Final report for ONE16-268
Project Information
This project examined two areas of scallop aquaculture: seed/spat production and growout using the ear-hanging method. Spat production can be made more effective and economical using traditional agriculture netting, when the netting provides sufficient surface area and physical stability to keep the spat collector inflated. When this occurs, growers might expect well over 2,000 scallops per collector, and this project provided estimates of 13-19 thousand spat per collector.
In this project, the industry standard material - called Netron - was compared to several alternatives. Over two seasons of tests, we found that a 1/4" square-mesh agricultural netting (OV-7822, Industrial Netting, Minneapolis, MN) performed at least as well as the industry standard, and much more economically. During spat tests we also found that collectors highest in the water column had highest catches of scallops, although settlement of conspecifics such as mussels were also highest.
During tests of ear-hanging culture, the single-ear (itimai-ake) method is better than drilling through both valves of the scallop (nimai-ake). Shell heights can be expected to grow at better than 0.06mm/day in cold weather, and in excess of 0.10mm/day in warmer months. Fouling control is important for good growth and survival.
Although this project was not able to make precise measurements of profitability, it did result in the development of a dynamic cash-flow statement. With this tool, growers can keep track of the important inputs and outputs, and begin to arrive at a conclusion regarding economic viability.
Some conclusions or observations for future consideration:
-Spat collection can be done effectively with less expensive materials than have been used in recent years. Flat-panel collector material may be more efficient than comparable material that arrives as a tube.
-The optimal height of collectors in the water column will vary from site to site; growers are advised to test different heights to learn more about their location.
-Fouling is a constraint to growth of scallops, and regular cleaning will help with better shell growth, meat weights, etc.
-When working with scallops, care should be taken to limit air exposure: work under shade, and keep the scallops in water to the extent practicable.
-Ear-hanging lines may do best with some weight (0.5-1.0kg) at the bottom each line, to keep them from abrading against one another or even wrapping around the headline.
The project proved to be an excellent platform for both collaborative research, as well as hands-on outreach for other producers, students, scientists and other members of the interested public. Outreach materials, which include the major findings of this work, should be helpful and of interest as more producers attempt to raise sea scallops through aquaculture, and as the scientific and regulatory communities grow to meet the opportunities and needs of this new sector.
Principal targets in this period were to:
- Seed 5 shellfish farms and one research farm with ear-hung scallops
- Visit deployment sites to make observations on growth rate, mortality, water temperature, fouling and other operational considerations.
- Deploy alternative spat collection materials, to evaluate effectiveness and profitability
- Begin to track labor and equipment costs
- Conduct outreach activities, and create outreach products, to inform interested parties of the details of the work
Introduction:
Diversification is the biggest need for Maine’s seafood harvesting community. Roughly half of the state’s 9000+ fishermen are lobstermen, the value of the lobster catch accounts for nearly 80% of the statewide landings value, and in the eastern portion of the state, the reliance on lobster is closer to 90% (Anonymous, 2018). This reliance is inherently precarious, but options to make money working on the water are highly constrained by low stock levels and/or limited availability of permits and licenses to diversify into other fisheries.
Aquaculture is presently the most promising avenue for diversification for seafood harvesters. Marine aquaculture in Maine is dominated by the salmon industry, which is typified by high capital investment, and multinational competition, therefore inaccessible to individual fishermen. There is a much smaller shellfish sector, and this sector is growing most rapidly in terms of new businesses being formed. Maine shellfish farming is dominated by production of eastern oyster (Crassostrea virginica) and blue mussel (Mytilus edulis). Several studies have indicated however that the sea scallop (Placopecten magellanicus) grows well in culture in the state’s waters (Pottle and Hastings 2001, Kuenstner 1996, Morse unpublished data), and the Maine scallop commands top dollar in the marketplace, when compared to trip-fished scallops or imported product.
Promisingly, the market for sea scallop meats is large, and increasing. The US sea scallop fishery is the largest in the world at over $500 million ex-vessel value, and yet ex-vessel prices have continued to increase over time, as supplies of scallops from other parts of the world have experienced setbacks. (http://www.undercurrentnews.com/2015/06/23/us-scallop-prices-to-increase-through-season-on-short-imports/). Moreover, imports of scallops and scallop products into the US amounts to roughly $300 million per year.
Taken together – a working-waterfront need for diversification, strong indications of good growth of scallops in culture, and an improving market – the opportunity for scallop aquaculture in Maine is clear.
This project builds on over a decade of experimentation in seed collection, stock enhancement and cage culture production in Maine. Maine fishermen can procure a steady supply of scallop seed, and can grow a high-quality product in an acceptable time frame (http://www.seagrant.umaine.edu/research/projects/dv/scallop-trials). We know that growth rates compare quite favorably with other work done in the state, and to growth rates published elsewhere (Pottle and Hastings, 2001; Kuenstner 1996; Smolowitz, 1999). Product sold into the live market in recent years has captured excellent value; $1-3 per piece (Morse, unpublished). The principal problems with cage-cultured, live-market production are twofold: live sales require a prohibitively expensive level of product testing to document safe levels of phycotoxins (those that cause so-called red tide poisoning); and secondly, that cage-grown scallops do not perform well at high stocking densities, meaning high capital costs for equipment.
The current project addressed two of the above challenges: the optimization of spat collection, and the use of an alternate growout technique, called ear-hanging, which is adapted from Japanese practice. The project also identified some factors in assessing economic viability using this new technique, and created opportunities for others to learn about the work, and to use the findings of this work to advance their own efforts in scallop aquaculture.
