Final report for LS22-363
Project Information
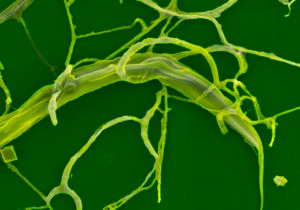
Grazing with the Fun Guy (or fungi). This project will show it is possible to provide a strategy to offer the fungus (see photo of parasite larvae trapped by Duddingtonia flagrans fungus), which controls gastrointestinal worms on pasture leading to improved animal health and productivity, and enhancing sustainability of livestock farms.
Since our first SARE R&E grant in 2002 (LS02-143), we have strived to include nematode-trapping fungus (Duddingtonia flagrans or Df) in the toolbox for livestock producers to control gastrointestinal nematode (GIN) parasites. However, commercialization of the fungus (BioWorma®) took 17 years! GIN are the major health threat for ruminant livestock confounded by dewormer resistance, leading to anemia, poor weight gains and death. The goal of sustainable farms is to eliminate chemical inputs and control GIN; the fungus brings this closer to reality. The fungus can remove much of the GIN on farm. Approximately 90% of GIN are found on the pasture and 10% in the animal; Df can reduce up to 90% of larvae on pasture. The product is considered costly by most farmers (between $0.20 to 0.60 per 100-pound animal per day). Thus, it is imperative to build a strategic program for farmers that considers effectiveness and economics. For example, can Df be fed every other day, or every other week or only in loose mineral supplements? These questions will be answered in research flocks/herds and on farm by examining changes in fecal egg counts, pasture larval counts and/or animal worm counts.
The fungus as part of the farm system will be considered among other management tools to minimize worms and optimize animal health. These include genetic resistance to lower GIN infection and increase economic value (current grant, OS19-124), and use of copper oxide wire particles (COWP) as a dewormer (included in CVs of PIs). These can lead to reduced time spent on GIN management, increased animal growth and production since the immune system is not overburdened, and a greater regenerative agriculture potential without the reliance on chemicals.
We have a rich outreach program through the American Consortium for Small Ruminant Parasite Control (www.wormx.info) which meets twice yearly including virtual meetings. Dr. Whitley is a small ruminant specialist with a strong extension program providing several farmer training sessions yearly (virtual or live) and on an ACSRPC extension committee to produce videos, fact sheets and translation of farmer publications to Spanish. We will communicate with K. Matthews, Delaware State University (LNE21-418), an ACSRPC member also investigating the use of Df. Thus, we can disseminate findings and build recommendations for use of the fungus in the farm system. We will work with Susan Schoenian, University of Maryland (expected to retire) ACSRPC website developer, on progressive development of the ACSRPC website to gauge the frequency of use of materials on the site. We will explore ACSRPC on Facebook and develop more infographics and sound bites of information to lead to more in depth info on the website.
- Examine practical approaches to administer Duddingtonia flagrans (Df) to sheep and goats to obtain good gastrointestinal nematode (GIN) control.
- Examine complementary approaches of using genetics, Df and other GIN control technology within the small ruminant grazing system.
- Develop new outreach materials as fact sheets, videos and webinars in collaboration with farmers, and increase use of available technology for GIN control to disseminate strategies of GIN control that minimize the need for deworming.
Cooperators
- - Technical Advisor
Research
Validation trial. Lambs had access to grass pastures contaminated with parasitic nematode larvae before trial began and were naturally infected. In early January 2023, Katahdin lambs, ~90 days of age, were randomly assigned to 2 treatments (8/treatment; 2 lambs/pen for feeding): 1) control (no D. flagrans or BioWorma), 2) D. flagrans or BioWorma (International Animal Health Prod. Pty. Ltd., Huntingwood, NSW, Aust; Lot no. 3167101, mfr Oct, 2022). All lambs received the same grain supplement and trace mineral mixed into feed fed per kg body weight (Premier Sheep Trace Mineral Premix mixed with salt at a rate of 5:50 per manufacturer recommendations). Lambs were acclimated to pen feeding for 7 days before beginning treatment diets for 7 days. Lambs weighed ~25 kg and were fed 60 mg BioWorma/kg body weight daily. Lambs were housed together and brought into pens in pairs for feeding daily then returned to plots after feeding, and had free choice bermudagrass plus alfalfa hay, water, and access to shelter/shade.
Sample Collection: Feces were collected on first day of diet acclimation, then, one week later there were three sample day collections every other day to determine fecal egg count (FEC). Feces were cultured to examine L3 recovery rate (L3 larvae/FEC) relative to control group and determine the predominant gastrointestinal parasite (H. contortus or Trichostrongylus spp.; plus Cooperia spp. or Oesophagostomum spp.).
