Final report for LNE22-450R
Project Information
Due to a short growing season, late-season root crops (e.g., potatoes and sweet potatoes) are particularly valuable in diversified farming systems in the northeastern United States. Reduced yield and quality resulting from infestations by common insect pests, including Colorado potato beetle (CPB) and wireworms, represent a serious risk for many farmers who rely on late-season revenue from these crops. Sustainable, low-impact options for managing these pests on both organic and conventional farms have been limited, and growers have often relied heavily on chemical controls. High levels of chemical pesticide use for CPB and wireworm management have increased the risk of pesticide resistance. The development and assessment of novel pest management strategies conducted in this study provided valuable information to expand the IPM toolbox available to Northeastern potato growers.
This project investigated two environmentally compatible approaches to reduce wireworm and CPB infestations in root crop systems. The project originated through a participatory action research (PAR) process that engaged approximately 30 growers across Vermont and New York. During PAR meetings and through distributed surveys, growers—particularly organic producers—expressed strong interest in new management options that could reduce dependence on a limited set of effective chemical controls (e.g., Entrust, neonicotinoids). In response to this need, the project developed and evaluated benchtop and on-farm applications of entomopathogenic fungi for managing wireworms and CPB. In addition, the project evaluated the efficacy of RNA interference (RNAi) products as a potential IPM tool for conventional growers.
The entomopathogenic fungi Beauveria bassiana and Metarhizium brunneum were the primary species cultured and evaluated during field and laboratory trials. These species were selected based on their relative efficacy and suitability for culturing. Benchtop formulations were produced using commonly available grain substrates (e.g., millet and barley), which allowed for comparison of cost-effective and efficient substrates relevant to research and on-farm contexts.
Laboratory experiments, field trials, and participatory evaluations were conducted to test the following hypotheses:
- grain substrates inoculated with entomopathogenic fungi reduced wireworm pressure when applied as soil treatments;
- foliar applications of entomopathogenic fungi and RNAi reduced CPB pressure in potato systems; and
- farmer training in fungal culturing principles expanded IPM knowledge and reduced reliance on chemical control strategies.
Across four years of trials, a clearer picture emerged regarding the performance, limitations, and practical implications of entomopathogenic fungi (EPF), RNA interference (RNAi) technologies, and integrated management approaches for CPB and wireworms in diversified Northeastern farms. Although several field seasons were affected by flooding, low pest pressure, and disease outbreaks, combined evidence from greenhouse, research farm, and on-farm trials supported several actionable conclusions for growers and informed future research directions.
Practical recommendations for farmers
Based on data collected during this project, the following recommendations were identified with reasonable confidence:
EPF (Beauveria, Metarhizium)
- EPF were effective as components of IPM programs, particularly for growers seeking reduced chemical inputs.
- Applications performed best under conditions of moderate humidity and consistent CPB pressure.
- EPF were not consistently reliable as stand-alone treatments in years with high pest variability.
RNAi (emerging technology)
- RNAi demonstrated strong potential for protecting foliage and maintaining yield.
- RNAi performed most effectively when used in combination with EPF.
- RNAi represented a promising option for growers seeking alternatives to spinosad-based controls.
Wireworm management
- EPF treatments did not provide consistent wireworm control under field conditions.
- Monitoring species composition and seasonal activity was critical for interpreting treatment outcomes.
- Future management strategies were likely to require species-specific or microbiome-informed approaches.
Overall conclusion
Despite adversity across multiple growing seasons, results from greenhouse and field trials demonstrated that biologically based CPB management was feasible, particularly when RNAi and EPF were combined. Although wireworm control remained unresolved, expanded surveys and laboratory assays provided essential ecological and species-level context to guide subsequent fungal efficacy trials.
Lab and field trials will be conducted to assess the efficacy of both cultured entomopathogenic fungi and RNAi technologies for the control of wireworms and Colorado potato beetle in root crops. This study will aid in the development of the essential knowledge and skills needed for the effective low-tech culturing of entomopathogenic fungi and subsequent field applications to control these pests. Data generated from RNAi trials will directly inform conventional potato growers of the best practices for utilizing RNAi technologies for CPB control. Both entomopathogenic fungi and RNAi applications will provide growers with innovative strategies to expand their IPM toolbox.
Cooperators
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
Research
The primary experiments conducted included:
- Laboratory trials assessing the most efficacious and cost-effective substrates for culturing Beauveria bassiana and Metarhizium brunneum.
- Field trials evaluating the efficacy of granular formulations of B. bassiana and M. brunneum for the control of Colorado potato beetle (CPB) and wireworms in potato and sweet potato crops, respectively.
- Development and evaluation of low-tech protocols for culturing B. bassiana and M. brunneum for research and demonstration purposes.
- Evaluation of the efficacy of commercially available RNAi products for the control of CPB on conventionally managed Northeastern diversified farms.
Hypothesis I
Laboratory trials (Table 1) included formulation of Beauveria bassiana and Metarhizium brunneum on different carriers (couscous, millet, and barley).
The formulations were evaluated based on substrate availability, cost, and efficacy for potential use in root crop pest management. Application of mycotized grains in furrows has been previously reported to be as effective as chemical treatments for the control of soilborne insects.
To produce mycotized grains, pure inocula of both fungi were obtained by culturing commercial products of B. bassiana (BotaniGard® ES, GHA; BioWorks, Victor, NY, USA) and M. brunneum (Met52® EC and Met52® G; Novozymes Biologicals, Franklinton, NC, USA) on Potato Dextrose Agar (PDA) and Sabouraud Dextrose Agar (SDA), respectively. Fungal formulations were prepared following the protocol of Kim et al. (2014) by inoculating cooked couscous, millet (Panicum miliaceum L.), and barley (Hordeum vulgare L.).
Quality control was assessed by measuring conidial germination and conidial density. Laboratory bioassays were conducted on wireworm larvae and Colorado potato beetle (CPB) to assess treatment efficacy. Each bioassay included ten replicates per treatment and control (Table 1). Approximately 2 g of mycotized grain formulation was applied to each treatment. All treatments were inspected daily to record mortality rates. The most effective fungal formulations were selected for subsequent field trials (Table 1).
To prepare fungal suspensions, label instructions were followed for B. bassiana. Because Met52 was no longer commercially available, suspensions were prepared at a concentration of 10⁸ conidia mL⁻¹.
Table 1. Treatments, substrates, target organisms, and application types.
| Trail | Treatment | Target Pese | Application Method |
| Lab | Control(no treatment) | Wireworms and CBP | NA |
| Couscous (C) | Wireworms and CBP | Contact exposure | |
| Millet (M) | Wireworms and CBP | Contact exposure | |
| Barley (B) | Wireworms and CBP | Contact exposure | |
| Beauveria bassiana GHA couscous (BBC) | Wireworms and CBP | Contact exposure | |
| Beauveria bassiana GHA millet (BBM) | Wireworms and CBP | Contact exposure | |
| Beauveria bassiana GHA barley (BBB) | Wireworms and CBP | Contact exposure | |
| Metarhizium brunneum F52 couscous (MMC) | Wireworms and CBP | Contact exposure | |
| Metarhizium brunneum F52 millet (MMM) | Wireworms and CBP | Contact exposure | |
| Metarhizium brunneum F52 barley (MMB) | Wireworms and CBP | Contact exposure | |
| Field trials | Control (grain) | Wireworms and CBP | In furrow |
| Most effective mycotized grain (B. bassiana) | Wireworms and CBP | In furrow | |
| Most effective mycotized grain (M. burnneum) | Wireworms and CBP | In furrow | |
| Control (Water) | CPB | Foliar | |
| B. bassiana GHA suspension | CPB | Foliar | |
| M. brunneum F52 suspension | CPB | Foliar |
Methods:
Field experiments were established using a randomized complete block design at three commercial vegetable farms and two UVM-associated research farms: the University of Vermont Horticultural Research and Education Center (South Burlington, VT) and Borderview Research Farm (Alburgh, VT). At each site, 15 sweet potato plots and 30 potato plots were established, with five replicate plots per treatment and control (Table 1). The same treatments and experimental design were applied across multiple field seasons. Individual plot size was 10 bd ft, with 10 bd ft buffer zones between plots.
To target wireworms, fungal treatments were applied in-furrow at planting at a rate of 5 g per plot. Fungal suspensions were applied as foliar sprays to manage CPB. All treatments were applied three times during the growing season at three-week intervals.
Data Collection:
For all field trials (fungal and RNAi applications), data were collected on above- and below-ground feeding damage and marketable yield. Wireworm damage was assessed by harvesting 20 sweet potatoes per plot and recording the number of wireworm galleries per tuber and marketable yield (lbs). Wireworm density was estimated using bait traps buried at 8–15 cm depth in each plot. Bait traps were prepared by soaking wheat and barley seeds in water for 24 hours to induce sprouting. Captured wireworms were counted and identified to species.
To assess CPB damage, all potatoes from each plot were harvested and weighed as total kilograms per meter of row. CPB populations were estimated by collecting ten stems (20–25 cm length) per plot at three sampling dates following treatment applications, at approximately four-week intervals. All live larvae were counted and categorized as early (first and second instars) or late (third and fourth instars).
Hypothesis II
Methods:
A low-tech protocol for fungal formulation was developed and demonstrated, including: (1) preparation of a sterile workbench; (2) media preparation by filtering cooked potato through cheesecloth and adding agar, dextrose, and water; (3) sterilization of media and grains at 121 °C using a pressure cooker; (4) inoculation of sterilized grains with fungal cultures; and (5) maintenance of cultures under controlled conditions.
Workshops were conducted during the project period to facilitate farmer-to-farmer learning and demonstrate fungal culturing principles and applications.