Spat collection
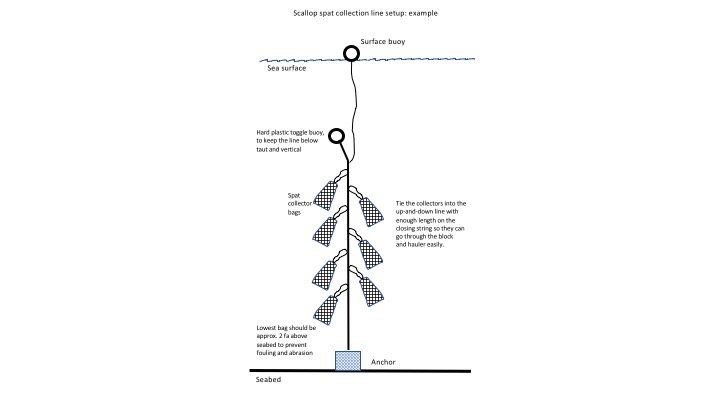
A scallop spat collector is constructed of two parts; an outer bag of polyethylene mesh, and an inner settlement substrate, which also usually consists of polythylene mesh, bunched up so that the collector resembles a pillow. The collector is closed by a drawstring at one end, which also functions to connect the collector to a rope. The rope is usually hung vertically in the water, held in place by an anchor of some type, and marked at the surface by a buoy.
Figure 1 - Left. Spat bags ready for deployment. Note the outer bag (orange) and inner settlement substrate (blue).
Figure 2 - Right. Typical setup for spat collector deployment.
The mechanism of collection is straightforward; scallop larvae in the water are able to pass through the mesh of the outer bag, and encounter the collector substrate, which is commonly referred to as 'spat bag stuffing.' If the larva is competent to settle, then the stuffing provides good substrate for it to do so: the larva goes through metamorphosis, and attaches to the stuffing via a byssal thread. Once attached to the substrate, it soon grows too large to be able to pass out again through the collector mesh, and is effectively captured. The job of the fisherman is to position the collector at the right place and at the right time, to encounter scallop larvae. Experiences along the Maine coast indicate that there are many locations where collections can exceed 2,000 individuals per collector, and sometimes over 10,000 (this report). Collectors are usually deployed between the last two weeks of September and the first two weeks of October, and are emptied the following spring, usually between April and June.
To date, the most common substrate used has been Netron; an extruded polyethylene mesh, blue in color and fairly rigid. Netron has become extremely expensive over time, changing from approximately $60 per 100m roll in 1999, to over $250 per roll in 2016. Given the cost, there has been ongoing interest to find a suitable and less expensive substitute.
Ear-hanging
The practice known as ear-hanging (Japanese: mimi-zuri) was developed in Japan in an effort to advance from lantern-net culture, which is labor- and equipment-intensive. The practice involves drilling a small hole, approximately 1.5mm diameter, into the left anterior auricle of the scallop (the left 'ear' near the hinge), such that the hole is positioned in the byssal notch of the right valve, and passing therefore only through the left valve. Once drilled, the scallop is mounted to a barbed pin (an 'age-pin') which has itself been inserted into a rope; the scallops hang vertically in the water in pairs.
Figure 3. Closeup of ear-hung scallops.
Ear-hung scallops have the benefits of excellent access to water flow, and to be separated from one another, avoiding the tendency of one scallop to clasp upon another, and damaging the sensitive mantle tissue. However, ear-hung scallops need to be cleaned periodically from biofouling organisms such as mussels, barnacles and ascidians. The present industry is highly mechanized and uses specialized equipment to drill and hang scallops, and to wash them once mounted to the age-pins. Here in Maine, there is interest in the method, and Coastal Enterprises Inc. has been successful in acquiring some machinery. This project helps to address the unknowns of how well Placopecten magellanicus, will grow in ear-hanging, and to gather essential data on growth and mortality. In so doing, we hope to contribute to our understanding of the potential for scallops as a profitable culture crop in Maine.
Cooperators
- (Researcher)
- (Researcher)
Research
This project undertook two principal activities:
- Examination of alternate spat bag substrates, to see if an effective, less-expensive alternative could be found.
- Documentation of growth, mortality and husbandry considerations for scallops grown with the ear-hanging method.
Additionally, we undertook to examine some of the considerations when evaluating the financial viability of ear-hanging as a culture method, and also to create opportunities for others to access the activities and results of this project.
SPAT COLLECTION SUBSTRATE EVALUATION
Spat collection Year 1
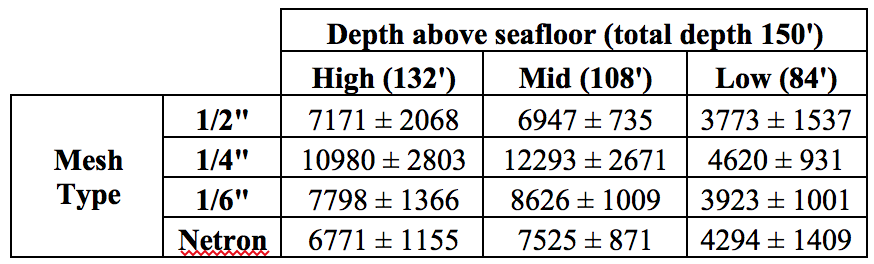
We deployed four lines of spat collectors in September and October 2016, approximately 3 miles SSE of Richmond Island, near Cape Elizabeth, Maine. Each line had four spat collectors (1.5 mm mesh opening) at three depths in the water column: high, mid, and low (n = 48). The collectors were placed in 150’ of water at approximately 132’ (surface),108’ (mid), and 84’ (bottom), above the sea floor. We tested 4 polyethylene mesh types: Netron (industry standard), and ⅙”, ¼”, and ½” agricultural netting. Alternative nettings were sourced from Industrial Netting Inc. of Minneapolis, MN, with product numbers as follows: 1/6” (OV-7100), ¼” (OV-7822), ½” (OV-1581). We retrieved the spat collectors on 21 June and 3 July, 2017.
