Final report for LS22-367
Project Information
The rapid expansion of global agriculture continues to generate large volumes of lignocellulosic crop residues that remain underutilized despite their high potential for recycling into productive resources. Biological recycling through mushroom cultivation offers an effective strategy to convert these residues into value-added products while promoting ecological sustainability. White-rot fungi such as Pleurotus spp. possess powerful ligninolytic enzymes capable of transforming agricultural waste into nutritious mushroom fruiting bodies with documented medicinal properties, including activity against gastrointestinal nematode (GIN) parasites. Following harvest, the resulting spent mushroom substrate (SMS)-rich in organic matter, minerals, bioactive compounds, and beneficial microbial communities-retain considerable value as feed, organic fertilizer, and soil amendment that enhances soil health, nutrient availability, and water-holding capacity. These attributes make mushroom-based bio recycling a promising approach for small and limited-resource farmers seeking low-cost, renewable inputs for sustainable production.
This study evaluated the agronomic, nutritional, and nematocidal potential of SMS within a lettuce production system. Two Romaine lettuce (Lactuca sativa L.) cultivars-Amadeus and Salivus were grown under greenhouse and field conditions using soil-SMS media with or without NPK (10-10-10) fertilizer. Morphological parameters (head height, root length, biomass), chlorophyll content, and yield were measured alongside proximate and chemical analyses of the substrates. Nematocidal assays were conducted using mushroom and SMS extracts against Haemonchus contortus eggs and larvae. Media enriched with SMS, particularly in combination with NPK, significantly enhanced chlorophyll concentration, plant vigor, and yield compared with soil-only controls, with stronger responses observed under greenhouse conditions. SMS also improved nutrient composition of the growing media, while mushroom and SMS extracts demonstrated dose-dependent inhibition of nematode egg hatch and larval development.
Education and outreach activities, including farmer demonstrations, student training, and resource development-further supported the adoption of sustainable bio recycling practices. Overall, integrating SMS into vegetable production systems offers a renewable, environmentally friendly strategy that enhances crop performance, reduces reliance on synthetic fertilizers, promotes soil health, and provides natural biocontrol benefits. The findings lay a foundation for expanded research and broader implementation of mushroom-based bio recycling and waste valorization across the southern United States and beyond.
- Evaluate the effect of varying soil-SMS media compositions, with and without NPK (10-10-10) fertilizer, on the growth and yield of two Romaine lettuce cultivars (Amadeus and Salvius) under greenhouse and field conditions.
- Determine which cultivar-media combination maximizes morphological development and biomass yield of Romaine lettuce.
- Evaluate the chemical and nutritional quality of mushroom, substrate and spent mushroom
- Determine the ability of mushroom and SMS extracts to act as nematicides against the gastrointestinal nematode parasite Haemonchus contortus in small ruminants.
Cooperators
Research
Research
Objectives 1 and 2.
Growth performance and yield of two romaine lettuce (Lactuca sativa L.) cultivars cultivated on different growing media in a greenhouse and field
Evaluate the effect of soil-SMS media with or without NPK (10-10-10) fertilizer on greenhouse lettuce growth and yield
Materials and Methods
The study was conducted under both greenhouse and field conditions at the Randolph Farm of Virginia State University, Petersburg, VA, USA (37°13′37.2″ N, 77°26′18.9″ W). The soil type used is characterized as sandy loam soil, typical of the soil type used for vegetable cultivation. The primary organic fertilizer source evaluated was spent mushroom compost (SMC) obtained from Full Circle Mushroom Compost LLC (Pennsylvania, USA), a by-product of the commercial mushroom industry. The SMC was a pasteurized Type B compost, officially registered with the Virginia Department of Agriculture and Consumer Services (VDACS) as a soil amendment and horticultural growing medium. It was used either alone or in combination with farm soil as a growth medium for Romaine lettuce (Lactuca sativa L.). A commercial NPK (10–10–10) fertilizer (Loveland Products, USA) was included as a reference to evaluate the fertility potential of SMC.