Data Collection.
Questionnaires were designed and distributed to participating farmers to evaluate the feasibility and clarity of the protocol. Feedback regarding challenges and suggestions was collected and used to refine educational materials. Fungal formulations produced during demonstrations were assessed for contamination and culture purity.
Data Analysis and Presentation of Results:
No statistical analyses were required for this hypothesis.
Hypothesis III
Methods:
At the outset of the project, a commercially available RNA interference (RNAi) product for CPB control had recently entered the U.S. market. During the project period, multiple attempts were made to obtain this product through direct communication with the manufacturer, including in-person meetings, virtual meetings, and follow-up correspondence. Despite these efforts, the product was not made available for research use, and purchase through standard commercial channels was not possible. Repeated inquiries resulted in referrals back to the same company representatives without resolution.
To ensure completion of the project objectives, an alternative RNAi product targeting CPB was sourced from a startup company based in Canada that offered a functionally similar RNAi formulation. This product was used as a replacement for the originally anticipated U.S.-based commercial RNAi product in laboratory and field evaluations. Product quality and quantity were assessed using agarose gel electrophoresis and NanoDrop spectrophotometry. Field trials were conducted using a randomized complete block design at two UVM-associated research farms: the University of Vermont Horticultural Research and Education Center (South Burlington, VT) and Borderview Research Farm (Alburgh, VT).
Individual plot size was 10 bd ft, with 10 bd ft buffer zones between plots. The RNAi product, provided in liquid form, was applied at labeled or experimentally determined rates during the project period. Trials were conducted across multiple growing seasons.
Data Collection:
CPB population density and plant damage were measured using the same methods described under Hypothesis I.
Data Analysis and Presentation of Results:
Data were analyzed using the same analytical approaches described under Hypothesis I, and treatment effects were compared between RNAi-treated and control plots.
2025 Progress Report
Hypothesis III: RNAi products will significantly reduce pest pressure associated with CPB infestations in conventionally managed potato crops.
Because several prior seasons were affected by unusually low pest pressure, persistent humidity, fungal disease, and storm events, we requested an extension to repeat the field trials under more favorable conditions. In the extension year, we re-established the complete research trial at the UVM research farm while also initiating complementary trials at two commercial organic farms to strengthen the robustness of the dataset.
Commercial Farm Trial
Our first on-farm trial, originally from Burlington, relocated their potato plantings to a temporary site in Hinesburg due to recurring early flooding in recent years. As a result, CPB pressure remained low throughout the season. Because beetle abundance never reached meaningful thresholds, this site did not generate interpretable treatment effects and is therefore described qualitatively but excluded from statistical analyses.
A second on-farm trial was conducted in Monkton, where potatoes were planted in mixed rows containing several cultivars, including La Ratte, Magic Molly, Amarosa, Pontiac Red, Russet Burbank, Kennebec, and Purple Viking. Two foliar applications of Beauveria bassiana (BotaniGard) were made in early and mid-July, and Colorado potato beetle populations were monitored four times across the season. The grower also applied spinosad as part of their routine management program. Because potatoes of multiple varieties were interplanted within each row, it was not feasible for the grower to selectively withhold their standard insecticide application from specific plots without compromising crop management. As a result, BotaniGard performance at this site is interpreted within the context of an active grower-managed IPM system rather than as an isolated biocontrol treatment.
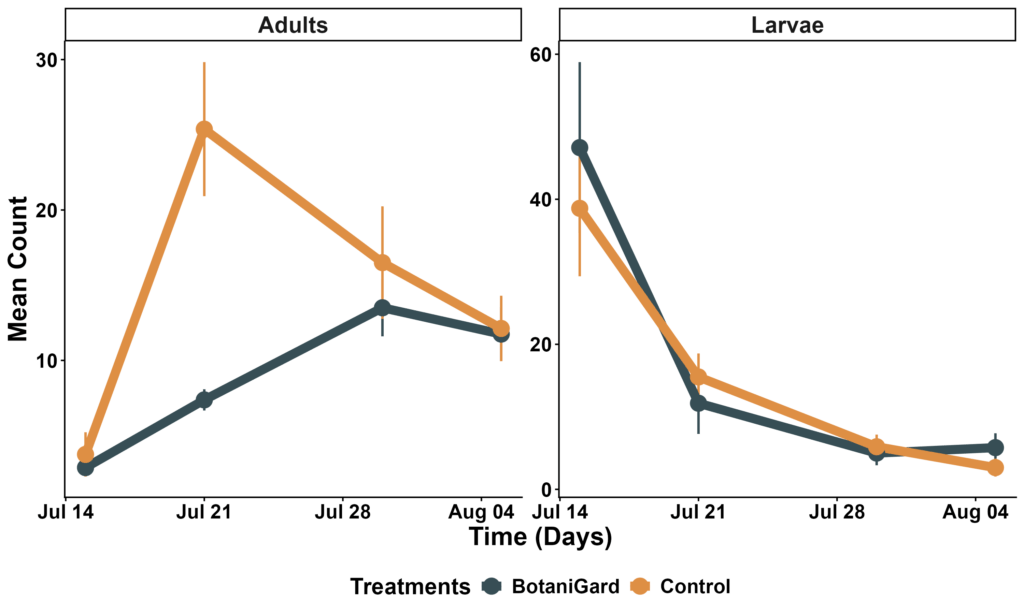
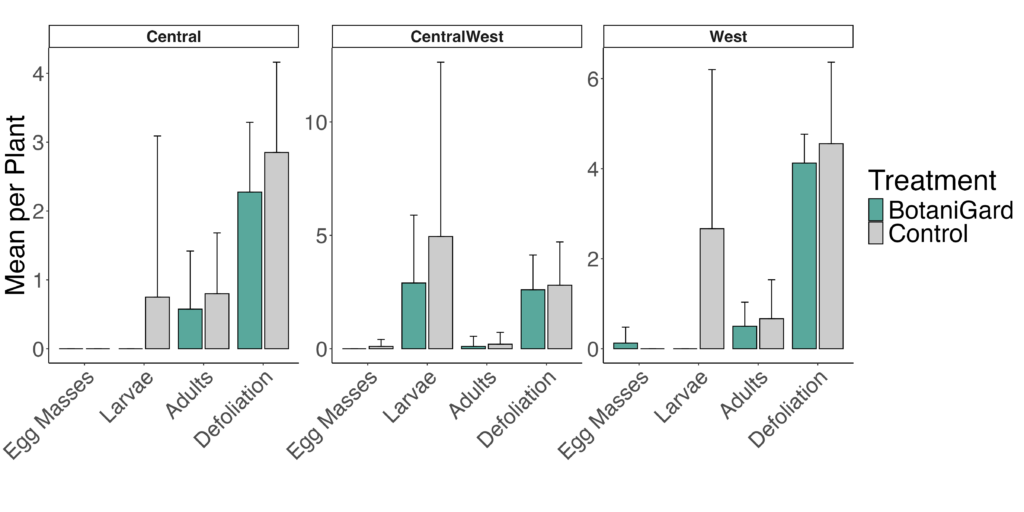
CPB populations showed strong seasonal patterns in the field, with larval densities declining steadily through the sampling period and adults peaking mid-season before dropping again. Larval abundance was strongly affected by sampling date (all p < 0.05), but Beauveria (BotaniGard) did not reduce larval numbers at any time point (treatment and interaction terms p > 0.40). Estimated marginal means confirmed that larval counts did not differ significantly between BotaniGard and control plots on any date (Figure 1).
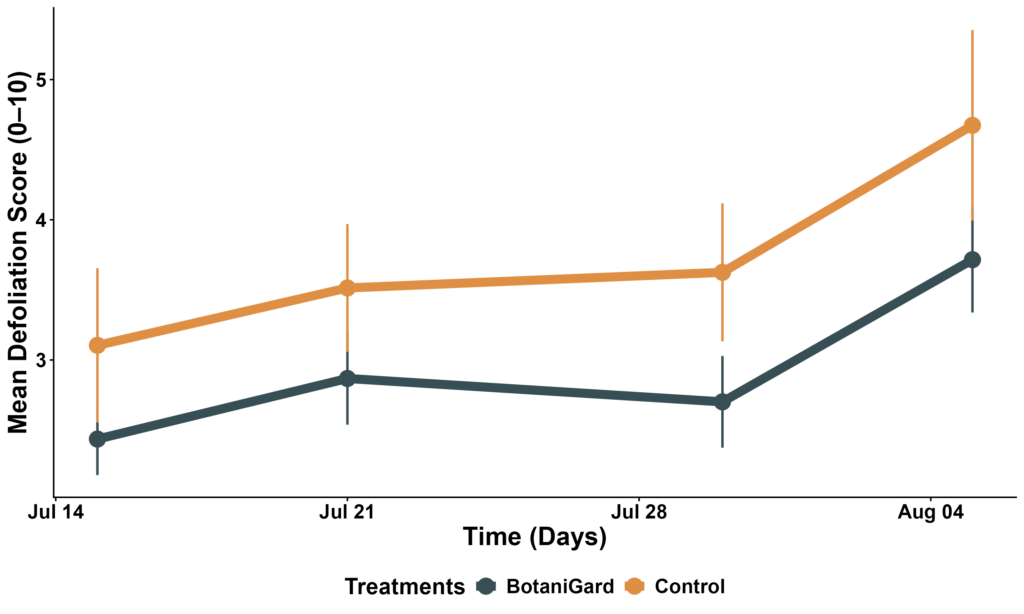
Adult CPB responded differently. Although there was no overall main effect of treatment, a significant treatment × date interaction occurred at the seasonal peak. BotaniGard plots had significantly fewer adults on 21 July (interaction: p = 0.022; EMM contrast: p < 0.0001), indicating a short-lived suppression of adult emergence (Figure 1). This effect did not persist on later dates. Defoliation increased over time (date effect p < 0.001), but fungal treatment did not reduce plant damage on any sampling date (all treatment effects p > 0.10) (Figure 2).