Full study. Katahdin lambs, 3 months of age, had access to grass pastures contaminated with parasitic nematode larvae before trial began and were naturally infected. Lambs were randomly assigned to treatments (8/treatment; 2 lambs/pen for feeding): 1) control (no D. flagrans; CON), 2) D. flagrans as BioWorma (BioWorma dose based on the heaviest animal within the group) as recommended top dressed on grain supplement (DfCON), or 3) D. flagrans as BioWorma (same dose as Trt 2) mixed in 7 g/head of a trace mineral mix (Premier Sheep Trace Mineral Premix mixed with salt at a rate of 5:50 per manufacturer recommendations; DfMIN). The mixed mineral and salt were mixed with BioWorma and stored for 7 days before top dressed onto lamb’s supplement. All lambs received the same supplement and trace mineral mix fed per kg body weight. Lambs were acclimated to pen feeding and diets (control) for 7 days before beginning treatment diets for 21 days. Lambs weighed ~25 kg and were fed 60 mg BioWorma/kg body weight daily for 28 days. Lambs had free choice bermudagrass plus alfalfa hay, water, and access to shelter/shade. A similar study was conducted in the field at Louisiana State University (see below).
Sample Collection: Starting on first day of diet acclimation, blood and feces were collected every 3 days (on Mon or Tuesday, and Thur) for determination of blood packed cell volume (PCV; every 7 d) and FEC. Feces will be cultured throughout the trial to examine L3 recovery rate (L3 larvae/FEC) relative to control group and determine the predominant gastrointestinal parasite (H. contortus or Trichostrongylus spp.; plus Cooperia spp. or Oesophagostomum spp.; still need to be identified). Lambs were weighed at start/end of trial.
Objective 2. Examine complementary approaches of using genetics, fungus and other GIN control technology within the small ruminant grazing system. One of the goals of this objective was to use Df in a mineral on farm by farmer collaborators. We learned through thorough investigation that there were not enough spores in the BioWorma used in the first year of study which set us behind. We were unable to conduct the study on inclusion of Df in mineral on pasture until summer 2023. We were unable to find farmer cooperators (some listed in the grant had sold their farm and others had too low parasite burden to test any hypotheses) to complete this goal. However, both the controlled and field study at LSU showed good results in including Df in a mineral mix for sheep. A similar field study in goats was to occur at Fort Valley State University in summer 2024 then 2025 but did not occur; no explanation was provided. The second goal was to examine complementary approaches of using genetics, Df and other GIN control technology within the small ruminant grazing system. The two farms (Coffeys and a replacement farm, Heifer Ranch, Perryville, AR) willing to use Df in a trace mineral had a very low incidence of parasitism in their flocks. These farms are good examples of excellent management practices for grazing and parasite control showing that parasites can be managed, even in Arkansas, so as not to cause undue health concerns in sheep. Their management includes rotational grazing, forage management, planting legumes, use of parasite resistant breeds or animals, and multi-species grazing (with cattle).
Finally, another goal was to consider genetic selection for GIN resistance which is feasible through the National Sheep Improvement Program (NSIP; www.nsip.org) and provides estimated breeding values for parasite resistance and other economic traits associated with farm data. The ARS flock has used NSIP for genetic selection since 2012 and has some of the most resistant sheep in the program. Two other farmer cooperators (J. Morgan and R. Newton for her farm and a collaborator) contributed data for this objective. Lambs (all) with NSIP breeding values were monitored between 60 and 150 days of age, the most susceptible time period for worm infection, for FEC, anemia and deworming events (dewormed when FAMACHA > 3 or packed cell volume < 19%) to determine a “safe” estimated breeding value, or a level that requires no deworming. Three farms (ARS, >200 lambs/year; Morgan; Newton) used 2 high FEC resistant breeding value rams to improve genetic resistance on farm compared with at least 2 less resistant or susceptible rams. Fecal samples were collected from all offspring per sire around 60 and 90 days of age to determine FEC, a measure of resistance. Differences in FEC among sire groups were compared. A report on the results was included under Products. Briefly, there were significant differences among offspring of sires for FEC and FAMACHA scores (measures of anemia). Sires selected for parasite resistance with high genetic accuracy (above 70%) produced offspring with lower FEC and FAMACHA scores (favorable). This is a reason for any flock challenged with parasites to use NSIP-determined parasite resistant sires.
Because not all on-farm objectives could be complete, another experiment was conducted at ARS. Copper oxide wire particles can be an important tool in controlling GIN. This experiment addressed a recent concern raised by a local farmer:
Revisiting copper oxide wire particles for control of Haemonchus contortus in lambs
ABSTRACT: Anthelmintic resistance in small ruminants is a global issue because of the health consequences associated with infection with resistant gastrointestinal nematodes (GIN). Copper oxide wire particles (COWP) used alone or in combination with anthelmintics can be used to aid in the control of GIN including Haemonchus contortus in sheep. Commercially available COWP included loose particles in a gel capsule. Recently, the formulation for cattle changed to a solid mass to be administered to adult cattle. Low doses (≤2 g) are recommended to sheep to minimize risk of copper toxicity; doses <2 g cannot be found commercially necessitating weighing COWP into small gel capsules. The objective was to determine whether the new solid formulation of COWP was effective in reducing fecal egg counts (FEC) associated with GIN in lambs. Naturally infected Katahdin lambs (n=30; 14 males, 16 females), were blocked by sex and previous FEC and randomly assigned to 1 of 3 treatments: control (grain included in the gel capsule; CON); 2 g Ultracruz® COWP capsule (ULT); 2g Copper Oxide (Copasure® for cattle, crushed, weighed and placed in gel capsules; COP; n=10/treatment). Capsules were administered with a sheep bolus gun after collection of fecal samples at 124.0 ± 2.2 days of age (Day 0; body weight, 33.0 ± 1.2 kg). Blood was collected that day to determine packed cell volume (30.8 ± 1.1%). FEC was determined on Days 0, 7, and 14. FEC reduction was calculated as [100 × (1 – FEC14/FEC0)]. Coproculture was conducted each week per treatment groups in replicate to determine genera. FEC reduction tended to be different among treatment groups (CON, 26.4 ± 13.8%; ULT, 64.8 ± 13.1%; COP, 66.4 ± 13.8%; P = 0.09); using orthogonal contrasts, FEC reduction was greater in the COP than CON group (P = 0.05) and both COWP treatments were reduced more than CON (P = 0.02). Coproculture indicated predominantly H. contortus in lambs. When determining percentage of H. contortus by day 14, the COP group had a lower H. contortus percentage (CON, 92.8; ULT, 87.4%; COP, 80.0 ± 1.0%; P < 0.001, treatment × day). The COP formulation effectively reduced FEC by day 14, and both COWP treatments reduced FEC. However, there may be resistance by GIN to the COWP products which warrants further research.