After retrieving each spat collector, we removed the scallops attached to the mesh, recorded the total dry volume of all organisms, and removed approximately 50 mL for later processing. In the lab, we recorded the dry volume of the sample and counted the total number of scallops, jingle shells (Anomia simplex), mussels (Mytilus edulis), and rock borers (Hiatella arctica). Additionally, we haphazardly selected 50 scallops from each sample and measured the shell height, or the perpendicular distance from the hinge to the opposite edge of the shell. These values were used to estimate the size and total number of scallops and other bivalves in the spat collector.
We tested for the effect of the two retrieval dates on the number of scallop spat, number of other bivalves, and shell height using t-tests, which compare differences between the mean values of two groups. We then tested for the effect of mesh type and depth on the shell height, total number of scallops, and total number of other bivalves in each spat collector using three Analysis of Variance (ANOVA) tests, which look for differences between the mean values of multiple groups or combinations of experimental treatments. We also tested for the effect of depth on the number of mussels per spat collector using an ANOVA test. Data was analyzed using the software packages R (version 3.4.0) and JMP (version 13).
Spat collection Year 2
The Year 2 spat collection experiment was not planned for in the original plan of work, but the partners felt it was important to confirm the prior experiment, and we had some funds left in the budget for materials; the collaborators donated their time in vessel use, sample processing and data analysis. We deployed four lines of collectors October 7, 2017, per the experiment from 2016/2017. Location was also approximately 3 miles SSE of Cape Elizabeth, in approximately 180 feet of water. Lines were paired; two vertical lines weighted by steel pipe anchors, and connected by a sink-rope ground line. We deployed four collectors at each of four depths, approximately 130, 100 and 80 feet from the seabed. Each group of four collectors was comprised of standard spat collector bags (1.5mm mesh size) holding approximately 21 square feet of collector material. Collector materials were: Netron and OV-7822, per the earlier trial, and two new materials, both made by the TENAX corporation: Purima and R9. These materials were chosen because they are broadly used in the shellfish aquaculture industry and are easily available.
On May 24, 2018, all collectors were retrieved. Each collector was emptied, and total volume of the catch was recorded. A sample of approximately 30ml was removed at random from the total catch of each bag, and frozen for later enumeration in the laboratory. Sample numbers were extrapolated to estimate the total number of scallops in the catch. Shell Heights were captured from 30 individuals in each sample, to construct length-frequency distributions for later comparison. A single-factor Analysis of Variance (one-way ANOVA) was conducted on the estimated yields for spat collectors on the two most successful substrates, to compare the effects of substrate type on collection success. The non-parametric Kolmogorov-Smirnov (K-S) test was applied to the length frequency distributions of the two highest-performing substrate types, to determine if the LF distributions were similar.
EAR HANGING TRIALS
The following trials of ear-hanging scallops were conducted on different sites throughout the midcoast of Maine, during 2016, 2017 and 2018. Each is reported separately, since the details of each deployment were different.
Atwood Site - 2016
Two lines of ear-hung scallops were deployed on June 11, 2016, on the site of the Maine Mariculture Company, in Home Harbor, on the northeast side of Pleasant Island, in the Mussel Ridge area of mid-coast Maine. Each scallop had 1.5 mm hole drilled through the shell near the hinge, with a diamond-dust-coated lapidary drill mounted into a Dremel Tool in a Dremel drill press (drill and press were purchased by the Maine Aquaculture Innovation Center and loaned for this project). One ear-hung line contained 34 pairs of scallop (68 individuals) and the other contained 32 pairs. Scallops for both trials were somewhat limited in supply; the site is relatively exposed and there had been damage and loss to the seed stock prior to beginning the trial.
One line (the 34-pair line) was drilled in the regular location; in the byssal notch located in the left anterior auricle, and through only the top (left) valve. This method is referred to as Itimai-ake in Japan, and is the common approach. The other method was deployed in the Nimai-ake method, where both valves are drilled through on the right anterior auricle; the method provides a stronger attachment but can result in excessive tissue damage.
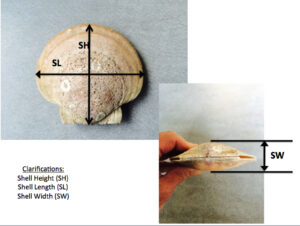
At the start of the project, shell dimensions were recorded with analog hand calipers, to the nearest millimeter: shell height (SH), shell length (SL) and shell depth SD). Figure 4 depicts these dimensions. A datalogger (Onset Instruments HOBO Pendant Temperature Logger) was deployed at the start of the project; tied to the headrope of the longline from which the ear-hanging lines hung.
Figure 4. Shell dimensions used to evaluate growth.
The site was visited on August 16 and December 2, 2016, at which times scallops were cleaned of bio-fouling, and measured according to the established. The data logger was retrieved during the December visit. Project staff intended to continue the trial on the site but all lines and equipment were lost on that site during the winter of 2016/2017, so no further data were available from that site. Growth through December is however summarized later in this report.
Wild Ocean Aquaculture 2017
On 5 January, 2017, three lines of pinned scallops were prepared in the workshop of Wild Ocean Aquaculture, on Commercial Street in Portland, ME. One line contained 30 pins, and two lines supported 20 pins each. Measurements on individual shell height/length/depth were taken by analog calipers to the nearest mm, and recorded. A check on the lines in late winter/early spring of 2017 revealed that all lines had been lost, due to tangling during storms. On 8/2/17, a replacement line with 119 individuals (30' length) was deployed, to the farm site near Bangs Island, in Casco Bay. An Onset Instruments HOBO temp/light logger was deployed near the hanging lines, beginning Aug 18, 2017, continuing every 6 hours until July 25, 2018. An unidentified line of scallops was brought to shore, checked and cleaned on 2/2/18. It appears possible that this line might have been from the original deployment, but any identifying tag had been lost. The August 2018 lines were not returned for measurement at this time. A final sampling of the August 2017 line occurred on July 25, 2018, when measurements on shell height/length/depth were taken, as well as weights for shell, meat, roe and viscera.