Greenhouse pot experiment was carried out in temperature- and humidity-controlled greenhouses at Virginia State University, Randolph Research Farm to evaluate lettuce growth response to various SMC and fertilizer treatments. The experiments followed a Randomized Complete Block Design (RCBD) consisting of 240 pots in each experiment. The treatment structure included two Romaine lettuce cultivars, ‘Salvius’ and ‘Amadeus’, and ten growth media treatments: (1) 100% soil; (2) 75% soil + 25% SMS; (3) 50% soil + 50% SMS; (4) 25% soil + 75% SMS; (5) 100% SMS; (6) 100% soil + NPK (150 kg ha⁻¹); (7) 75% soil + 25% SMS + NPK (150 kg ha⁻¹); (8) 50% soil + 50% SMS + NPK (150 kg ha⁻¹); (9) 25% soil + 75% SMS + NPK (150 kg ha⁻¹); and (10) 100% SMS + NPK (150 kg ha⁻¹). Each treatment was replicated four times with three observations per replicate, resulting in a total of 240 pots per experiment.
The treatment media were prepared by thoroughly mixing the components in plastic mixing tubs based on their designated volume ratios. Romaine lettuce seeds of both cultivars, supplied by Paramount Seed Company (USA), were germinated in seed starter trays under controlled conditions. Seedlings were transplanted into 0.6-gallon black plastic pots (NSI Classic® Nursery Pots) filled with the prepared media. The pots were placed on tables lined with a plastic sheet maintaining approximately one inch of water, allowing capillary absorption from the base. Water levels were monitored twice weekly to maintain consistent moisture at the upper soil surface. Environmental conditions inside the greenhouse were automatically monitored, controlled, and recorded every 10 minutes using a Micro Grow / Greenhouse Systems Inc. / Johnson Controls automated system. The temperature was maintained between 10–21 °C during the day and 7–13 °C at night, with relative humidity between 50–70% and at least 6 h of direct sunlight daily. Pots were observed weekly for any visible nutrient efficiency or deficiency symptoms. The first experiment was seeded on January 6, 2023, transplanted on February 8, 2023, and harvested on March 22, 2023.
For the field experiment, similar treatment combinations were established following a Randomized Complete Block Design (RCBD) with eight treatments and three replications for each cultivar. The plots were fertilized with NPK at a rate equivalent to 150 kg ha⁻¹, consistent with standard lettuce cultivation practices. Regular drip irrigation was maintained to sustain optimal soil moisture, and all agronomic practices were applied uniformly across treatments.
At harvest, both growth and yield parameters were recorded, including head height, head width, crown diameter, root length, fresh and dry biomass, and chlorophyll content. Chlorophyll content was measured using a MC-100 Apogee chlorophyll meter (µmol m⁻²). The composition of the mushroom, substrate, SMS and SMS-soil mixtures were analyzed before start of the experiment.
Objective 3
Evaluate the chemical and nutritional quality of mushrooms, substrate and spent mushroom Substrate.
Materials and Methods
Crude protein, crude fat, crude fiber, ash, and carbohydrate contents were determined using standard laboratory procedures (proximate analysis) following AOAC (2019) protocols. Moisture was determined by oven drying at 105 °C, ash by incineration at 550 °C, protein via Kjeldahl digestion (N × 6.25), and fat by Soxhlet extraction with petroleum ether (40–60 °C). Mineral contents (Ca, Mg, Fe, Zn, Na) were determined using Atomic Absorption Spectrophotometry (AAS, PerkinElmer Analyst 400) after acid digestion.
Objective 4
Determine the ability of mushroom and SMS extracts to act as nematicides against the gastrointestinal nematode parasite Haemonchus contortus in small ruminants.
Materials and Methods
The mushroom extracts were prepared for nematocidal evaluation. For the hydroalcoholic extract (HE), 44 g of dried mushroom powder was mixed with 100 mL of a 60:40 (v/v) ethanol–water solution and incubated for 24 h at 25 °C with occasional shaking. The mixture was filtered through Whatman No. 4 filter paper, concentrated under reduced pressure using a rotary evaporator, and lyophilized to dryness. The HE was stored at −20 °C until use. For the aqueous extract, 3 g of lyophilized mushroom powder was extracted with 30 mL of methanol–water (80:20, v/v) for 6 h at 20 °C, followed by 15 min ultrasonication and centrifugation at 4000 × g for 10 min. The supernatant was filtered through Whatman No. 4 paper, and the residue was re-extracted twice with the same solvent. Combined filtrates were concentrated under reduced pressure at 40 °C, freeze-dried, re-dissolved in water (200 mg mL⁻¹), and stored at −20 °C.