Objective 3. Develop new outreach materials as fact sheets, videos and webinars in collaboration with farmers, and increase use of available technology for GIN control to disseminate strategies of GIN control that minimize the need for deworming.
LSU has continued to supply FAMACHA© cards to trained producers and veterinarians. FAMACHA© cards are an aid producers use to identify sheep that are suffering from barber pole worm infection and need treatment. Many of the outreach efforts of the project were accomplished through the website of the American Consortium for Small Ruminant Parasite Control. The project provided support for a project cooperator, Susan Schoenian, who maintained and updated the website, and accomplished several other outreach efforts for the project, including making weekly blog entries and Facebook posts, editing and publishing new Timely Topics on the website of the American Consortium for Small Ruminant Parasite Control, updating other Timely Topics. In addition, Ms. Schoenian reviewed exams and applications for certified FAMACHA© instructors.
Benefits to students, farmers, and other stakeholders:
The main finding of the project that Df retains its efficacy against strongylid nematode larvae when aged in mineral mix will be of considerable importance for probably hundreds of producers who are unable to feed the Df mixed in supplements, as prescribed by the manufacturer. In addition, the on-pasture study conducted in Louisiana provided data for a master’s student, Elisa Preston, to compile her thesis. Also, the project provided the opportunity for a two-day short training course to be offered in the morphological identification of third-stage infective larvae of sheep to three graduate students (University of Rhode Island, University of Arkansas, and University of Arkansas at Pine Bluff) and a senior technician (USDA ARS Booneville, AR), building capacity in these individuals to further their own research work.
Duddingtonia flagrans included in trace mineral mix or feed for control of gastrointestinal nematodes in sheep
J.M. Burke, K. Petersson, E. Kass, J.E. Miller, A. Vatta, M. Acharya, S. Rohila
Objective was to determine efficacy of Duddingtonia flagrans (Df) included in a mineral mix compared with a feed supplement in Katahdin ewe lambs weaned Jan 2022 (76 ± 2.0 d of age; 21.2 ± 1.1 kg). Lambs were supplemented in pairs with a 12% CP sweet feed (226 g and 6 d later 450 g/lamb) that was thoroughly mixed daily with trace mineral mix [7.1 g salt (87.7%), Sheep Trace Mineral Premix (8.8%), and Vitamin ADE mix (3.5%), Premier 1] and coccidiostat (0.2 g of Decoquinate Type A as Deccox®, Premier 1). Lambs were randomly assigned to one of three treatments (n = 8/treatment; 2/pen): 1) control (CON) of no Df, 2) Df (BioWorma® Int. Anim. Health Prod.) mixed in supplement as recommended (DfC; weighed daily at 1.7 g/lamb), or 3) Df added in mineral (DfM; trace mineral was mixed with 13.6 g Df for 8 lambs then stored for 7 d in a Ziploc bag) which was then added to the supplement. The Df dose used was for the heaviest lamb. Pair feeding lasted 1 h, then lambs had access to free choice bermudagrass hay and water. Fecal samples were collected for fecal egg counts (FEC) and coproculture in pairs twice weekly, and blood collected to determine packed cell volume (PCV) weekly between d -2 and 27. Larval recovery was calculated per pair: (L3/g feces/average FEC) × 100. GIN were 51.3% Trichostrongylus spp., 44.9% Haemonchus contortus and < 2% each of Cooperia spp., Oesophagostomum spp. and Teladorsagia spp. Most samples contained some Strongyloides. FEC were log transformed; FEC, PCV, larval recovery were analyzed using repeated measures over time. There was a treatment × date interaction for FEC (P = 0.02) but DfM was similar to CON throughout. PCV were similar among treatment groups (P > 0.10). Surprisingly, larval recovery was similar among treatments (P > 0.10). In working with the Df manufacturer, it was determined that spore count of the BioWorma of this lot number was ~10-fold lower than anticipated (possibly due to mixing) or required for expected larval recovery. In addition, the coccidiostat mixed in the feed may have had an adverse effect on Df viability and deserves further attention. A subsequent study was conducted using BioWorma from a new lot, validating the spore viability and repeating the objective. Results will be presented in next report. Implications are that Df products should be tested for viability before use. This has prompted us to develop simple in vitro cultures to be conducted before research studies. These in vitro studies will be presented in the next report.