Maine Scallop Company (MSC) ear-hanging trial 2017
On June 22, 2017, two lines of ear-hung scallops were prepared in the shop of the Maine Scallop Company, on Deake's Wharf, Portland, Maine. Day 0 data included individual shell dimensions (height, length, depth) and weights of shell, meats, roe and viscera were taken on seven individuals as a general indication of starting condition. The two lines were deployed immediately thereafter to the MSC lease site, near Basket Island in Casco Bay, where they hung from a horizontal longline, which was positioned approximately 3 meters below the sea surface. Each line supported 35 pins; Line A had 69 scallops attached (one barb went without a scallop) and Line B had 70 scallops attached. The lines were checked, measured and cleaned on August 31, 2017, February 2nd, 2018, and finally on August 24, 2018, when a destructive sampling took place. The final sample included shell dimensions, and weights for shell, meat, roe and remaining viscera. A temperature logger deployed to the site began sending useable data on 20 February, 2018 to the final date of the experiment, August 24, 2018.
Note: Originally, the project called for scallops to be deployed to sites owned by M. Green and P. Stocks. However, subsequent to the time of the proposal, Green and Stocks jointly began the Maine Scallop Company, so deployments to their respective sites turned out to be a single deployment to the Basket Island site.
Clark's Cove - March 2016 cohort: cleaned vs fouled shells
On 11 March, 2016, five lines of 25 scallop pins each (50 individuals) were drilled at the Darling Marine Center, using a 1.5mm lapidary drill, set into a Dremel drill press. One line was marked with Sharpie permanent marker, on the right valve of each scallop; the valve was dried prior to marking. Scallops were drilled through the byssal notch, so only the top (left) valve was pierced. Shell measurements were taken on the marked line, and HOBO TidBit temperature logger was deployed. Scallop lines were set on a submerged longline that ran between the anchor chains of a mussel raft of the Pemaquid Mussel Company, in Clark's Cove on the Damariscotta River. The longline sat in approximately 65' of water, and was held at about 4' below the surface, by several lobster pot buoys tied to the longline. Additionally, 30 individuals from the same cohort of scallops were used for destructive sampling, yielding information on shell height/length, depth, as well as wet weights of shell, gonad, adductor muscle, roe (if present) and remaining viscera at the start of the study. All weights were recorded on an Ohaus Model CL 201 digital scale, to 0.1 grams.
On May 13, 2016, the long line in Clark's Cove was visited, at which point investigators discovered that the Sharpie marker had not persisted, and was effectively gone from the line we had marked. We then chose one of the existing lines, and re-measured the shell parameters, attaching a small Tyvek tag to one scallop of each pair, so that individuals could be identified. At this point, four of the five lines of pinned scallops were cleaned by hand, to free them of fouling organisms such as tunicates, algae and mussels. One line was deliberately left in its fouled condition, for the remainder of the trial.
Scallops in Clark's Cove were checked on August 30th, and November 2nd, and cleaned each time, except for the unmaintained line. Final sampling occurred on March 25, 2017, at which point both the tagged line and the unmaintained line were measured for all shell parameters, and wet weights of shell, adductor muscle, gonad and remaining viscera. Weights were performed to the nearest 0.1 gram, on an Ohaus Model CL 201 digital scale.
June 2016 Cohort at Darling Marine Center (ABCD position mixed on same lines study)
Beginning on June 16, 2016, three lines containing 20 age-pins each (40 scallops per line) were drilled at the Darling Marine Center, and deployed to a longline within the Darling Marine Center's lease, in Lowe's Cove, on the Damariscotta River. The purpose of the trial was to evaluate drilling position as it related to growth and survival, modeled after the work of Hamada et. al. (2001). Figure 5 shows the drill locations that were investigated. Each line contained individuals of each treatment. For example, Pin 1 would have one scallop each from positions A and B, and then Pin 2 would have one scallop each from positions C and D. Since the pins could be located, and the drill locations were different on each pin, no tagging was necessary to identify individuals. Scallops were cleaned by hand on January 30, 2017, and were retrieved for final sampling on July 12, 2017. At the final sampling, shell dimensions were recorded and individual wet weights were obtained for shell, adductor muscle, gonad (if present) and all remaining viscera. Weights were recorded to the nearest tenth of a gram.
Figure 5. Shell drilling positions, after Hamada et al, (2001). Shown in the figure is the right (lower) valve of the scallop, and position A is located at the byssal notch.
August 18, 2016 Cohort at Clark's Cove
Beginning on August 18, 2016, four lines of ear-hung scallops were deployed on a longline on the Darling Marine Center's aquaculture lease, in Clark's Cove, on the Damariscotta River. Each line contained 30 pins (60 scallops) spaced at 15cm (6") intervals. As with the June 16th cohort, scallops were drilled at differing locations along the hinge, per the work by Hamada et al (2001), although in this cohort, each of the four lines contained only scallops of one drill position. i.e.: one line was comprised completely of scallops drilled at position A, another line was of scallops drilled at position B, etc. Lines were measured and cleaned on December 13, 2016 and May 31, 2017, and were cleaned once more on August 7, 2017. Final sampling took place on Aug 23, 2017, at which point the scallops were cleaned, and records made on shell dimensions and weight, soft tissue weights (adductor muscle, gonad, and remaining viscera), and observations on mortalities or missing individuals. Summary statistics were performed on the survivors, in an effort to describe any effects from the various drill positions on growth, survivorship, and/or loss from drop-off.