The nematocidal bioassay of the mushroom extracts against Haemonchus contortus was performed using Egg Hatch Inhibition (EHI) and Larval Mortality (LM) tests. Fresh H. contortus eggs were isolated from the feces of naturally infected meat goats using salt–flotation and sieving techniques. Eggs were placed in 24-well plates containing mushroom extracts at concentrations of 10 and 20 mg mL⁻¹, alongside Albendazole (0.1 mg mL⁻¹) as a positive control and distilled water as a negative control. Plates were incubated at room temperature 20-22 °C for 48 h, and hatched larvae were counted microscopically to determine egg hatch inhibition percentage. For larval mortality, infective third-stage larvae (L₃) were exposed to the same concentrations for 24–48 h, and mortality assessed. Each test was performed in triplicate, and results were expressed as mean ± standard error (SE).
Statistical Analysis
All quantitative data were subjected to a two way analysis of variance (ANOVA) to evaluate the effects of cultivar, treatment, and their interactions on measured parameters. Separate analyses were conducted for the greenhouse, field, and laboratory (nematocidal) experiments according to the respective experimental designs: a two-way ANOVA for the greenhouse and field trials (Cultivar × Treatment).
The general linear model used was:
where represents the observation for the replicate of the cultivar under the treatment; is the overall mean; is the effect of cultivar; is the effect of treatment; is the cultivar × treatment interaction; and is the residual error term.
Analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA). For each experiment, data were analyzed under a completely randomized design (CRD) or randomized complete block design (RCBD), as appropriate. Mean separation among treatments was conducted using the Least Significant Difference (LSD) test at a 5% probability level (p ≤ 0.05), and results were reported as mean ± standard error (SE).
For the nematocidal assays, dose-response data were analyzed using one-way ANOVA to determine significant differences in egg hatch inhibition and larval mortality among treatments. Mean separations were performed using post hoc tests at p ≤ 0.05, and the results were expressed as percentages of inhibition or mortality.
Objectives 1 and 2.
Chlorophyll Concentration (CHC) _ (µmol m⁻²)
Figure 1 shows the chlorophyll concentration response across media treatments for both cultivars.
Chlorophyll concentration by treatment (µmol m⁻²). Amadeus and Salvius responded positively to compost and NPK, peaking at TRT 8.
Chlorophyll content increased with increasing compost proportion up to 50% and was further enhanced by NPK fertilization. Treatments containing both compost and NPK (TRT7–TRT9) yielded the highest chlorophyll values, indicating synergistic effects of organic and inorganic nutrient sources. Amadeus consistently exhibited slightly higher chlorophyll concentrations than Salvius across most treatments, particularly in compost–soil combinations supplemented with NPK.