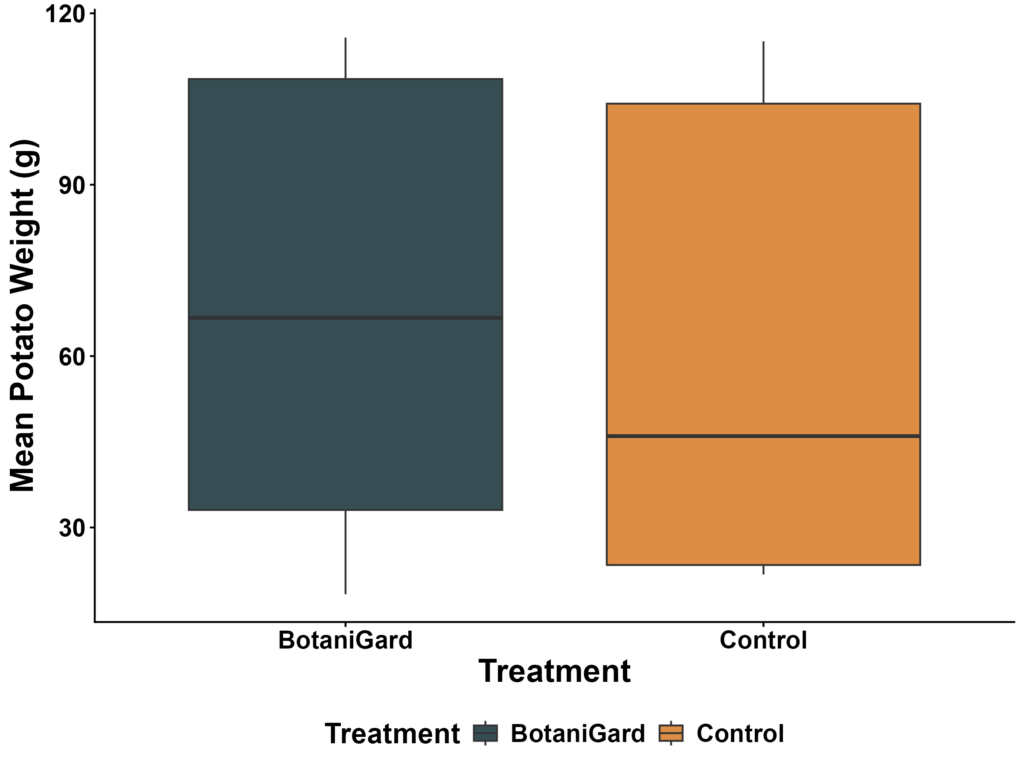
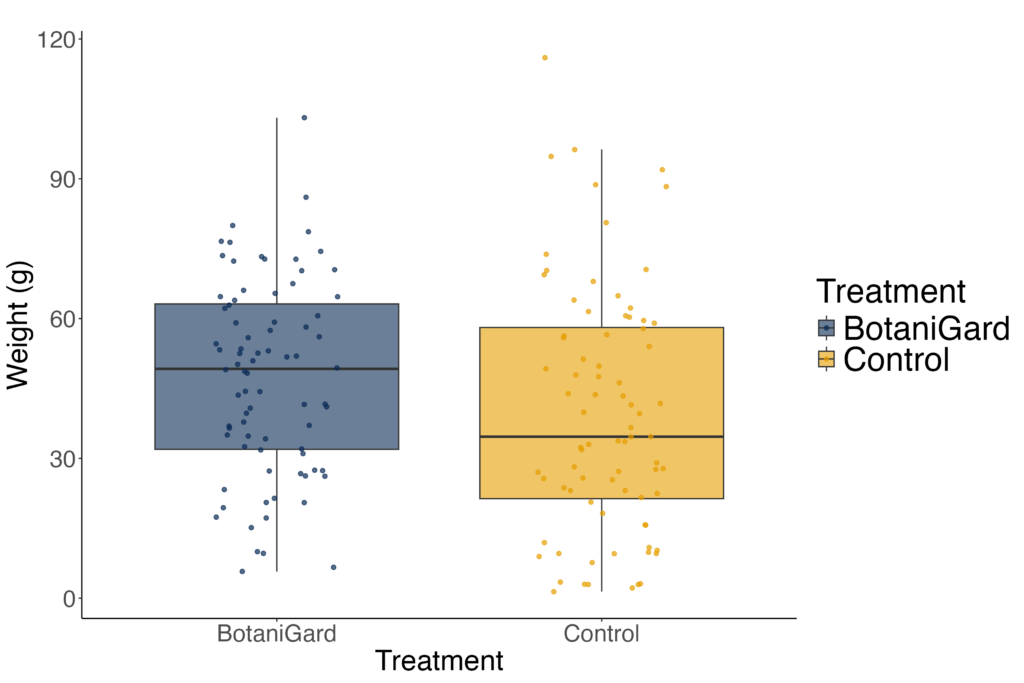
Mean tuber weight did not differ between the BotaniGard and Control treatments. A log-transformed linear model indicated no treatment effect on yield (F₁,₁₄ = 0.13, p = 0.72). Back-transformed marginal means were 48.4 g (95% CI: 27.8–84.4 g) for Control plots and 55.4 g (95% CI: 31.8–96.5 g) for BotaniGard, with confidence intervals overlapping extensively. Pairwise contrasts confirmed no detectable difference between treatments (p = 0.72). These results show that BotaniGard applications did not affect tuber weight within this trial (Figure 3).
Taken together, sampling date was the dominant driver of CPB population dynamics and crop injury. BotaniGard caused a brief mid-season reduction in adult beetles but did not influence larvae, defoliation, or yield. The discrepancy between larval and adult responses likely reflects unobserved mortality during the pupal stage—where Beauveria commonly acts—as well as larval movement among neighboring field plots, which can obscure treatment effects in open field conditions.
Research Farm Trial
We conducted the research trial at the University of Vermont Horticultural Research and Education Farm. Our research plot consisted of 8 rows each with 10 8 ft plots with 6ft buffer plot planted with cover crop in between. We used Norland Red potato variety. Applications were done in first and second week of July. Potatoes were harvested in mid August (Figure 8–9).
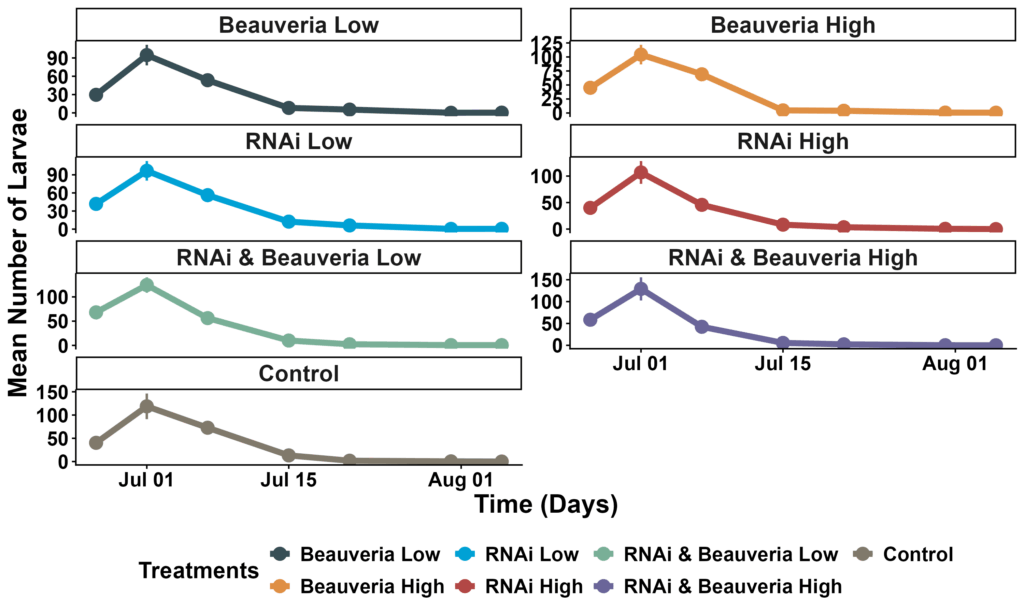
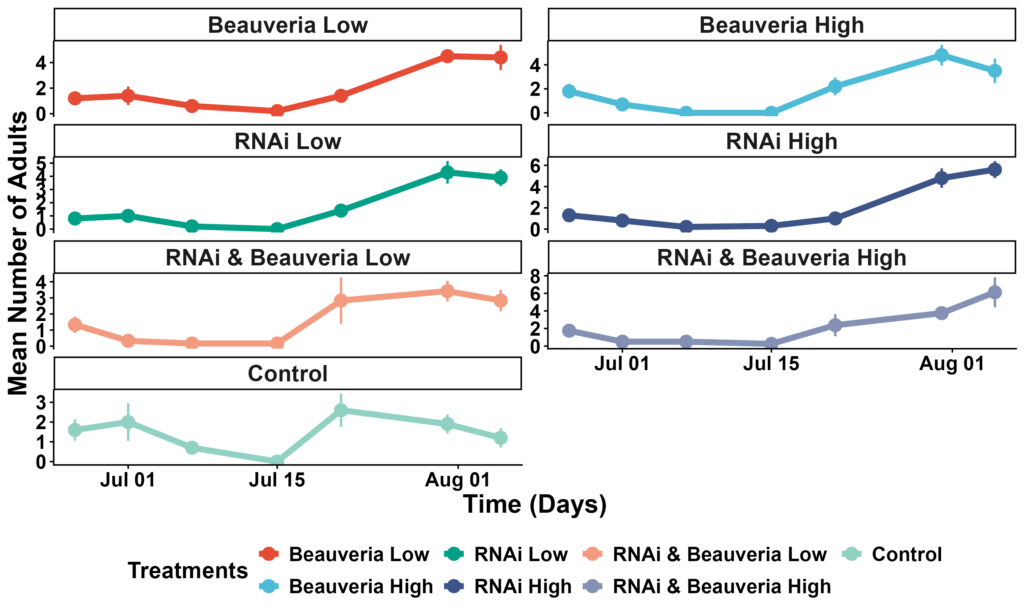
Across the full dataset, larval abundance did not differ significantly among the seven treatments (glmmTMB with negative binomial distribution, all P > 0.05). Estimated marginal means (back-transformed) ranged from ~27 to 38 larvae per plant, and pairwise contrasts showed no statistically distinguishable differences after Tukey adjustment. Seasonal timing was the dominant driver of CPB pressure: larvae were highest at the early-season sampling (∼95–130 per plant on July 1), declined sharply by mid-July (∼5–13 per plant), and were nearly absent by July 31 (≤1 per plant across treatments). The two-date model (Treatment × DateF) confirmed this pattern—very strong date effects (P < 0.001) but no significant treatment × date interactions. Together, these results show that larval pressure collapsed uniformly across the field regardless of treatment, and no treatment produced measurable suppression relative to the others (Figure 4).