Df in mineral to sheep on pasture at Louisiana State University. The objective was to determine the effectiveness of mixing Df in mineral to sheep on pasture. The Df spores must survive long term exposure to mineral to be successful on pasture. In 2023, at LSU, 150-day old Katahdin and Hampshire lambs of mixed sexes weighing 34 kg (mean) were randomly assigned to 1) a control of no Df, 2) Df to be fed as recommended top dressed on a corn/soybean meal supplement, or 3) incorporated at a rate estimated to be the same in a loose trace mineral mix (based on determined intake of trace mineral mix prior to initiation of study), n = 16/treatment, 2 reps/treatment (48 lambs total). Lambs will be fed for 14 d to acclimate and 28 days on treatment. All groups had free choice mineral and same feed per unit body weight according to NRC recommendations. For the Df/mineral, the combination was “aged” by mixing 7 days before feeding and replaced every 7 days. Measurements: Blood was collected every 7 days for determination of packed cell volume, and feces for FEC and fecal culture twice weekly, described above. As in the last subobjective, larval recovery was the most important measurement for this objective. Lambs were weighed every 7 days and need for deworming was recorded. Data were analyzed using mixed models with repeated measures over time (SAS). Variables in the model included treatment, sex, breed, replicate, day or week, and all possible interactions.
Gastrointestinal nematode larvae were predominantly H. contortus. The mean FEC among treatments was 3343 eggs/g and mean PCV was 23.1 +/- 0.8%; no differences were detected among treatments. Importantly, larval recovery of both Df treatments was reduced (P < 0.001) compared with control, and no difference detected between the two Df treatments. Thus, Df remained viable in the aged mineral in a field study in which sheep were allowed ad lib access to a mineral mix that contained Df.
Interaction between Duddingtonia flagrans and coccidiostat on viability of Haemonchus contortus larvae from lambs. (JM Burke, S Rohila, JE Miller, K Petersson, A Vatta). Providing dietary spores of Duddingtonia flagrans leads to fewer gastrointestinal nematode (GIN) larvae on pasture, but little is known about interaction with dietary coccidiostats. The objective was to determine the efficacy of D. flagrans (Df) fed to sheep with or without decoquinate (Study 1 and 2) or lasalocid (Study 3) to reduce the number of GIN larvae (predominantly Haemonchus contortus) cultured. All studies used a 2 × 2 factorial design (± Df × ± coccidiostat). Study 1: An in vivo experiment used 8 lambs/treatment using the recommended 5 × 105 chlamydospores/g carrier, but in retrospect, the package contained 10-fold fewer spores. There was no reduction in cultured larvae associated with Df. Study 2 and 3: anthelmintic-treated lambs were fed a supplement with coccidiostat (+C; decoquinate or lasalocid in Study 2 or 3, respectively) or without (-C) for 7 (Study 2) or 14 (Study 3) days before fecal collection; within diet, Df was administered (+DF) or not (-DF) 1 to 2 days before fecal collection. Feces were cultured on agar plates for 7 days at 2 g/plate + 300 L3 of H. contortus or 5 to 6 g/plate + 1000 L3 (n = 3 to 4/lamb) at 25°C. On the 2 g plates, Df reduced L3 recovery (P < 0.001) without an effect by coccidiostat in Study 2 (P = 0.38) or a slight increased recovery (P = 0.05) in Study 3, and no interaction between Df and coccidiostat. Using these methods, dietary coccidiostats did not impair the action of Df (5 × 105 chlamydospores/g) on reducing infective larvae in feces.
Impact of sire selection on managing gastrointestinal worm parasites in lambs
Joan Burke
Introduction
It does not take long to realize animal health complications in lambs in a southeastern U.S. summer, particularly if a worm management plan was not implemented. In fact, poor lamb health can occur in almost every U.S. state if there is enough moisture in pastures used for grazing. Unfortunately, dewormer resistance has been detected in all three classes of dewormers across the U.S. which means that once a lamb succumbs to barber pole worm, a blood sucking nematode parasite that causes anemia, available dewormers may not help. Farmers need to have a solid plan to manage barber pole worm which includes good nutrition, good grazing practices, and good genetics.
What does genetics have to do with anything? Animal resistance to these worm parasites is a heritable trait. That means that a dam or sire that is resistant to parasites will pass that trait to its offspring. Resistance can be measured by counting the eggs in a fecal sample, which relates to the intensity of the worm infection. Fecal egg counts (FEC) can be measured in a group of lambs around the time of weaning and then again post-weaning. The National Sheep Improvement Program (NSIP) uses this information along with similar data on parents and relatives to give an estimated breeding value for that trait. The parasite resistance traits, weaning FEC or WFEC, and post-weaning FEC or PFEC, can be used to select the most resistant lambs in a group to be used as replacements for future generations. Over time, the farm population will become more resistant to parasites and there will be less reliance on dewormers. It was of interest to determine how important WFEC or PFEC of a sire is on offspring FEC on working farms. It was thought that sires with greater resistance (lower WFEC and PFEC EBV or more negative) would produce lambs with lower FEC.