Morphological Traits
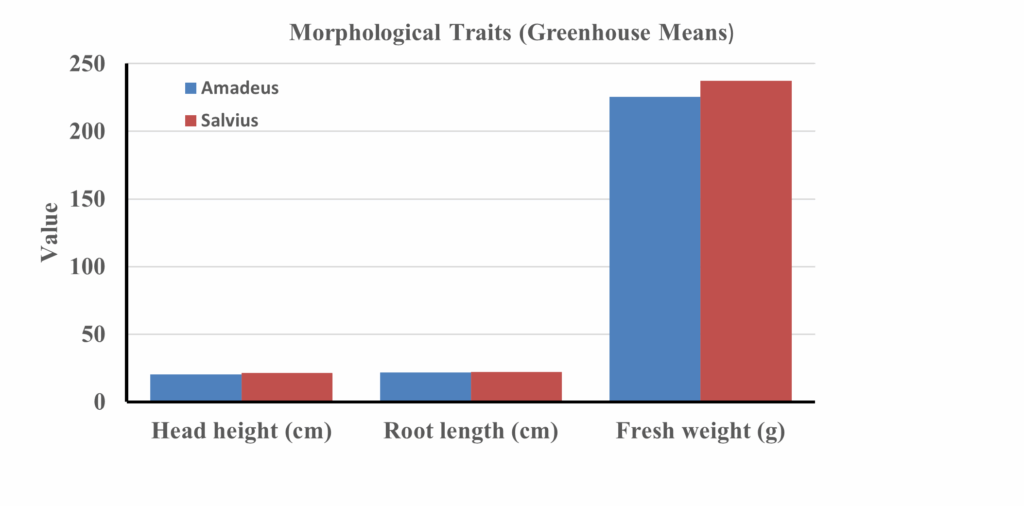
Figure 2 Details greenhouse morphological means (height, root length, fresh weight) for Amadeus and Salvius in the greenhouse
Average morphological performance of Amadeus and Salvius lettuce cultivars under greenhouse conditions is highlighted by three key growth indicators: head height, root length, and head fresh weight. Both cultivars responded positively to compost-enriched media, with improved root architecture and canopy development. Salvius exhibited slightly taller heads and longer roots than Amadeus, suggesting a marginally stronger vegetative vigor. The increase in root length across both cultivars reflects improved substrate aeration and nutrient availability in compost-amended media. Fresh weight—an important commercial trait—was also higher in Salvius, indicating superior biomass accumulation. Overall, the morphology data emphasize that organic amendments, particularly when combined with balanced fertilization, enhance structural growth attributes that directly contribute to marketable yield.
Head Dry Weight

Figure 3. Head dry weight (HDW) by treatment details head dry weight by treatment with standard error bars (±0.3 g).
Head dry weight (HDW) by treatment. Maximum dry matter occurs at 40% compost + NPK (TRT 8). It is a critical indicator of true biomass production, independent of transient water content. Treatments incorporating compost and NPK consistently produced higher HDW values, with peak performance recorded under the 40% compost + NPK treatment (TRT 8). This synergy reflects the combined benefits of slow-release nutrients from compost and immediate mineral availability from NPK fertilizer. Salvius consistently accumulated more dry matter than Amadeus across most treatments, reaffirming cultivar-level differences in resource-use efficiency. The strong alignment between HDW trends and fresh weight patterns suggests that increases in fresh biomass were supported by real gains in structural and physiological biomass, not merely water absorption. These findings highlight HDW as a robust indicator of treatment effectiveness in organic–inorganic integrated nutrient systems.
Objective 2.
Materials and Methods: Eight compost–soil–fertilizer treatments were tested in field plots using the same cultivars. Fertilizer was applied at 150 kg ha⁻¹ NPK. Growth and yield parameters were measured at harvest.
Results and Discussion
Figure 4 compares field yield (t ha⁻¹) for Amadeus and Salvius across treatments (aggregate means shown)
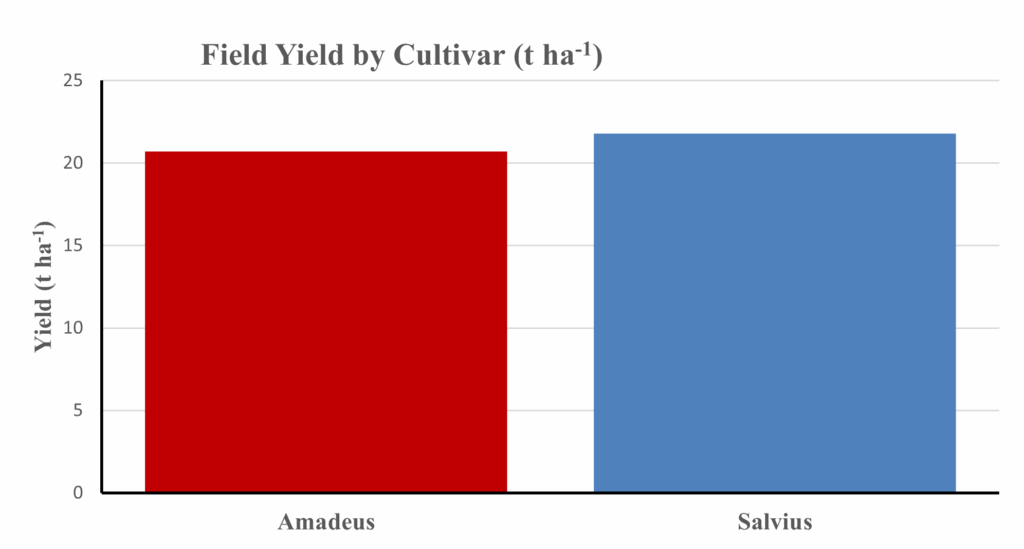
Figure 4. Field yield by cultivar (t ha⁻¹). Salvius demonstrates a slight yield advantage in field conditions.