Adult beetle counts remained low throughout the season, and treatment differences were again statistically non-significant (zero-inflated nbinom2 model, all P > 0.05). Back-transformed emmeans ranged from ~2.0 to 3.2 adults per plant, indicating modest and relatively homogeneous adult pressure across the field. Adults followed the same seasonal trajectory as larvae, with the highest densities at the beginning of July and almost complete disappearance by the final sampling date. No treatment achieved detectable adult suppression once multiple comparisons were applied, suggesting that phenology overwhelmingly structured adult abundance (Figure 5).
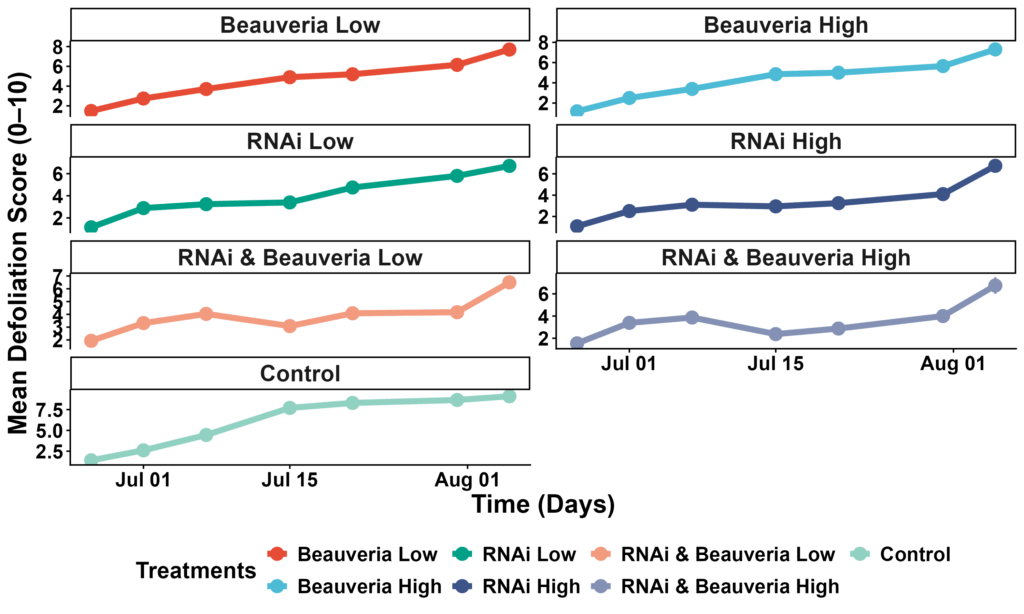
Foliar damage provided the clearest evidence of treatment effects. In the full model, several treatments showed significantly lower defoliation than the Control. Defoliation in the Control averaged ~6.0 on the 0–10 scale, compared with 3.4–4.6 in fungal, RNAi, and combined treatments. The two-date model revealed strong date effects (P < 0.001) and several significant treatment × date interactions: by mid- and late-season, RNAi High, RNAi+Beauveria Low, and RNAi+Beauveria High all produced significantly lower defoliation compared with Control (Tukey-adjusted P < 0.05). These reductions were most pronounced on July 15 and July 31, when Control plots showed the steepest increase in leaf injury. Treatments involving Beauveria alone (High or Low) showed moderate but non-significant reductions relative to Control (Figure 6).
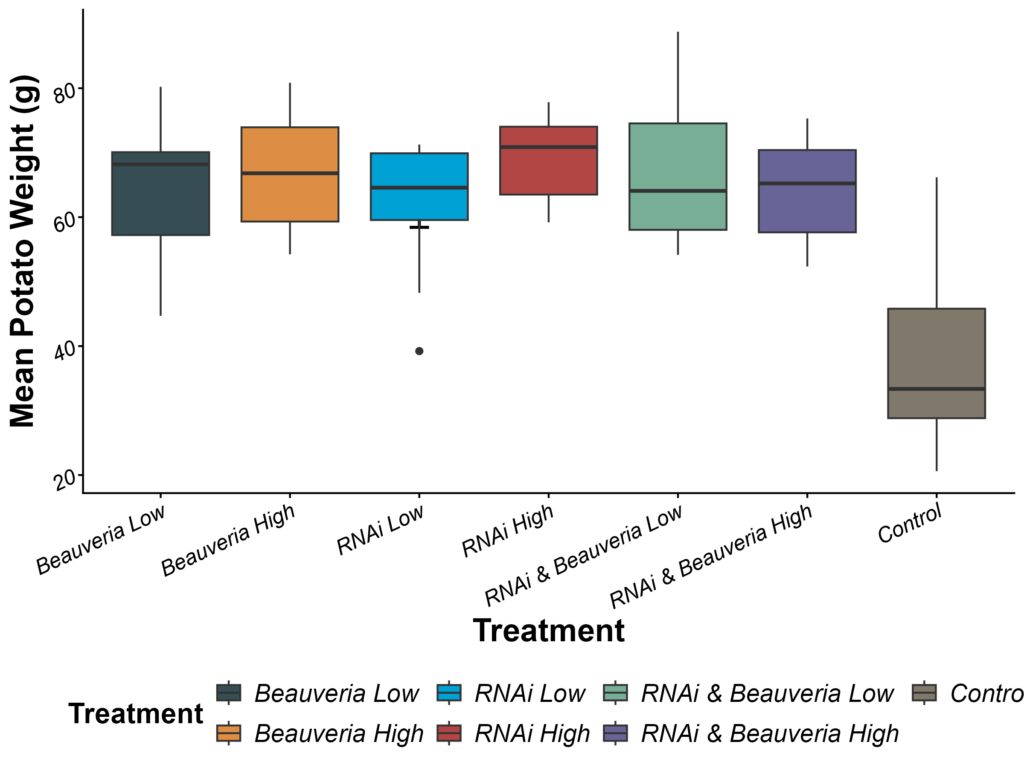
Mean tuber weight differed significantly among treatments (F₆,₆₃ = 13.85, p < 0.0001). After back-transformation from the log scale, estimated mean tuber weights ranged from 31–40 g in the Control plots to 56–79 g across all other treatments. All Beauveria, RNAi, and combined RNAi + Beauveria treatments produced substantially heavier potatoes than the untreated Control, with pairwise contrasts showing that Control plots were consistently and significantly lower than every other treatment (Tukey-adjusted p < 0.0001). In contrast, no significant differences were detected among the six non-control treatments themselves, whose 95% confidence intervals showed considerable overlap (all adjusted p > 0.90). Overall, any treatment involving entomopathogenic fungi, RNA interference, or their combination maintained higher tuber weights relative to the untreated control, but these treatments did not differ from one another in yield performance (Figure 7).
Wireworm Bioassay
In laboratory assays using Melanotus similis—the most abundant species identified across all sites in our 2024 survey—exposure to mycotized formulations of Metarhizium brunneum F52 resulted in approximately 50% mortality, whereas Beauveria bassiana GHA produced lower and more variable mortality (around 20%).
This variation in different fungal species pathogenicity underscores the need to better understand the genetic, physiological, and microbiome-mediated factors that influence EPF susceptibility in wireworm populations.
Figure 1. Trend in larval and adult Colorado potato beetle abundance under control and BotaniGard treatments. Points and lines represent mean counts per plot across four sampling dates.
Figure 2. Mean defoliation scores (0–10 scale) across sampling dates in control and BotaniGard plots.
Figure 3. Mean tuber weight per plot for control and BotaniGard treatments. Boxplots show the distribution of plot-level mean weights.
Figure 4. Mean larval abundance across treatments. Mean number of larvae (± SE) per plant from late June through early August at HREC.
Figure 5. Adult Colorado potato beetle counts over time for each treatment. Mean adult abundance (± SE) across the growing season.
Figure 6. Plant defoliation over time under different treatments. Mean defoliation scores (± SE) on a 0–10 scale.