ECONOMIC EVALUATION
Based on discussions with producers in the study, and through experiences with producers outside the study, H. Cowperthwaite (Coastal Enterprises, Inc.) developed a cash-flow worksheet was designed in Excel, to encompass the principal categories of expense and income for a scallop farm. The worksheet is a fillable document; items from the various columns and row can automatically populate other segments of the sheet (such as totals), and is easy to modify. The number and type of columns and rows, subject headings and formulas within cells are possible to change, depending on the need. Advice to producers when using this tool will include the recommendation to keep a version as the template, and then to create a separate version as a working document; in this way, the original layout and formulas will remain intact.
This study did not ultimately conduct work at a scale necessary for real evaluation of the profitability of ear-hanging as a production method. However, by outlining the major areas of expense and revenue, and perhaps providing advice to producers about how to measure those parameters, the cash flow document can be a useful tool as each grower makes estimations about economic viability on his/her own site.
The Scallop farm Cash flow template, and all other materials will be posted to the Maine Sea Grant web page devoted to the project, at: seagrant.umaine.edu/extension/northeast-sustainable-agriculture-research-and-education-scallop-project. NOTE: At the time this report is being submitted (Jan, 2020), the web pages concerning shellfish aquaculture are undergoing updates. Upon completion of those updates, all project information will be available at the above link.
RESULTS AND DISCUSSION
Spat collection Year 1
Mesh Type
Mesh type had no effect on the number of scallops per collector (ANOVA, p = 0.07, Fig. 1), the number of other bivalves per collector (ANOVA, p = 0.62, Fig. 2), nor the average shell height of scallops (ANOVA, p = 0.95). Mean numbers of scallops collected at each depth and mesh combination are listed in Table 1.
Table 1. Spat yields by mesh type and depth, 2016-2017 collection season.
Depth
Depth significantly affected the number of scallops and other bivalves per collector (ANOVA, p < 0.001 & p = 0.027), but not scallop shell height (ANOVA, p = 0.91). Additionally, the number of mussels was significantly higher near the surface of the water (ANOVA, p = 0.001, Fig. 3). The mean number of scallops per collector (reported as the average, ± the standard error) was highest at mid depths with ¼” mesh (12293 ± 2671). The mean number of all other bivalves per collector was highest at mid depths (1084 ± 150), and the mean number of mussels per collector was highest near the surface of the water column (393 ± 94). Mussels were analyzed separately because they can cause substantial damage to scallop spat. The mean scallop shell height across all depths was 9.41 ± 0.29 mm.
Retrieval Date
Retrieval date had no effect on the mean number of scallops per collector (t-test, p = 0.24) nor the mean shell height of scallops (t-test, p = 0.49). Retrieval date affected the mean number of other bivalves per collector (t-test, p = 0.004), but because scallops grow slowly the two-week difference in collection date did not have a large effect and we treated all data equally, with four replicates of each factor. Two spat bags were lost during the experiment: one at the surface with ¼” mesh and one at mid depth with ½” mesh.
Note: a poster and update to producers from this segment of the project is given as additional material to this report.
Spat collection Year 2
Forty-six collectors were retrieved, of the 48 deployed. The two missing collectors were both in the bottom position of their lines, and filled with OV-7822. The yield-per-collector and length-frequency analyses that follow were based on two approaches. In the first case, all available bags containing Netron and OV-7822 were used. In the second case, only collectors that had direct counterparts in both materials and depths were used. The sample size for the latter analyses were lower, but they were done to reduce biases, principally by depth.
Yield per collector:
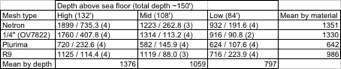
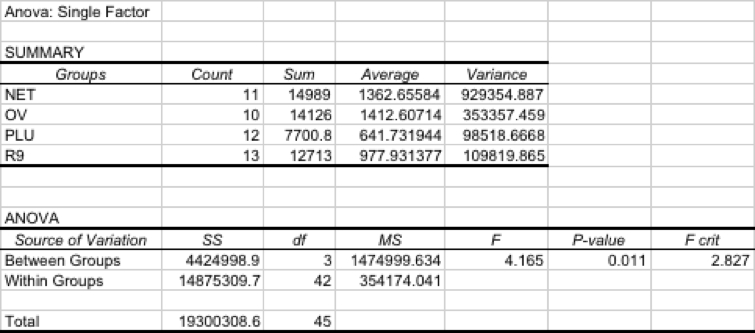
As in the prior trial, the spat collectors highest in the water column performed the best, and those closest to bottom performed the worst. Netron collected slightly more scallops per bag on average than OV-7822 or R9, and much more than Plurima (Table 2). Here we also see the same pattern as in 2016/2017, with spat collectors near the surface outperforming the two lower depths.
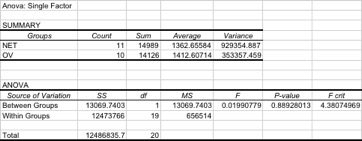
Three subsequent single-factor ANOVA were then run. The first ANOVA compared all mesh types, with all bags included in the analysis, and all depths grouped by mesh type. The low p-value (0.011) indicated a significant difference in collection between mesh types (Table 3). The second and third ANOVA's compared Netron to OV-7822, to compare these two best performers. Initially, we compared OV to Netron with all collectors retrieved. The high p-value (0.889) indicates no significant difference detected between the catches of both mesh types, and results are summarized in Table 4. Next, we included only those collectors that had direct counterparts between netting types. For example, if the Netron LOW position had only three bags returned out of the possible four, then the corresponding OV LOW bag was removed from the analysis. As in the prior analysis, the results of this analysis indicate no different in catch rates between mesh types (p=0.939), given in Table 5.