Despite similar growth patterns, Salvius demonstrated a consistent yield advantage, harvesting approximately 1 t ha⁻¹ more than Amadeus. Field yields were higher overall than greenhouse values due to greater solar radiation, deeper rooting potential, and richer soil–microbial interactions that enhanced nutrient cycling. The responsiveness of both cultivars to compost in field conditions confirms the value of organic matter in improving soil structure, moisture retention, and nitrogen mineralization. The superior performance of Salvius suggests inherent physiological traits possibly including higher chlorophyll stability, stronger photosynthetic rate, or more efficient water uptake contributing to its field adaptability. This result underlines the importance of cultivar selection when scaling bio-recycling strategies to open-field systems.
Objective 3. Evaluate the chemical and nutritional quality of mushroom, substrate, and SMS
Materials and Methods : Soil and leaf samples from selected treatments were analyzed for organic matter and nutrient concentrations. SMS, compost, and control soils were compared for nutrient enrichment potential.
Results and Discussion
Figure 5 presents the stacked composition (leaf N, P, K) across selected treatments.
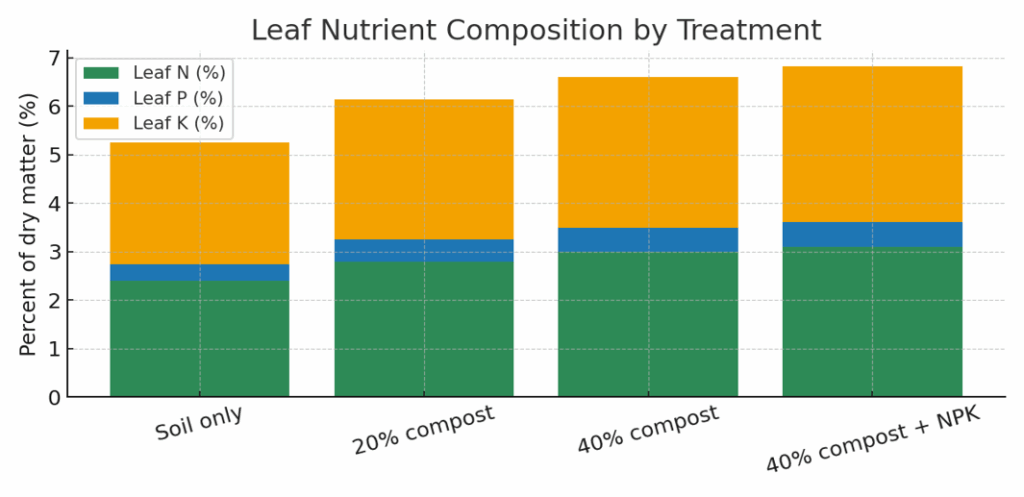
Figure 5. Leaf nutrient composition by treatment. Nutrient uptake increases with compost proportion; NPK addition yields the highest totals.
Nutrient uptake (N, P, K) increased markedly with compost proportion, demonstrating compost’s role in providing slow-release macronutrients and enhancing soil cation exchange capacity. The 40% compost + NPK treatment exhibited the highest leaf nutrient concentrations, confirming that integrated nutrient management maximizes plant nutrient accumulation. Elevated nitrogen levels indicate improved amino acid synthesis and chlorophyll development, while increased potassium supports turgor pressure and stomatal regulation. Phosphorus enrichment—essential for energy transfer and root development—also rose with compost and fertilizer synergy. These patterns align with the observed improvements in biomass (Figures 2–3), confirming that nutrient uptake efficiency is tightly linked to growth performance. The results validate SMS as nutrient-enhancing amendments that enrich leaf quality, supporting both yield and nutritional value.