Figure 7. Mean tuber weight per plot across all treatments. Boxplots show the distribution of treatment-level mean weights.
Figure 8. Our team members harvesting potatoes.
Figure 9. Last day of our research trial at the research farm.
Scheduled presentations at several professional and farmer-focused meetings, including:
- Participatory Action Research (PAR) Meeting (March 12, 2025). The meeting focused on CPB pressure, wireworm species updates, and planning for the 2025 field season. Participants: 8.
- Izzo, V. (March 19, 2025). What’s New in Colorado Potato Beetle Control? VVBGA Webinar. Live participants ~25–30; 172 recorded views by Nov. 30, 2025.
- Izzo, V. (March 19, 2025). Master Gardener Education Class – CPB & IPM Session. Participants ~190.
- Izzo, V. (August 14, 2025). UVM Horticultural Research & Education Center Field Day. Demonstrations included CPB monitoring, fungal application methods, and field trial design, presented by project staff. Participants ~35.
- Nouri-Aiin, M. (December 16, 2025). Biological Control and IPM in Cropping Systems. NOFA, MOFGA, & PCO Northeast Growers Symposium.
- Nouri-Aiin, M. (January 13, 2026). Biological Control Using Microbial Insecticides. Maine Ag Trades Show Winter Symposium.
- Nouri-Aiin, M. (February 14, 2026). New Tools for Smarter Pest Management on Diversified Farms. NOFA-VT Winter Conference.
2024 Progress Report
Hypothesis I: Benchtop granular formulations of B. bassiana and M. burnneum will significantly attract and infect wireworms and CPB larva and result in reduced pest pressure
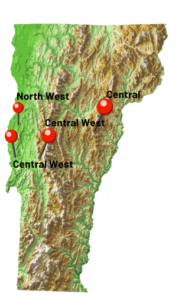
The formulation was tested at the University of Vermont Horticultural Research and Education Center and University of Vermont Greenhouse. The on-farm trails were conducted in four fields in Vermont—Intervale Community Farm in Burlington (North West), Last Resort Farm in Hinesburg (Central West), Bear Root Farm in Williamstown (Central), and Shelburne Farm in Shelburne (West)—to evaluate the efficacy of Beauveria bassiana (BotaniGard) in managing CPB populations (Figure 1). Plots were established with treatment areas ranging from 50 to 200 feet, with an equivalent-sized control plot assigned in each farmer’s field. BotaniGard was applied three times at 5- to 7-day intervals, and data on egg masses, larvae, adults, and defoliation rates were recorded in both the treatment and control plots.
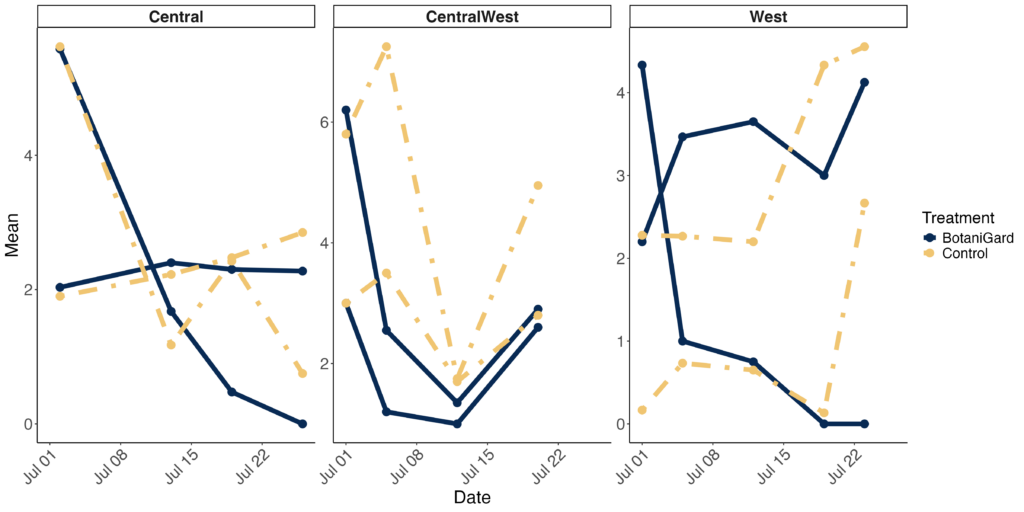
Results varied across sites. The Northwest location experienced flooding, so no usable data were collected there. At the Central site, BotaniGard did not reduce larval numbers, but it did significantly decrease defoliation. This reduction in plant damage is likely due to infected beetles feeding less in the BotaniGard-treated plots (Figure 1).
At the Western Site, plots treated with BotaniGard initially had higher CPB pressure compared to control plots. Following treatment, larval numbers decreased and remained low throughout the season. Defoliation in BotaniGard-treated plots also stabilized for three weeks after application (Figure 1). At the Central West Site, BotaniGard-treated plots showed significant reductions in both larval numbers and defoliation. After the storm, defoliation increased in both treated and control plots. However, BotaniGard-treated plots produced significantly higher potato yields compared to the control, demonstrating the efficacy of the treatment even in challenging conditions (Figure 1, 2).
Hypothesis III: RNAi products will significantly reduce pest pressure associated with CPB infestations in conventionally managed potato crops.
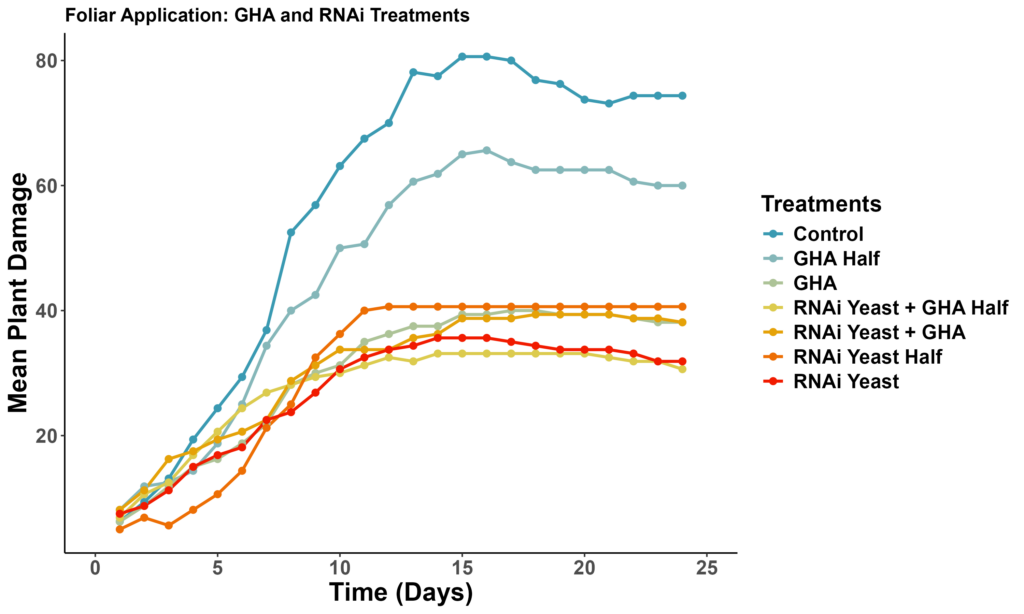
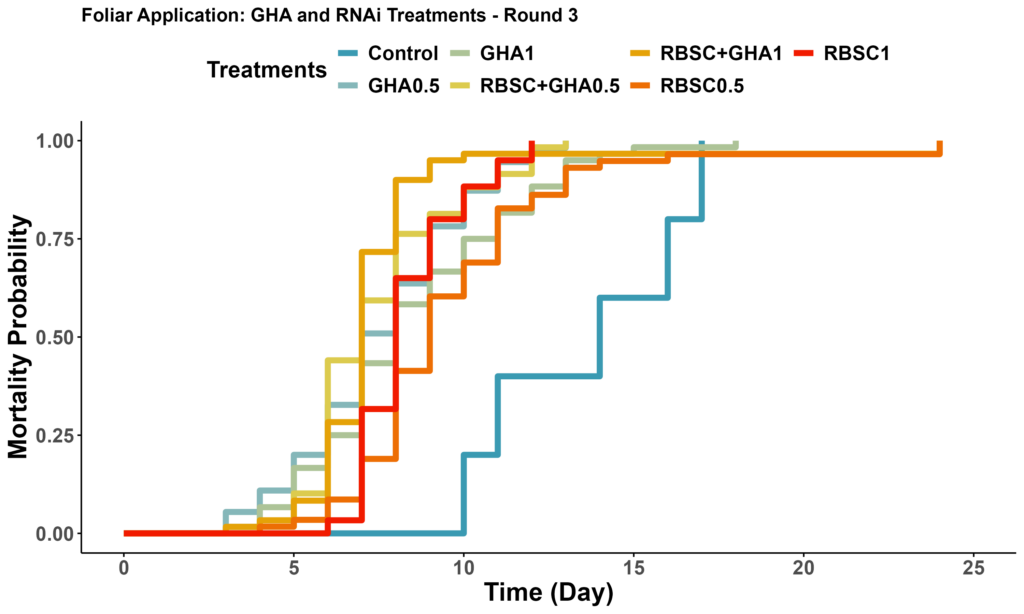
We employed the same setup for the treatments and plant damage recording as our initial bioassay (Figure 4). In our initial bioassay, B. bassiana (GHA) exhibited higher mortality and a shorter LT50 compared to M. brunneum (M52). In the subsequent bioassay, our objective was to optimize dosing. The treatments included a control, RNAi yeast applied at 2.5 kg/ha and 5 kg/ha, and B. bassiana (GHA) applied at 10^6 conidia/ml and 10^8 conidia/ml. Additionally, we tested combination treatments: RNAi yeast at 2.5 kg/ha with B. bassiana (GHA) at 10^6 conidia/ml, and RNAi yeast at 5 kg/ha with B. bassiana (GHA) at 10^8 conidia/ml.
