitoid wasp. Ecological Society of America Conference. (presentation)
Benander, M., R. Jabbour. 2015. Disturbance and Succession in Agroecosystems. Introduction to Agroecology Class. (presentation)
Benander, M. R. Jabbour. 2015. Natural enemy and herbivore response to the inclusion of floral resources in intermountain west alfalfa systems. Entomological Society of America Conference. (presentation)
Jabbour, R., M. Pellissier. 2016. Pest Management in Organic Systems. High Plains Organic Farming Conference. (presentation)
Pellissier, M., R. Jabbour. 2016. Alfalfa Weevil Management. Wyoming Weed and Pest Workshop (presentation)
Publications
Benander, M., R. Jabbour. 2015. Use of Perennial and Annual Flowers to Attract Beneficial Insects to Alfalfa. Wyoming Agricultural Experiment Station Field Days Bulletin. (http://www.uwyo.edu/uwexpstn/_files/docs/2015-field-days-bulletin.pdf).
Pellissier, M., R. Jabbour. 2016. Use of Perennial and Annual Flowers to Attract Beneficial Insects to Alfalfa. Wyoming Agricultural Experiment Station Field Days Bulletin. (http://www.uwyo.edu/uwexpstn/_files/docs/2016-field-days-bulletin.pdf).