Leaf nutrient composition (N, P, K %) of lettuce across media treatments. Increasing compost proportion enhances nutrient accumulation, with highest N and K levels observed in the 40 % compost + NPK treatment.
Objective 4. Determine the nematocidal potential of mushroom and SMS extracts against Haemonchus contortus
Materials and Methods: Mushroom fruit bodies and SMS were air-dried and extracted in water/ethanol. Serial concentrations (0.5–5%) were tested in vitro for egg-hatch inhibition at 48 h.
Results and Discussion
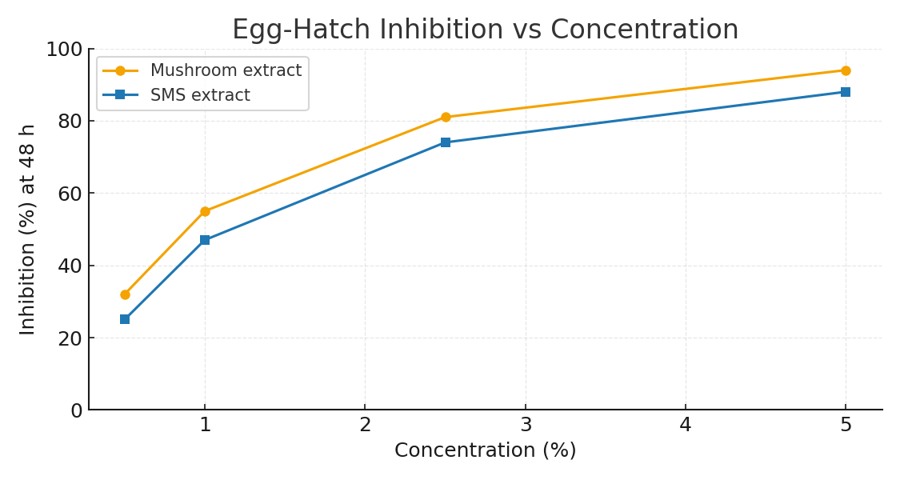
Figure 6 depicts dose–response curves for egg-hatch inhibition by mushroom and SMS extracts.
Figure 6 presents dose–response relationships for mushroom and SMS extracts tested against Haemonchus contortus egg hatch. Both extracts exhibited strong bioactivity, with inhibition levels rising sharply beyond 2.5% concentration. Mushroom extract performed slightly better than SMS across all concentrations, achieving nearly 94% inhibition at 5%, compared to 88% for SMS. This suggests a higher concentration of bioactive secondary metabolites—likely phenolics, terpenoids, or enzymatic residues—in mushroom tissues relative to the spent substrate. The clear, concentration-dependent inhibition trend confirms a true biochemical effect rather than random mortality. These findings demonstrate exceptional potential for mushroom-derived products as low-cost, farm-produced nematicides, providing alternatives to synthetic anthelmintics and supporting integrated parasite management in small ruminants.