The LT50 results revealed that the most effective treatment was the combination of RNAi yeast and B. bassiana (GHA), which achieved 50% mortality by approximately Day 7. Moderate effectiveness was observed for RNAi yeast alone, B. bassiana (GHA) alone, and RNAi yeast combined with a half-dose of B. bassiana (GHA), all reaching LT50 around Day 8. Slower responses were recorded for the half-dose of B. bassiana (GHA) at approximately Day 12 and the half-dose of RNAi yeast at approximately Day 10. The combination of RNAi yeast and B. bassiana (GHA) provided the fastest action with the lowest LT50. Both RNAi yeast and B. bassiana (GHA) treatments consistently reduced plant damage compared to the control, although the lower concentration of B. bassiana (GHA Half) did not effectively reduce plant damage. Despite LT50 values ranging from 8 to 12 days, treated groups experienced significantly less plant damage, demonstrating the effectiveness of these treatments in mitigating crop losses (Figure 5-left). Plant damage was significantly lower in all the treatments compared to the untreated control (Figure 5-right).
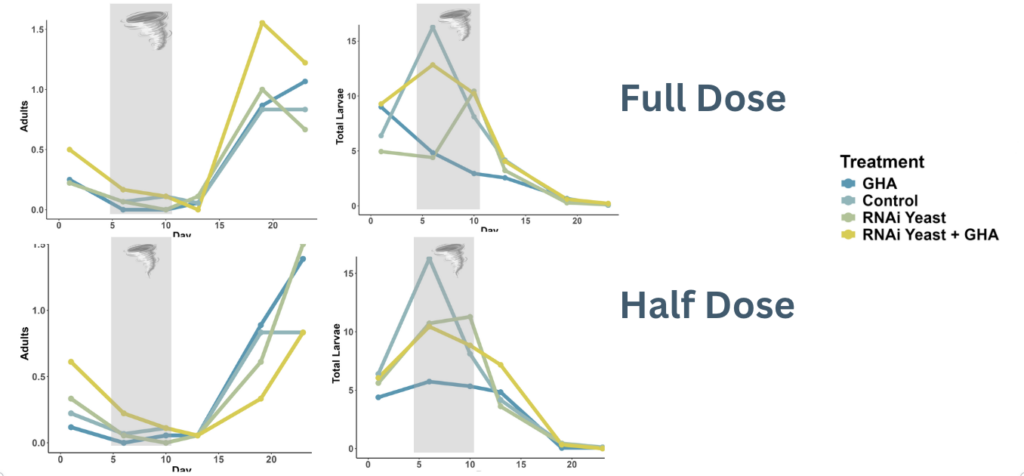
Field trials were conducted in 10-foot by 10-foot plots with six replicates per treatment at the University of Vermont Horticultural Research and Education Center (Figure 6). Treatments were applied three times at 5-day intervals. Monitoring involved counting eggs, larvae, and adults on 10 stems per plot and assessing plant damage before and after each application. The treatments mirrored those tested in the bioassay: control, RNAi yeast at 2.5 kg/ha and 5 kg/ha, B. bassiana (GHA) at 10^6 conidia/ml and 10^8 conidia/ml, and their combinations. At the Horticulture Research Center, the trial demonstrated a clear and consistent reduction in CPB populations. However, following a storm on July 10th, potato plants began to decline due to a fungal disease, resulting in minimal tuber production despite the reduced pest pressure (Figure 7).
Due to the flood and excessive humidity leading to fungal disease in potatoes, some of the results from this year were inconclusive. As a result, an extension request was submitted and approved, allowing for further investigation and validation of our findings in subsequent trials.
Wireworm Survey
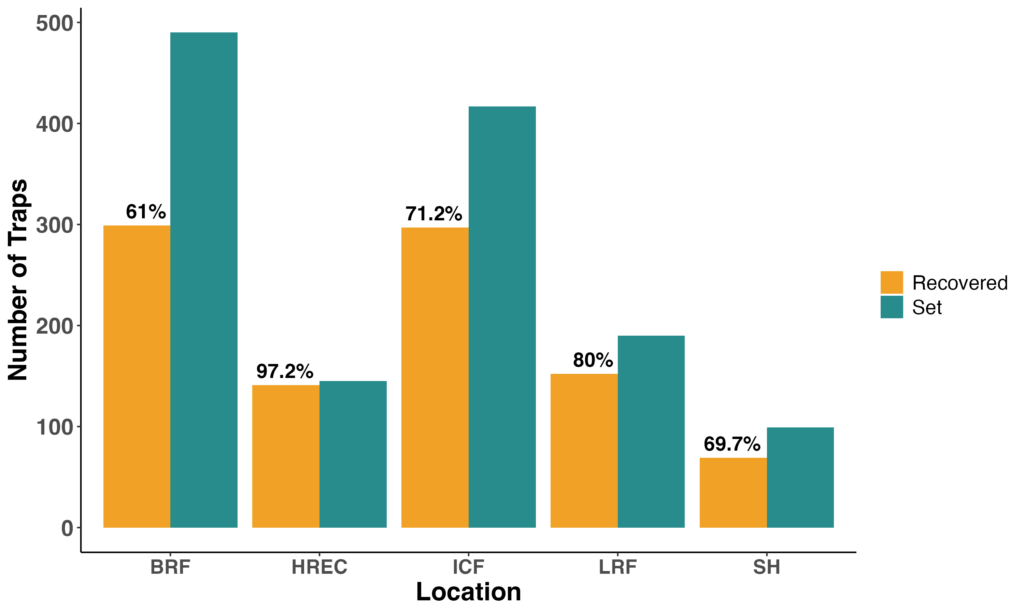
To strengthen our understanding of wireworm communities and improve the design of future fungal efficacy trials, we expanded our trapping effort across five locations (Bare Root Farm (BRF), UVM Horticultural Research and Education Center (HREC), Intervale Community Farm (ICF), Last Resort Farm (LRF), and Shelburne Farm (SH)) throughout the growing season. Across sites, we deployed over 1,500 baited traps, with recovery rates ranging from approximately 60% to 97%, depending on location (Figure 9). These traps were set repeatedly from April through October to capture seasonal activity patterns.
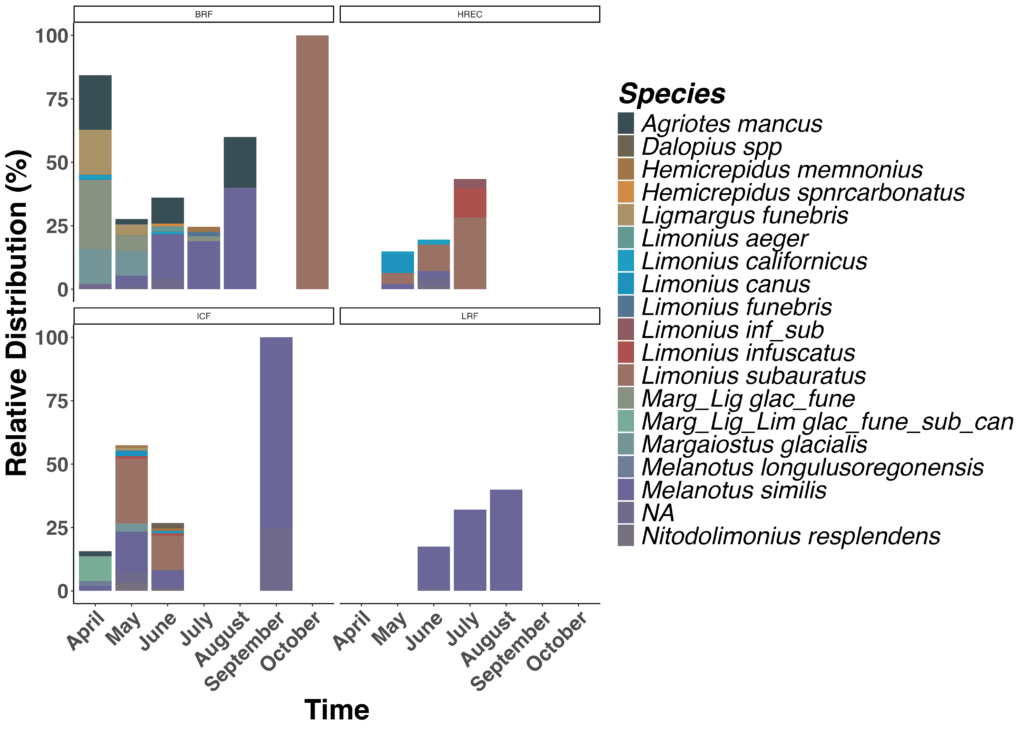
The survey revealed a diverse assemblage of wireworm species, with each farm hosting a distinct community composition and clear shifts in relative abundance across months (Figure 10). Species richness and turnover varied substantially among sites, and several locations showed strong early-season dominance by Melanotus spp., while others were dominated by Limonus or Margaiostus species later in the season.
These findings highlight the ecological complexity of wild wireworm populations. The combination of high species diversity, strong spatial patchiness, and seasonal fluctuations in activity helps explain the variable performance of entomopathogenic fungi observed in earlier field trials. In addition, differences in species-specific susceptibility—shaped by cuticular defenses, life stage, and microbial associations—likely contributes to inconsistent infection rates under field conditions. This survey provides a much clearer baseline for interpreting fungal trial outcomes and will guide the placement, timing, and species-specific focus of our next round of lab and field experiments.