Table 2: Mean number of scallops per collector, standard error of the mean, and sample size, for scallops collected during 2017/2018 trial. All retrieved collectors are included in this summary.
Table 3: One-way ANOVA for spat per collector, for all mesh types tested; depths are all grouped into the mesh type.
Table 4: One-way ANOVA comparing spat per collector, between the two best performers: Netron and OV-7822. All collectors in each treatment group were included in this analysis.
Table 5: One-way ANOVA comparing spat per collector, between the two best performers: Netron and OV-7822. Only collectors with direct counterparts in both mesh types were included in this analysis.
Length-frequency analysis.
The purpose of this analysis was to examine if one mesh type (Netron or OV-7822) yielded spat of a larger size than the other. To accomplish this, we used the Kolmogorov-Smirnov test, one commonly used to examine frequency of observations; the tests were run in an on-line environment hosted by the College of St. Benedict and St. Johns: http://www.physics.csbsju.edu/stats/KS-test.n.plot_form.html
In the first run, as in the first ANOVA listed above, all bags retrieved for OV and Netron were included in the analysis, and therefore the sample sizes are different (Table 6).
Table 6. Output for sample size, mean and standard deviation from Kolomogorov-Smirnov test, 2017-2018 spat collectors, all collectors included.
N Mean S. D.
Netron 330 10.97 3.16
OV-7822 270 11.72 3.01
The K-S test returned a p-value of 0.021; indicating that the two length frequencies were statistically different, at the 95% confidence level.
In the second run, the only collectors included in the analysis were those that had a direct counterpart on each line: an intact bag with either Netron or OV7822. In this analysis, the K-S test returned a different result (p=0.873, Table 7), indicating that we could not detect a difference in length frequency, between the two substrates.
Table 7. Output for sample size, mean and standard deviation from Kolomogorov-Smirnov test, 2017-2018 spat collectors, using collectors with direct counterparts.
N Mean S. D.
Netron 210 11.71 3.18
OV-7822 210 11.54 3.01
Given the influence of depth on collector yield and the more precise comparison for the second run, we are inclined to conclude that the second K-S test was more reflective of the actual case, and that there was no real difference between the length frequency distributions of scallops caught with the two settlement substrates.
Also notable in the spat collection trials, was that the 2016-2017 spat collection season was much more productive than the 2017-2018 season. This phenomenon was reported along the coast and is reflected in the numbers of this study. For example, in the first year, nine collectors were estimated as having over 10,000 scallops inside, with the maximum estimated at 19,180 individuals - 2.8 liters of seed were captured in that collector alone (OV-7822, mid height). The average for all bags in that year was 6977 individuals per collector (N=46). In the second year, average yield per collector was merely 1076 (N=46), and the largest yield was an estimated 3977 individuals (Netron, high position). Given that the experiments occurred in roughly the same area at roughly the same time of year, we view this as an indication of year to year variability, and the value of longer-term datasets. In addition, it gives us a 'sense of the possible' - that nearly 20,000 scallops could be captured from a single collector is an encouraging observation.
Ear Hanging Trials
Maine Mariculture Company
The two lines originally hung at the Pleasant Island site of the Maine Mariculture Company were stocked with scallops from the same nursery cohort, which had been raised on site in bottom cages. The two lines supported 34 pairs of Itimai-ake-drilled scallops ('single drilled'), and 32 pairs of Nimai-ake-drilled ('double drilled') scallops. Shell heights for both groups were slightly different, 57.6mm for single-drilled and 55.4mm for double-drilled: an F-test for homogeneity of variance gave a p-value of 0.31, indicating similar variances of the mean, and a subsequent T-test gave a two-tailed p-value of 0.21, indicating that the two means were significantly different at alpha=0.05.
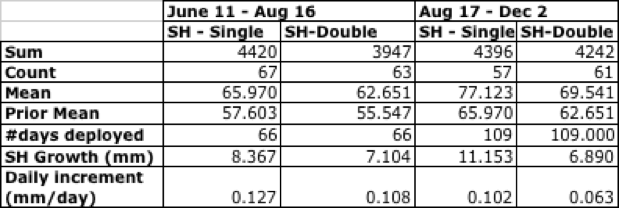
Scallops were checked on August 16, 2016 and December 2, 2016, at which point the scallops were cleaned of most bio fouling and measured again for shell dimensions. The following summary refers to Shell Height however, as the most common method of describing scallop size. Growth was higher during the summer months, and single-drilled scallops grew more quickly over both time intervals, as shown in Table 8; note that this summary includes all scallops of both kinds that survived until Dec 2.
Table 8. Summary growth statistics for single- and double-drilled scallops, Maine Mariculture Company.
During the winter of 2016/2017, all lines on the farm were lost and the farm subsequently shut down, so the December sampling was the final one.
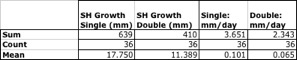
Next, we examined the growth of only those scallops that had a direct counterpart at the Dec. 2 sampling, or 175 days post-deployment. For example, if individual #15t (pin 15, tagged) was present for both single- and double-drilled scallops, it was included. In this summary, we identified 36 individuals of both types (72 individuals total) that met this criterion, and summarized them in Table 9.
In keeping with the prior observation where all scallops were included, the single-drilled scallops grew much more quickly, both in terms of overall growth (in mm) and growth rate (mm/day). The final measurements for individual growth were highly significant according to a Paired-Sample T-Test (p<.001), as were the final growth rates (p=0.001); both favoring single-drilling.