Overall Yield by Environment
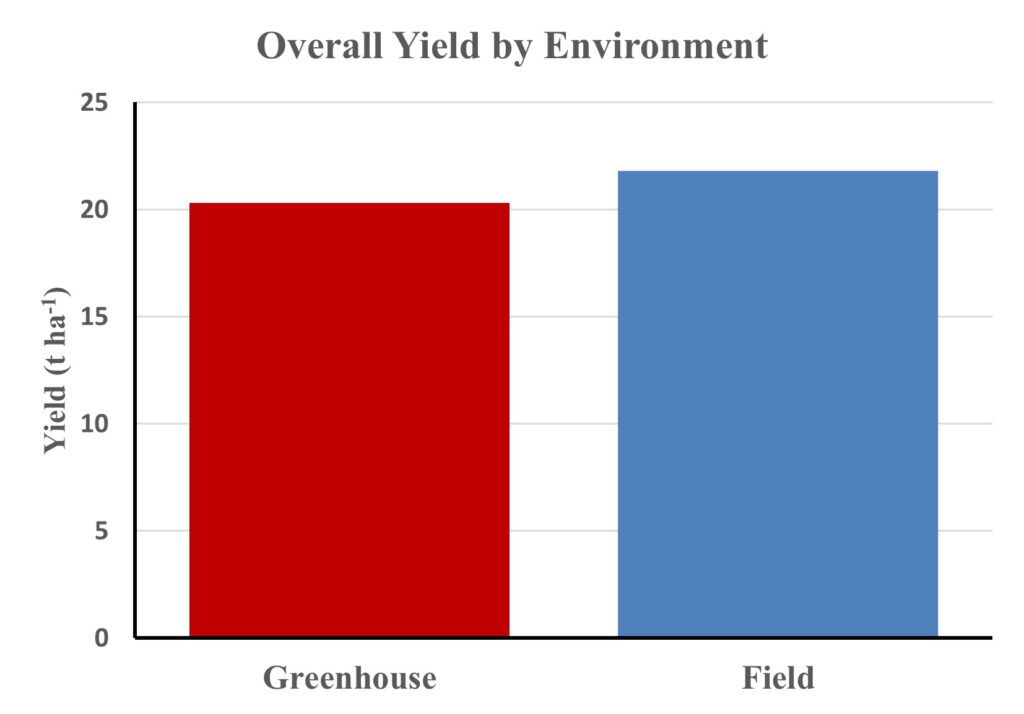
Field production exceeded greenhouse yield by approximately 7%, reflecting the more favorable microclimatic and edaphic conditions of open-field systems. Field-grown plants benefited from full-spectrum sunlight, improved soil structure, and enhanced microbial activity—factors that collectively boost nutrient mineralization and root exploration. The greenhouse environment offered greater control but limited natural airflow and radiation, slightly constraining maximum yield potential. The modest yet consistent yield advantage of field conditions reinforces the adaptability of compost-based bio-recycling systems across growing environments. These results affirm that SMS can be effectively integrated into both controlled and open-field production systems without compromising yield performance.
Conclusion and Recommendations
Conclusion
The integrated evaluation of greenhouse, field, nutritional, and nematocidal studies demonstrates the strong potential of SMS and compost as sustainable components of lettuce production systems. Across multiple experiments, media enriched with SMS—especially when supplemented with moderate NPK fertilizer consistently improved plant growth, structural development, chlorophyll content, and marketable yield. These results confirm that SMS provides both physical and biochemical benefits to soil, enhancing water retention, organic matter content, and nutrient availability.
Between the two cultivars tested, Salvius exhibited greater adaptability, superior physiological vigor, and higher biomass yield in both greenhouse and field settings, indicating its suitability for integrated nutrient management systems. Nutritional analyses further showed that compost and SMS amended treatments increased leaf nutrient concentrations, enhancing the dietary value of lettuce grown within circular production systems.
The laboratory assays demonstrated that mushroom and SMS extracts possess significant nematocidal activity, achieving high egg-hatch inhibition rates against Haemonchus contortus. This finding presents a promising, environmentally safe alternative to conventional chemical anthelmintics, offering dual value from mushroom production residues.
Collectively, these outcomes confirm the viability of a circular bioeconomy model, where agricultural wastes often considered environmental burdens are transformed into productive inputs for food and feed production while simultaneously offering potential pest-management solutions. This approach contributes to soil health, reduces dependency on synthetic inputs, and enhances farm profitability, aligning with the goals of sustainable and regenerative agriculture.
SMS and compost media, when combined with NPK, significantly enhanced lettuce growth, yield, and nutritional quality. Salivus showed superior adaptability and biomass yield. Mushroom and SMS extracts demonstrated strong nematocidal activity, offering eco-friendly options for parasite control in ruminants. These outcomes validate circular bioeconomy principles by converting waste into productive resources.