Methods
Farm G. In each of 2022, 2023, and 2024, fecal samples were collected from ewe and ram lambs at weaning (mean age of 72 days, std. dev. of 9.2) and post-weaning (average age of 104 days, std. dev. of 9.6) to determine FEC using the McMaster’s procedure. Body weight and FAMACHA score (1 = healthy and 5 = severely anemic) were also recorded on the same day. Four to five sires were used in each year representing between 12 and 31 offspring per sire each year. There were 112, 96, and 99 lambs used in each successive year. Any lamb that had been dewormed within 30 days prior to sampling was deleted.
Farm T. In 2024, lambs were born from 10 rams with a minimum of 8 offspring (offspring from 6 rams with 1 to 6 lambs each were removed from the data). Fecal samples were collected from ewe and ram lambs at weaning (n = 241; mean age of 71.5 days, std. dev. of 9.0), early post-weaning (n = 265; mean age of 104 days, std. dev. of 11.1), and post-weaning (n = 119; mean age of 154 days, std. dev. of 11.1). Body weight was recorded at weaning and post-weaning. Lambs were not dewormed before the post-weaning measurement.
Statistical analysis. For the purpose of this study, the November 2025 NSIP EBV was used for the sire’s WFEC and PFEC EBV. A proc mixed (SAS) analysis was used for FEC, FAMACHA (Farm G), and body weights. FEC was log transformed because of a non-normal distribution; untransformed means are presented for clarity. For Farm G, independent variables included sex, rear type (single, twin, or multiple; rear type of 3 or more was considered multiple or triplet), year and sire with age of lamb used as a covariate. For Farm T, independent variables included sex and sire with age of lamb used as a covariate. Interactions were tested and if not significant, removed from the model. Regression analysis was used to determine the relationship between the offspring’s FEC or FAMACHA score (Farm G) at each age and the sire’s PFEC EBV (WFEC EBV was not used because of fewer observations or lower FEC at the WFEC collection; WFEC and PFEC are highly correlated). The shape of the curve was tested and found to be linear for FAMACHA scores and for FEC in Farm T. The curve was cubic for FEC of Farm G lambs. However, because the shape of the curve was nearly linear, for simplicity of this presentation, the linear untransformed regression equations will be presented along with the actual significant model.
Results and Discussion
Farm G. There was a sire effect on offspring’s WFEC and PFEC (P < 0.001). Year was significant for WFEC (P = 0.005) and PFEC (P = 0.05) but there was no effect of sex or rear type. There was also a sire effect on offspring’s FAMACHA score for both time points (P < 0.001). There was no effect of year, sex, or rear type. This means that the sire influenced the resistance (FEC) and the resilience (FAMACHA or level of anemia) of the offspring, though not uniformly among sires. There was no effect of sire on offspring’s body weight, but as expected, sex (P < 0.001), rear type (P < 0.001), and year (P < 0.001) affected body weight at both time points.
The regression analyses showed a positive relationship between the offspring’s FEC and FAMACHA score and the sire’s PFEC EBV. WFEC: y = 6.27 + 0.0068x – (3.7 × 10-6)x2 (P < 0.001; R2 = 0.09) where y = WFEC and x = sire’s PFEC EBV (x will be the same for the following equations). More simply, for every 1 unit improvement (more negative) in the sire’s PFEC EBV, the mean WFEC of offspring decreased by 9.5 eggs per gram.
PFEC: y = 7.18 + 0.011x – (6.5 × 10-6)x2 (P < 0.001; R2 = 0.18) where y = PFEC. More simply, for every 1 unit improvement (more negative) in the sire’s PFEC EBV, the mean PFEC of offspring decreased by 21.2 eggs per gram. In other words, a sire with -100 PFEC EBV had lambs with 622 eggs/g at weaning and a sire with -50 PFEC EBV had lambs with 953 eggs/g. That could be a difference between lambs not becoming sick from parasites (from the -100 sire) or lambs that may become sick (from the -50 sire).
For both weaning and post-weaning FAMACHA: y = 1.58 + 0.0025x (P < 0.001; R2 = 0.07) where y = WFEC or PFEC. Or, for every 1-unit improvement in sire PFEC EBV, the FAMACHA scores improved by 0.016. Recall that FAMACHA is a 5-point scoring system. For a sire with a PFEC EBV of -100, his offspring will have a mean FAMACHA score of 1.3 compared with a sire with a PFEC EBV of 0 whose offspring FAMACHA score would be 1.6. That means that within the sire’s offspring, sires with less resistance will have more lambs that might need to be dewormed, especially if their FEC are higher and there are more worms on pasture. Or, a sire with -100 PFEC EBV had lambs with 791 eggs/g at post-weaning and a sire with -50 PFEC EBV had lambs with 1562 eggs/g. Again, that could be a difference between lambs needing to be dewormed (from the -50 sire) or lambs that don’t need to be dewormed (from the -100 sire).