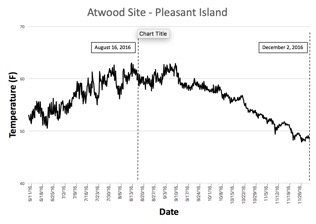
Temperature - Atwood Site, Pleasant Island
Data retrieved from the temperature logger are displayed in Figure 5. Logging began on June 11, 2016 and ended on December 2, 2016; four readings per day were taken at regular 6-hour intervals. Mean temperatures before and after the August 16th visit were nearly identical; 56.83F prior, and 56.01 afterward. Without chlorophyll and turbidity data, we speculate that the higher growth in the first period was due to better food resources, although rough weather later in the year could certainly have been a factor as well.
Figure 5. Temperature plot for Pleasant Island, Maine Mariculture Company
Wild Ocean Aquaculture
The replacement line deployed on August 8, 2017 had 69 live individuals at the end of the experiment, of the original 119, or a survival rate of 58.0%. Most of the losses appear to be drop-offs, since 39 individuals were missing: either the shell or the age-pin had broken.
The line brought to us for measurement on 2/2/18 was of unclear origin, so the numbers that follow reflect findings from the one chance we had to measure the 8/8/17 line, at the end of the experiment. Mean total growth over the 357 days of deployment was as follows: Shell Height - 23.1mm, Shell Length - 22.6mm and Shell Depth - 6.9mm. Daily incremental growth for SH averaged 0.065mm/day, and the maximum SH growth seen in an individual was 46.3mm, or 0.130mm/day (1.86in/yr).
The final group had 67 individuals with observable gonad material; 36 males and 31 females. Mean wet weights of dissected tissues were as follows; percentages are given to two decimal numbers, which may result in rounding error (Table 10)
Table 10. Wet weights of shell and soft tissue, for cohort at Wild Ocean Aquaculture, deployed 8/8/17.
Wt (g) % of total wet wt.
Adductor Muscle 16.3 16.01%
Gonad 11.5 11.29%
Viscera 23.3 22.89%
Shell 50.7 49.80%
Mean whole wet weight 101.8 (0.2 lb)
Since scallop meats are sold by the number to a pound (referred to as X-count), the 16.3 g. average meat translates to 27.8 meats to the pound, or '28-count.' At the 2018 statewide average ex-vessel price of $10.54/lb, each scallop meat was worth $0.37.
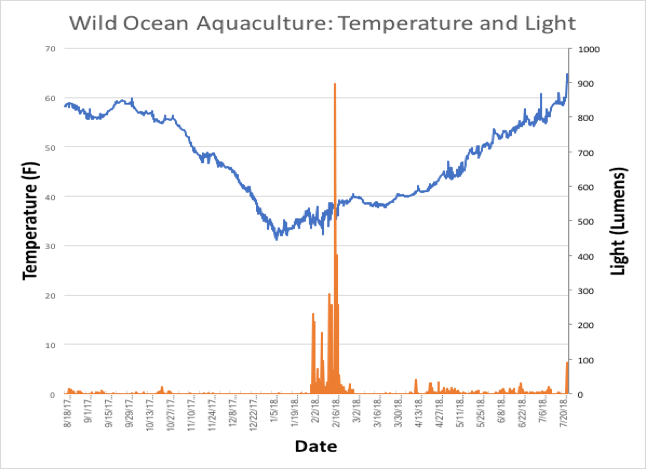
Temperatures at the site varied around the 50F mark; approximately 6 months of the sampled year was spent above and below 50F, and only rarely above 60F. Minimum temperature on site occurred on 1/1/18, at 31F. Daylight was measurable with the datalogger giving return values, although there were regular distortions of the data between 1/30/18 and 2/21/18. The cause for this is unknown, but clearly the data during this period is unreliable. It may be better in future to have a light logger attached to the hanging line or a separate dropper, rather than directly to a scallop production line. Temperature and light data are shown in Figure 6, below.
Figure 6. Temperature/light plot from Wild Ocean Aquaculture, 8/8/17 cohort.
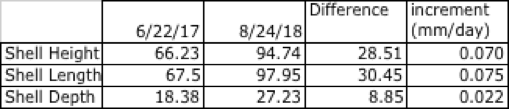
Maine Scallop Company
Two lines of pinned scallops were deployed on 6/22/17. Both lines contained 35 pins, but one pin on Line 1 missed a scallop (N=69), whereas Line 2 was fully stocked (N=70). Mean shell heights for lines 1 and 2 were 66.28mm and 65.1mm, respectively. A two-sample z-test for means indicated no significant difference between the SH's of both groups at Time 0 (one-tailed p-value = 0.0554).
Lines were checked on 8/31/17, 2/2/18, and 8/24/18. On the final date, it was discovered that Line 2 had become wrapped around the head rope of the long line, and all scallops had been lost. Thirty-nine individuals survived on Line 1, or 56.5%. Of these, 13 were missing, and 11 were empty shells.
A summary of growth for all shell dimensions measured, over the 408 days of the study is given in Table 11.
Table 11. Shell growth in mm over the term of the study, and daily growth increment for Line 1, Maine Scallop Company.
Mean wet weights for shell and tissues, and their relative proportions of total wet weight were as follows. Total wet weights are given to two decimal numbers, which may produce rounding error (Table 12).
Table 12: Mean wet weights for shell and soft tissue, Maine Scallop Company.
Mean wet weight (g) % of total wet weight
Adductor Muscle 20.20 15.69%
Gonad 17.81 13.84%
Viscera 29.74 23.11%
Shell 60.75 47.20%
Total 128.71
At 20.2 grams, the average adductor muscle was 0.71 ounces, or 22-count. At the 2018 statewide average ex-vessel price of $10.54/lb, each scallop meat was worth $0.47.
Clark's Cove March 2016 trial - cleaned vs. untended/fouled line
For this analysis, we make the following comparisons
- Growth characteristics of the tagged line between the time it was tagged on 5/13/16, and when it was terminated on 3/25/17, or a deployment of 316 days.