Figure 1. Study sites for Beauveria bassiana (BotaniGard) bioassay trials in Vermont.
Figure 2. (top) Change in the number of CPB larvae, and defoliation rank over the time at 3 different commercial organic farms. (bottom) Mean number of CPB larvae, adults, and defoliation rank on the last day at 3 different commercial organic farms.
Figure 3. Potato crop yield comparison between plots treated with fungus compared to untreated control.
Figure 4. (top) Greenhouse bioassay setup and (bottom) plant damage assessment reference.
Figure 5. Bioassay results: (top ) Mean plant damage in different treatments compared to untreated control. (bottom) Mortality probability by different treatments compared to untreated control.
Figure 6. Plot setup for the bioassay at the University of Vermont Horticultural Research and Education Center.
Figure 7. Result of two different dosages of each treatment on CPB larvae and adults population at the research field trail.
Figure 8. Potatoes plant affected by white wall sclerotia (Sclerotium sp.)
Figure 9. Trap deployment and recovery counts across five Vermont farm locations. Number of baited traps set and recovered at each site during the seasonal wireworm survey.
Figure 10. Seasonal relative abundance of wireworm species across locations. Community composition and temporal dynamics of wireworm species captured from April through October at four Vermont farms.
Our results were presented at several professional and farmer-focused meetings, including:
- Nouri-Aiin, M. (2024, December 17). On-farm innovations for Colorado potato beetle management beyond Entrust. New England Vegetable & Fruit Conference, Manchester, NH, USA. Participants ~ 180
- Nouri-Aiin, M., Lewins, S., & Izzo, V. (2024, November 13). Eco-friendly pest management strategies for late-season root crops: Integrating entomopathogenic fungi and RNA interference in Northeastern diversified farms. Entomological Society of America Annual Meeting, Phoenix, AZ, USA. Participants ~ 60
- Eller, K., Izzo, V., & Nouri-Aiin, M. (2024, November 11). Wireworm populations in Vermont. Entomological Society of America Annual Meeting, Phoenix, AZ, USA. Participants ~ 20
- Vermont Entomology Participatory Action and Research Meeting with Farmers: This meeting engaged 19 farmers, though 9 attended, with 2 unable to join due to emergencies. We presented our findings, discussed experiments, and received valuable feedback to refine our protocols.
2023 Progress Report
Hypothesis I: Benchtop granular formulations of B. bassiana and M. burnneum will significantly attract and infect wireworms and CPB larva and result in reduced pest pressure
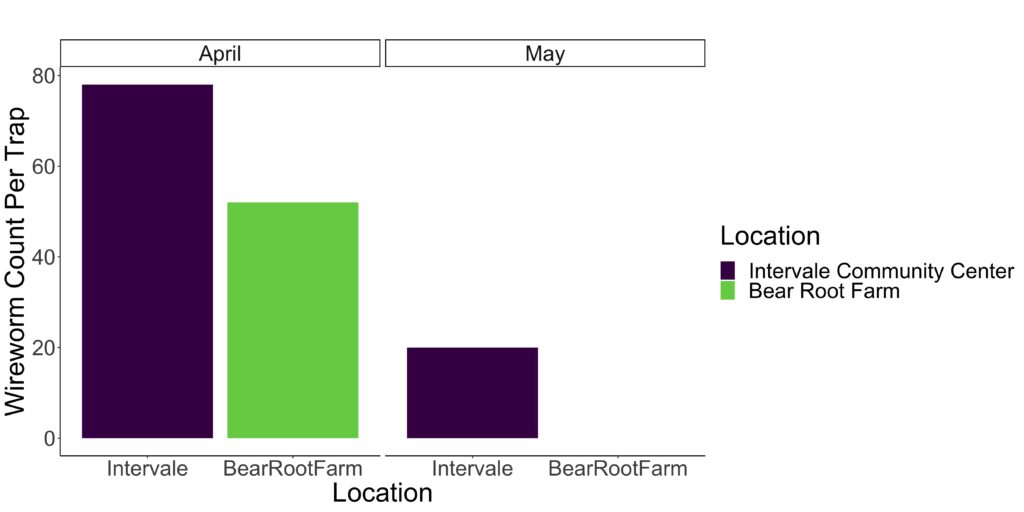
Throughout April, May, and June, an extensive array of traps baited with sprouted wheat enclosed in nylon stockings were deployed at the Intervale Community Center, Bear Root Farm, and Burnt Rock Farm to assess wireworm activity. Subsequently, all captured wireworms underwent morphological identification and were relocated to an insect cage within a greenhouse setting. In January 2024, a bioassay was conducted to on the wireworm population in the colony, utilizing both Beauveria bassiana and Metarhizium burnneum.
Prior to and during the experiments conducted in April, May, and June, wireworm traps were deployed at the Intervale Community Center, Bear Root Farm, and Burnt Rock Farm. The results from April and May at the Intervale Community Center and Bear Root Farm are displayed in Figure 1. Conversely, no significant wireworm pressure was observed at Burnt Rock Farm. Furthermore, by June, the wireworm population had diminished to such an extent that a significant number were no longer detected in the traps, and as a result, the data for this month is not presented. Morphological identification of wireworms was performed for each field separately to determine species, as depicted in Figure 2A and B. The composition of the wireworm population varied between the fields, with Bear Root Farm exhibiting a greater variety of species. Notably, Limonius infuscatus was the most abundant species at the Intervale (Figure 2A), while Athous sp. predominated at Bear Root Farm (Figure 2B). It is worth noting that identifying the specific species of Athous requires DNA barcoding.
A field trial was carried out across four farms, each with two treatments—BotaniGard and M52—alongside an untreated control. Each treatment was replicated five times per farm. Plots (10 ft each) were arranged in a randomized complete block design, with approximately 20 potato plants per plot (100 plants per treatment), and buffer zones were maintained within rows to minimize plot-to-plot interactions. All treatments were applied twice at three-week intervals: the first during the third week of June, incorporated into the furrows, and the second applied using the same method three weeks later.
Across sites, field conditions varied considerably and strongly influenced trial outcomes. At the Intervale Community Center, the first treatment was applied on June 20th, 2023, but extensive flooding occurred before the second application, forcing the discontinuation of the experiment. At Burnt Rock Farm (Hinesburg) and the UVM Horticultural Research and Education Center, wireworm pressure remained extremely low throughout the season—despite high numbers of adult click beetles at HREC—resulting in insufficient larval activity to evaluate treatment effects. These sites were therefore excluded from analysis, and we expanded the project by adding locations with a documented history of higher wireworm pressure next season. The only site with sufficient pressure to complete the assessment was Bear Root Farm, where treatments were applied on June 21st and July 14th, 2023, and potatoes were harvested for damage evaluation on October 3rd, 2023. In October, twenty moderately sized tubers were collected from the center of each plot, washed, and evaluated for wireworm damage incidence and severity (number of galleries per tuber).
Across all farms, our data showed no significant differences among treatments and the control. This outcome is partly attributable to the highly patchy spatial distribution of wireworms, which produced large variability within and among plots. In addition, wild wireworm populations are known to exhibit variable susceptibility to fungal infection, influenced by species-specific defense traits, such as cuticular structure, immune responses, and potentially their associated microbiomes. Because each farm hosted multiple wireworm species, this diversity likely contributed to inconsistent responses and complicates interpretation of treatment effects.
To address these uncertainties, a broader regional survey was carried out in the following season to more comprehensively document species composition and distribution across farms. Based on those findings, we also plan to run controlled laboratory bioassays using field-collected wireworms to better evaluate species-specific susceptibility to Beauveria and Metarhizium under standardized conditions.
Hypothesis II: Farmers will independently produce their granular formulation of fungi using a low-tech protocol
We held one early workshop demonstrating the biology and ecology of entomopathogenic fungi and introducing simple culturing concepts for educational purposes at the NOFA-VT winter conference 2023, titled "D.I.Y Beneficial Fungi: Grow Your Own Fungal Insecticide at Home".
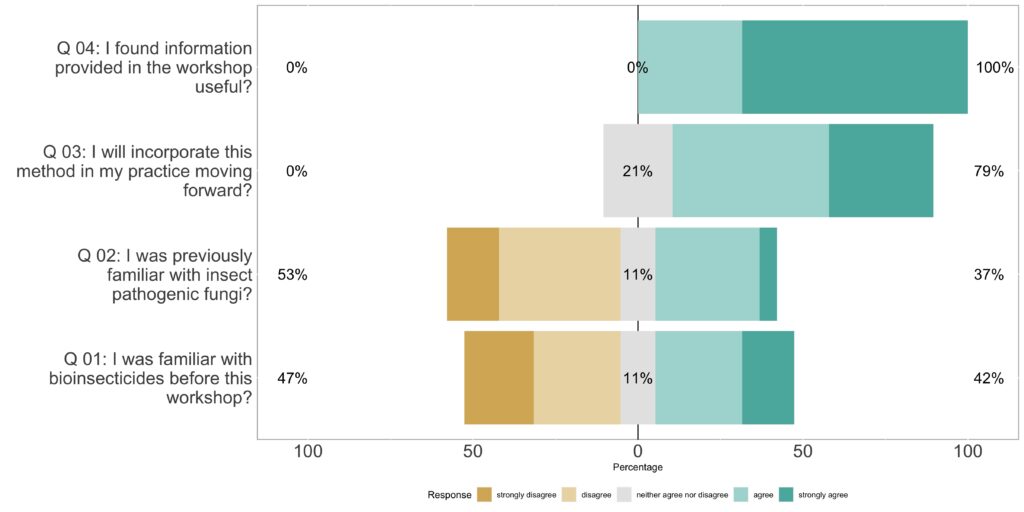
We provided a handout to participants including a detailed explanation and visual representation of each step involved in growing entomopathogenic fungi. The workshop attracted 28 participants, with 19 of them completing our survey. Feedback from the participants indicated a high level of satisfaction with the information presented during the workshop, and they expressed a strong interest in implementing the method in their own practices (Figure 3). As we refined the project, we confirmed that any fungal material intended for pest management must be an EPA-registered commercial product, and therefore on-farm production is not a permissible pathway for grower use.