Farm T. There was no effect of sire or any variable on WFEC or PFEC. The mean WFEC for sire groups ranged from a low of 476 to 1412 eggs/g. The mean PFEC for sire groups ranged from 656 eggs/g to 3686 eggs/g but there were only 119 observations due to lambs being removed from the farm. There was an effect of sire on EPFEC (P = 0.007) even though the mean FEC for sires groups were low (the highest mean was 800 eggs/g and the lowest was 111 eggs/g). There tended to be an effect of sire on weaning weight (P = 0.09) with a significant effect of sex (P = 0.004) and rear type (P < 0.001) as expected. There was a sire effect on the offspring’s post-weaning weight (P = 0.002); the weight was influenced by rear type (P < 0.001).
For the regression analyses, there was a relationship between sire PFEC EBV and offspring’s WFEC: y = 6.9 + 0.012x (P = 0.003; R2 = 0.03) where y = EPFEC. Or, for every 1-unit improvement in sire PFEC EBV, the mean EPFEC improved by 6.9 eggs/g.
EPFEC: y = 5.7 + 0.009x (P = 0.03; R2 = 0.03) where y = WFEC. Or, for every 1-unit improvement in sire PFEC EBV, the mean WFEC improved by 12.8 eggs/g.
PFEC: y = 8.0 + 0.016x (P = 0.009; R2 = 0.06) where y = PFEC. Or, for every 1-unit improvement in sire PFEC EBV, the mean PFEC improved by 35.3 eggs/g.
Summary and impacts. Data was collected on NSIP lamb traits from two farms. Farm conditions (environment, management, resources) will always be different, and climate, especially rainfall, will influence lamb parasitism or FEC. Typically, wetter years will yield higher FEC and FAMACHA scores and more reliable differences among sire genetics, although if FEC are allowed to increase too much across all lambs, parasitism can overwhelm even the best genetics. Multiple year studies can yield more information than single year studies, but changes in sires among years will influence offspring production data.
The impact of the study shows clearly that sires with greater parasite resistance yield offspring better adapted to parasite exposure. Their lambs will have lower FEC and FAMACHA or anemia scores, leading to fewer cases of deworming and deaths, and lower parasite load on pasture.
Education
The American Consortium for Small Ruminant Parasite Control performs outreach and teaching through the extension specialists and technology transfer specialists within the group (see www.wormx.info). In addition, knowledge gained through research conducted by the ACSRPC has been integrated into curriculum for veterinary students at the University of Georgia, Louisiana State University, Virginia Tech, Michigan State University, Ohio State University, Tuskegee University, St. George’s University in Grenada, and Ross University in St. Kitts. The website has continued to update fact sheets (at least 14 published) and other materials available (Timely Topics; Other Worm fact sheet series; Infographics; Podcasts; Videos), primarily focused for producers and extension specialists (ACSRPC | Best Management Practices (wormx.info)). The blog focused on synopsis of research articles: Weekly Blog | wormx. Several articles are available in Spanish (ACSRPC | En español (wormx.info)). In December 2023, there were 61,153 unique visitors to the website, primarily from the U.S., Canada, and Australia.
Other education included the following:
Gave invited presentation at Lincoln University on control of parasites in small ruminants, April 2023. Train-the-trainer event attended by 20 extension specialists.
Small Ruminant Field Day, DBSFRC, Booneville, AR, April 2023. Topics included parasite control in small ruminants, grazing and forages, nutrition, health of sheep and goats. Attended by 50 farmers and extension specialists.
ACSRPC meeting, LSU, Baton Rouge, LA, May 2023, attended by 30 members and students. Included presentation and discussion of the SARE nematode-trapping fungus research, outreach and education, collaboration. Virtual meeting, December 2023. Attended by 20 members. Gave update on SARE research.
NC-214 Efficiency of Sheep Production committee meeting, Dubois, ID, June 2023, attended by 30 sheep researchers, extension specialists, and graduate students. Presented Station Report on SARE nematode-trapping fungus research.
Congressional Field Day, DBSFRC, Booneville, AR, August 2023, attended by 50 USDA administrators, farmers, and extension specialists. Gave presentation on research which included SARE nematode-trapping fungus research.
American Dairy Goat Association Annual Conference, Tulsa, OK, October 2023. Gave invited presentation on control of parasites in goats; attended by 20 farmers.
Small Ruminant Field Day, Heifer International, Heifer Ranch, Perryville, AR, November 2023, attended by 50 farmers. Gave invited presentation on control of parasites in small ruminants.
American Sheep Industry Association Annual Conference, Denver, CO, January 2024. Gave invited presentation on nematode-trapping fungus, including SARE research results. Attended by 70 farmers, veterinarians, and extension specialists.
Southern Section American Society of Animal Science Annual Meeting, Louisville, KY, January 2024. Gave abstract presentation in Small Ruminant Production session on SARE research on nematode-trapping fungus. Attended by 40 animal scientists, students, extension specialists, and educators.
Novel Approaches for Parasite Control, Grenada, West Indies (gave virtual presentation), March 2024. Gave invited presentation on genetic control of parasites in sheep.
River Valley Small Ruminant Conference, Russellville, AR, April 2024. Gave presentation on alternative methods of parasite control in sheep and goats.
North Central Arkansas Small Ruminant Conference, April 2024. Gave presentation on alternative methods of parasite control in sheep and goats.