Recommendations
- Adopt SMS-based growing media in both greenhouse and field systems
Routine incorporation of spent mushroom substrate as fertilizer improves soil structure, nutrient availability, and lettuce productivity. Its consistent performance across environments supports broader adoption among growers. - Apply moderate NPK fertilizer (~150 kg ha⁻¹) alongside SMS for optimal nutrient balance
While SMS contributes organic nutrients, the addition of modest levels of NPK ensures a balanced and readily available nutrient supply. This integrated nutrient management approach maximizes growth while reducing reliance on synthetic fertilizers. - Conduct multi season and multi-location field trials
Long-term evaluations are needed to understand cumulative effects of SMS on soil health, microbial communities, crop rotation outcomes, and potential residual benefits. Expanding trials will also support region-wide recommendations for Extension programming. - Advance research on standardized extraction of mushroom bioactive
Further biochemical characterization and purification of mushroom-derived compounds can lead to low-cost, natural nematicide products. Standardization will improve efficacy, safety, and scalability for livestock producers seeking alternatives to chemical dewormers. - Expand education, outreach, and farmer-training programs
Continued investment in organizing workshops, field demonstrations, and student research experiences will accelerate the adoption of biological recycling of large and cheaply available agricultural wastes-based systems. Strengthening such programs ensures that producers, students, and stakeholders gain practical skills and scientific understanding of sustainable agriculture technologies.
Educational & Outreach Activities
Participation summary:
A comprehensive set of Education and Outreach activities was conducted to extend the project’s findings and support capacity-building across communities, producers, and students. A flagship one-day workshop, “Bio-Recycling Agricultural Wastes to Value-Added Products with Mushrooms,” was hosted at Virginia State University (VSU). Participants, including farmers, students, and Cooperative Extension agents, engaged in hands-on demonstrations covering mushroom and lettuce cultivation and substrate and SMS management.
Multiple outreach events were implemented:
- February 28, 2025: Training and technical assistance were provided to the Powhatan County Cooperative Extension Agent in preparation for an upcoming county-level workshop on bio-recycling and mushroom cultivation.
- May 22, 2025: Mushroom spawn and production materials were supplied to Powhatan County Cooperative Extension for a hands-on Oyster Mushroom Production Workshop attended by 11 participants. Each participant received spawn and growing supplies. Agent John Few led training sessions on both mushroom cultivation and culinary preparation techniques.
- August 21, 2025: Value added products derived from agricultural waste-including mushrooms, lettuce, and composted substrates-were exhibited at the VSU Trojan Farmers Market, where the project reached an estimated 500 visitors, increasing public awareness of waste-to-resource technologies.
- September 12, 2025: A field-day event (see flyer below) attracted 15 producers interested in bio-recycling agricultural wastes for mushroom cultivation. Post-event evaluations showed 100% of participants increased their awareness and knowledge of converting straw and other agricultural wastes into productive mushroom substrates.
- Ribickie Farms, a substitute cooperating farmer located on the North Carolina-Virginia border and known for high-quality gourmet mushroom production, also participated and shared insights from their commercial experience.
In addition to producer outreach, the project supported student engagement and experiential learning. Through funding from the SARE “Yes Grant,” two undergraduate students gained hands-on research experience in sustainable agriculture by participating directly in the lettuce experiments. They learned experimental design, data collection, statistical analysis, interpretation, scientific writing, and presentation skills. Both students presented their research in the Virginia State University Undergraduate Research Symposium (2024 and 2025) and submitted electronic posters to SARE, contributing to regional knowledge dissemination and strengthening future capacity in sustainable agricultural practices.
Project Outcomes
This project demonstrated that biological recycling using mushrooms and spent mushroom substrate (SMS) offers a practical, sustainable strategy for improving crop production, soil health, and natural parasite control. Greenhouse and field experiments showed that integrating SMS into soil–compost media significantly enhanced Romaine lettuce growth, chlorophyll concentration, and biomass yield, especially when combined with moderate NPK fertilization. Cultivar comparisons revealed that Amadeus and Salivus responded differently to SMS-enriched substrates, enabling identification of optimal cultivar–media combinations for improved performance. Physicochemical analyses confirmed that mushrooms, raw substrate, and SMS contain valuable nutrients and bioactive compounds that support their use as organic fertilizers and potential feed resources. Laboratory assays further demonstrated that mushroom and SMS extracts exhibit dose-dependent inhibitory effects against Haemonchus contortus eggs and larvae, providing a promising natural alternative to synthetic anthelmintics. Collectively, the project establishes mushroom-based biorecycling as an effective circular-agriculture innovation that transforms agricultural residues into food, feed, fertilizer, and natural biocontrol resources for sustainable farming systems.