From that point forward, our outreach shifted toward helping farmers understand how entomopathogenic fungi function, how they integrate into IPM programs, and how to evaluate commercially available products. At the same time, our research trials using mycotized grain formulations continued as planned. These trials are generating performance data that can be made publicly available, offering a foundation that commercial manufacturers may use to develop or refine EPA-approved soil-applied formulations for wireworms and other soil-dwelling pests.
Hypothesis III: RNAi products will significantly reduce pest pressure associated with CPB infestations in conventionally managed potato crops.
At the UVM Horticultural Research Center, a field consisting of 60 plots, each measuring 10 ft with 15 ft buckwheat buffer in between to prevent spraying drift, was prepared. Throughout the week, the number of CPB was diligently recorded to assess population levels ahead of RNAi and EPF applications. However, the presence of a high abundance of predatory insects has prevented the realization of significant CPB pressure required for the application of our treatments.
To tackle this challenge, we've launched a greenhouse bioassay at the UVM Greenhouse, focusing on CPB larvae and employing the same treatments. This ongoing experiment is aimed at generating results that will be presented next year. Currently, we are testing six treatments, including a control, B. bassiana, M. burnneum , RNAi, B. bassiana + RNAi, and M. burnneum + RNAi, with each treatment consisting of 9 replications. Due to the controlled environment of the assay, we intend to explore various approaches, such as targeting different larval stages and pupae within the soil. Our objective is to determine the most effective method for applying fungi in preparation for the upcoming field season.
Figure 1. Comparison of wireworm populations collected at the Intervale Community Center and Bear Root Farm during April and May, revealing a greater abundance of wireworms captured in April at both locations.
Figure 2. Distribution of wireworm species captured in traps at (top) Intervale Community Center and (bottom) Bear Root Farm, expressed as a proportion of the total captured specimens.
Figure 3. Survey results assessing knowledge acquisition during the workshop and gauging interest in implementing this practice by the participants.
2022 Progress Report
Hypothesis I: grains inoculated with entomopathogenic fungi (I.e. benchtop culturing) will reduce wireworm pressure when applied as a soil application
Laboratory trials were postponed to the winter of 2022–2023 because we were unable to collect a sufficient number of field-caught wireworm larvae. Despite multiple trapping efforts using recommended bait protocols, captures remained too low to support replicated bioassays. As a result, lab experiments were rescheduled for the winter and mealworms—an easily reared surrogate with comparable biology—were used for initial screening. RNAi trials were similarly postponed, as commercial suppliers were unable to provide the quantities needed for experimentation. In addition, we prioritized calibrating fungal dosages for below-ground applications before proceeding with a combined above- and below-ground trial.
During the 2022 field season, we conducted our first wireworm trial at Intervale Community Farm in Burlington, VT. Sweet potato slips were planted in 40-inch beds with four rows per bed and 1-foot spacing within rows. Plots (10 ft each) were arranged in a randomized complete block design with four treatments and five replicate blocks. Each plot contained approximately 20 plants (100 plants per treatment), and buffer zones were used to minimize interaction among plots.
Treatments included untreated control, BotaniGard (Beauveria bassiana; labeled rate), M52 (Metarhizium brunneum; labeled rate), and Seduce (spinosad bait; 44 lbs/acre). Applications were made on June 16, August 1, and September 14 by placing materials through the plastic around each plant and incorporating them into the soil. Harvest occurred in October 2022. Twenty sweet potato tubers from the center of each plot were washed and evaluated for wireworm damage incidence and damage severity (galleries per tuber). These metrics were analyzed using a generalized linear model.
Results showed no significant differences among treatments in mean damage incidence, reflecting very high within-plot variability and the well-known patchiness of wireworm distributions. Wild wireworm populations also vary in susceptibility to fungal infection due to species identity, cuticular and behavioral defenses, and potentially their associated microbiomes. Because each farm contained multiple species—and species abundances differed among sites—these factors likely contributed to the lack of detectable treatment differences.
To better understand these patterns, we plan to conduct a broader field survey in the upcoming season to characterize wireworm species composition, spatial aggregation, and seasonal activity. These data will help determine whether wireworm populations are concentrated in specific microhabitats or soil conditions and will guide the design of targeted lab bioassays using field-collected individuals. This additional survey will improve our ability to interpret treatment outcomes and refine fungal application strategies for future trials.
Across four years of trials, a clearer picture emerged regarding the performance, limitations, and practical implications of entomopathogenic fungi (EPF), RNA interference (RNAi) technologies, and integrated management tactics for Colorado potato beetle (CPB) and wireworms in diversified Northeastern farms. While several field seasons were affected by flooding, low pest pressure, and disease outbreaks, the combined evidence from greenhouse, research farm, and on-farm trials supports several actionable conclusions for farmers and informs future research directions.
Practical recommendations for farmers
Based on the data collected to date, the following recommendations were identified with reasonable confidence:
EPF (Beauveria, Metarhizium)
- Were useful as part of an IPM program, particularly when growers prefer reduced chemical inputs.
- Applications were most effective during periods of moderate humidity and consistent CPB pressure.
- EPF were not consistently reliable as stand-alone treatments in years characterized by high pest variability.
RNAi (emerging technology)
- RNAi showed strong potential for protecting foliage and maintaining yield.
- RNAi performed best when used in combination with EPF.
- RNAi represented a promising alternative for growers seeking options beyond spinosad-based controls.
Wireworm management
- Did not provide consistent control from EPF treatments under field conditions.
- Monitoring species composition and seasonal activity was critical for interpreting treatment outcomes.
- Future management strategies may require species-specific or microbiome-informed approaches.
Overall conclusion
Despite adversity across multiple growing seasons, results from greenhouse and field trials demonstrated that biologically based CPB management was feasible, particularly when RNAi and EPF were combined. Although wireworm control remained unresolved, the expanded survey and laboratory assays provided the ecological and species-level context needed to refine subsequent fungal trials.
Education & outreach activities and participation summary
Educational activities:
Participation summary:
Project outreach included a combination of workshops, webinars, PAR meetings, field demonstrations, and conference presentations reaching farmers, agricultural educators, and researchers across Vermont and the Northeast. A participatory PAR meeting engaged six growers in reviewing CPB and wireworm data and refining research directions. A statewide CPB webinar hosted through VVBGA reached over 170 online viewers and continues to receive views via YouTube and podcast platforms. The UVM Master Gardener Education Class included approximately 190 participants, providing an opportunity to educate home gardeners and small-scale growers about CPB management and biological control. A field day at the UVM Horticultural Research & Education Center brought ~35 participants for hands-on demonstrations of plot design, monitoring, and microbial pesticide applications. Project findings were also presented at major regional conferences, including the New England Vegetable & Fruit Conference (180 attendees), the Entomological Society of America Annual Meeting (60 attendees oral and 20 attendees poster sessions), Vermont’s farmer-led PAR meeting (9 growers), NOFA/MOFGA/PCO Northeast Growers Symposium 2025 (40 attendees), the Maine Ag Trades Show 2026 (30 attendees), and the NOFA-VT Winter Conference 2026 (~ 40 attendees).
Learning Outcomes
Farmers and agricultural educators demonstrated gains across several areas central to soil-dwelling pest ecology and biological control:
- Awareness of wireworm diversity in Vermont.
Participants learned that Vermont farms host a wider and more variable assemblage of wireworm species than previously documented. The survey was the first state-level dataset many had seen, and growers expressed surprise at both species richness and strong seasonal turnover. - Understanding of species-specific implications for management.
Farmers recognized that wireworm damage and fungal efficacy can differ by species. This shifted their expectations away from “one-size-fits-all” approaches and toward more nuanced, species-informed management decisions. - Improved knowledge of entomopathogenic fungal products (Beauveria and Metarhizium).
Growers reported a clearer understanding of how these fungi work, especially that they act slowly, are sensitive to weather, and often target pupal stages rather than larvae directly. Educators noted improved clarity on integrating microbial insecticides into IPM programs. - Enhanced awareness of application timing relative to CPB phenology.
Participants learned that treatment success depends heavily on matching microbial pesticide application to CPB life stage—particularly aligning sprays with early larval peaks or anticipated adult emergence. - Greater understanding of why treatment effects can be inconsistent.
Workshops and field discussions helped farmers understand the influence of flooding, humidity, disease outbreaks, predator pressure, and larval movement—all factors that can obscure biocontrol performance in open-field conditions. - Better understanding of re-application intervals needed to target multiple CPB life stages.
Improved awareness that a single application rarely affects eggs, larvae, pupae, and adults simultaneously. They learned that microbial insecticides and RNAi products require timed, repeated applications—aligned with early larval emergence and again before the next adult cohort—to maintain pressure across generations. - More realistic expectations of RNAi and fungal tools.
Growers shifted from assuming “quick-kill” performance to recognizing these technologies as preventative, integrative tools that reduce feeding damage and protect yield—even when they do not strongly suppress beetle numbers. - Stronger interest in soil ecology and below-ground pest dynamics.
The discussions around microbial communities, environmental drivers, and cross-species susceptibility broadened participants’ ecological thinking about pest management.