American Sheep Industry Podcast, May 2024. How BioWorma can help control parasites. https://podcasts.apple.com/au/podcast/how-bioworma-can-help-control-parasites-with-dr-joan-burke/id1270278004?i=1000657053587
NC-214 Regional group on Efficiency of Sheep Production, Hopland, CA (attended virtually), June 2024. Gave scientific presentation on comparison of including BioWorma in feed or mineral mix.
Small ruminant conference, Missouri Extension (gave virtual presentation), June 2024. Integrated Parasite Management.
University of Florida Small Ruminant Field Day, Gainesville, FL, September 2024. Gave two presentations on holistic control of parasites and genetic control of diseases including parasites. Attended by ~75 producers, animal scientists and veterinarians.
American Sheep Industry Association Annual Conference, Scottsdale, AZ, January 2025. Gave invited presentation on genetic control of worm parasites in sheep. Attended by 80 farmers, veterinarians, and extension specialists.
FAMACHA training and alternatives to dewormers, Greenwood, AR, May 2025. Gave slide presentation and hands-on training to local sheep and goat producers.
Eastern Alliance for Production Katahdins, Elizabethtown, KY, July 2025. Gave presentation on vector-borne viruses to sheep producers. Also had private conversations with many about genetic selection and control for worm parasites of sheep.
Outreach and Education for LSU:
Theses:
Elisa M. Preston (MS). LSU School of Animal Sciences. Thesis: The use of Duddingtonia flagrans aged in mineral for gastrointestinal parasitic nematode control in small ruminants. Graduated May 2024.
Training courses:
Two-day short training course in June in morphological identification of third-stage infective larvae of sheep to three graduate students (University of Rhode Island, University of Arkansas, and University of Arkansas at Pine Bluff) and a senior technician (USDA ARS Booneville, AR), June 18-19, 2024, LSU School of Veterinary Medicine, Baton Rouge, Louisiana.
Scientific outreach:
Vatta, A.F., 2025. Diagnostics for the management of parasite infections. In: Miller, J.E., Schoenian, S., Burke, J.M. (Eds.). Management practices for controlling nematode parasites of small ruminants. CABI, Wallingford, United Kingdom.
VATTA, A.F., 2024. Parasites—Anthelmintics in sheep and goats: Making the best use of what we have. 2024 Louisiana Veterinary Medical Association Winter Meeting, Shreveport, Louisiana, January 26-28, 2024.
VATTA, A.F., 2023. Deworming sheep and goats? What you should know about anthelmintic resistance and the correct use of dewormers. American Association of Small Ruminant Practitioners Webinar, November 16, 2023.
VATTA, A.F., 2023. A new old parasite—liver fluke. Oral presentation with article published in: Proceedings of the American Association of Bovine Practitioners 56th Annual Conference, American Association of Small Ruminant Practitioners Session, Milwaukee, Wisconsin, September 21-23, 2024.
Educational & Outreach Activities
Participation summary:
The website of the American Consortium of Small Ruminant Parasite Control has been updated to include several Spanish translations of fact sheets and infographics: ACSRPC | En español (wormx.info). In addition there were 10 fact sheets published on Other Worms: ACSRPC | Other Worms (wormx.info), and 11 infographics (ACSRPC | Infographics (wormx.info)). An article was written for the Eastern Alliance Katahdins on how selection for parasite resistance in sheep affects other important traits: How Does Selection for Parasite Resistance in Katahdin Sheep Affect Other Important Traits? - Eastern Alliance for Production Katahdins (easternalliancekatahdins.com). The ACSRPC held their annual meeting at Louisiana State University in May 2023 and May 2024 at West Virginia University to present research reports. Other meetings held twice yearly were virtual.
A field day at Dale Bumpers Small Farms Research Center, Booneville, AR occurred in April 2022, presenting an update to use of nematode-trapping fungus to control parasites on pasture, and other alternative measures of parasite control. A webinar on worm control in small ruminants was presented at a train-the-trainer event at Lincoln University and at a producer's conference in Texarkana in April 2023. A train-the-trainer event was held at Tuskegee University and PIs presented information on small ruminant parasite control. Other various field days and small ruminant events are listed below.
Publications:
Peer-reviewed articles
Murphy, T.W., Burke, J.M., Hess, A.S., Miller, J.E., Wood, E.L., Acharya, M., 2024. Impacts of season and production system on gastrointestinal parasitism and growth in Katahdin lambs. Transl. Anim. Sci. 8, txae174. https://doi.org/10.1093/tas/txae174.
Burke, J.M., Rohila, S., Preston, E., Williams, C.C., Scully, C.M., Delcambre, B.A., Petersson, K., Kass, E., Vatta, A., Miller, J.E., Acharya, M., 2025. Viability of feeding Duddingtonia flagrans spores in trace mineral mix to lambs for control of gastrointestinal nematodes. Vet. Parasitol. 334, 110114. doi.org/10.1016/j.vetpar.2025.110414
Burke, J.M., Rohila, S., Miller, J.E., Petersson, K., Vatta, A.F., 2026. Interaction between Duddingtonia flagrans and coccidiostat on viability of Haemonchus contortus larvae from lambs. Vet. Parasitol. submitted.
Abstracts
Rohila, S., Burke, J.M., Vatta, A.F., Miller, J.E., Bowdridge, S., 2025. Determining interaction between Duddingtonia flagrans and dietary lasalocid on in vitro recovery of Haemonchus contortus larvae. J. Anim. Sci. 103 (Suppl. 2), 96 (abstr.).
Burke, J.M., Lewis, R.M., Murphy, T.W., Freking, B.A., 2024. Use of genetics to estimate resistance of gastrointestinal nematode infection in sheep. The 9th Novel Approaches Conference, St. George’s University, Granada, West Indies. March 2024.
Preston, E., Williams, C., Burke, J., Delcambre, B., Scully, C., Vatta, A., 2024. Effectiveness of the nematophagous fungus, Duddingtonia flagrans, in consuming infective stage larvae following aging of the spores in sheep mineral mix. American Association for Veterinary Practitioners.
Forbes, R.M., Murphy, T.W., Burke, J.M., Lewis, R.M., 2024. Adding parasite resistance to the breeding objective in hair sheep. (abstr. Midwest Sec. ASAS).
Nilson, S.M., Burke, J.M., Becker, G.M., Murdoch, B.M., Petersen, J.L., Lewis, R.M., 2024. Genomic and pedigree diversity of Katahdin sheep: The impacts of breeding and selection. (abstr. Int. Plant Anim Gen.; accepted 11/27/23).
Nilson, S.M., Burke, J.M., Becker, G.M., Murdoch, B.M., Petersen, J.L., Lewis, R.M., 2024. Genomic diversity of Katahdin sheep and impacts on genomic prediction. (abstr. Midwest Sec. ASAS).
Burke, J.M., Rohila, S., Petersson, K., Kass, E. Vatta, A.F., Miller, J.E., 2024. Determining in vivo and in vitro predatory activity of Duddingtonia flagrans and interaction with dietary coccidiostat. J. Anim. Sci. 102 (Suppl. 1), 91-92 (abstr.).
Gunes, H.Y., Howard, R., Fudolig, M., Burke, J.M., Lewis, R.M. 2023. Fit of the zero-inflated negative binomial model to analyze fecal egg counts. J. Anim. Sci. (abstr. National ASAS, July 2023).
Lewis, R.M., Freking, B.A., Heaton, M.P., Gore, K., Burke, J.M., Pejsar, B.G., Burgett, R.L., Brown, D.J., 2023. Determining genetic conditions in U.S. sheep with a medium density ovine bead array. J. Anim. Sci. (abstr. Western Sec. ASAS).
Popular
Burke, J.M., Lewis, R.M., Notter, D.R., 2023. How does selection for parasite resistance in Katahdin sheep affect other important traits? Eastern Alliance for Production Katahdins Newsletter.
Burke, J.M., 2025. Impact of sire selection on managing gastrointestinal worm parasites in lambs. Eastern Alliance for Production Katahdins Newsletter.
Learning Outcomes
Farmers gained knowledge in use of copper oxide wire particles as an alternative to resistant dewormers to control parasites, and proper use of feeding fungus for pasture control of worms.
Farmers gained knowledge on practical application of feeding a nematode-trapping fungus, and an understanding of how the product works, what it does and does not do.
Farmers gained knowledge on the genetics of parasite resistance and its importance in controlling parasites within a flock/herd.
Project Outcomes
In the first year, there were issues with the Duddingtonia flagrans (fungus) product used and the initial research study had to be repeated three times, including a study at University of Rhode Island. Once a new lot of the fungus became available, we determined that the fungal spores were viable and the product worked as expected in reducing larval recovery when animals were fed according to manufacturer's directions, and the spores included in a loose mineral worked even better than that treatment. In vitro techniques were learned in the lab, which reduced turn-around time to validate the quality of the fungal spores used for research (and that can be use on-farm), reduced the number of research animals needed for studies to examine interactions with common feed ingredients such as coccidiostats. This led to the successful completion of field studies conducted at LSU and ARS. In sheep, it was determined that the nematode-trapping fungus can be included in the mineral offered to sheep to reduce larvae on pasture. This practice worked as effectively as including the product in feed, but allows farms that do not feed grain to use the product for parasite control.
The two producer collaborators who were to work on the genetics objective failed to meet the requirements outlined. We have found additional farmers to contribute data for the genetics objective (one from Arkansas and two from Georgia). There will be three year's worth of data on examining differences in offspring parasite infection of known parasite resistant rams and unknown sires. An extension publication geared toward farmers will be forthcoming.
It is very difficult to find livestock farmers to contribute meaningful research data since their farm objectives can be quite fluid. I found two farms willing to feed BioWorma in the mineral, but their parasite load was not high enough for meaningful observations. It's been a common frustration over the last 20 years trying to include farmers on my grants in meaningful ways, other than the education component. Appreciate everything SARE has done for my research program!!
Information Products
- Impact of sire selection on managing gastrointestinal worm parasites in lambs
- Sustainable Control of Internal Parasites in Small Ruminant Production
- Efficacy of Duddingtonia flagrans spores fed in trace mineral mix to lambs in reducing the development of gastrointestinal nematode larvae in feces
- Interaction between Duddingtonia flagrans and coccidiostat on viability of Haemonchus contortus larvae from lambs