- Growth characteristics of the tagged line vs. untended line, over the length of the study: 3/11/16 to 3/25/17, or a deployment of 379 days.
The initial sampling of 30 individuals on 3/11/16 yielded the following group averages:
SH: 48.2mm, SL: 48.0mm, SD: 10.7mm
Mean tissue wet weights and percentage of overall wet weights are listed below, for the 30 individuals on 3/11/16. Gonad development was at such a light stage as to be nearly negligible, so the weights for viscera include gonadal tissue as well:
Adductor: 1.27g
Viscera: 4.51g
Shell: 5.27g
Total: 11.05g
Tagged line growth:
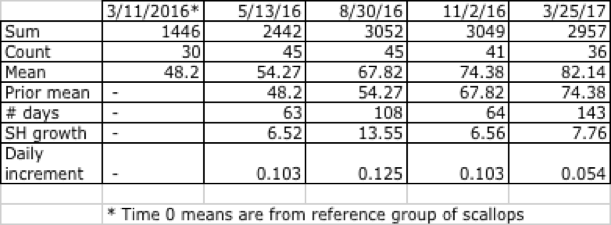
Table 13 summarizes growth and daily increment for shell height, on the tagged line between 3/11/16 and 3/25/17, or 378 days deployed. Note that the Time 0 averages on 3/11/16 were based on the scallops measured and destructively sampled, referred to in the above paragraphs. Subsequent measurements were based on the tagged scallops themselves.
Table 13. Summary shell height statistics, Clark's Cove location, tagged (cleaned) line.
Tagged vs. fouled/untended line:
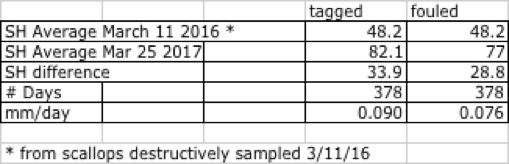
Scallops on the tagged line were cleaned during checks for measurement, and demonstrated higher growth than did the untended/fouled line. Final shell heights for scallops on the tagged line vs. the fouled line are given in Table 14. Note that the initial shell height of 48.2mm was derived from the destructively sampled lot of representative scallops on 3/11/16.
Table 14 - Final shell height for cleaned vs. fouled ear-hanging lines, Clark's Cove Road.
Tagged vs. fouled - weights
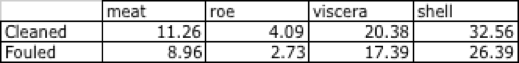
The tagged line outperformed the fouled line, in terms of wet weights of all tissues measured, illustrated in Table 15.
Table 15 - Wet weight in grams for various tissue compartments: fouled vs. cleaned line, Clarks' Cove: 3/11/16 to 3/25/17.
Meat weights for clean and fouled lines in ounces were 0.40 and 0.32, or 40- and 50-count respectively. At the 2018 statewide average ex-vessel price of $10.54/lb, these were worth $0.26 and $0.21 respectively.
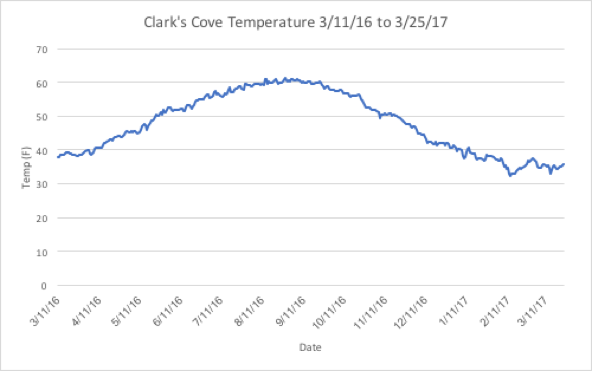
Temperature:
Figure 7 below gives the temperatures encountered on side during the length of the study at Clark's Cove.
Darling Marine Center, June 2016 Cohort
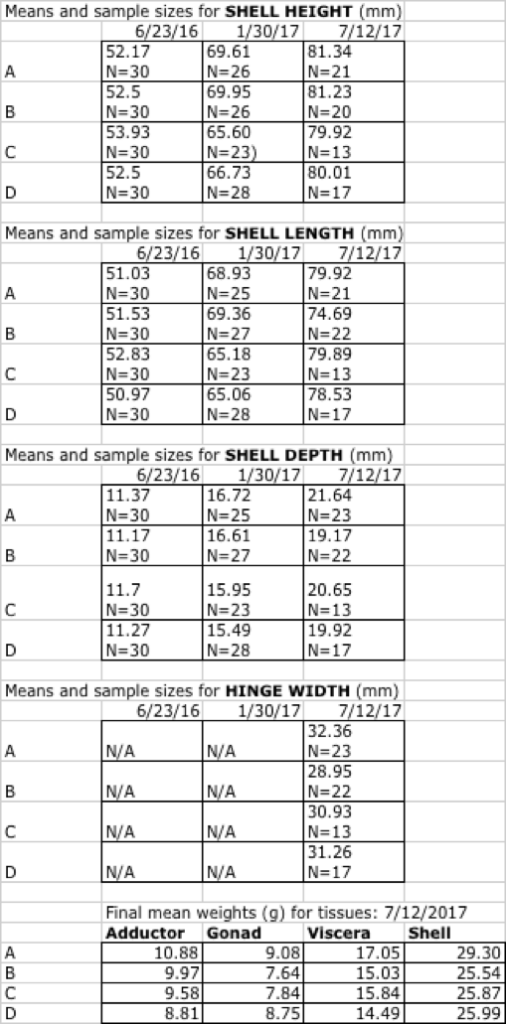
The three lines deployed on June 23, 2016 were cleaned on December 13, 2016 (day 142), measured on January 30th, 2017 (day 190) and measured again at the end of the experiment on July 12, 2017 (Day 385), at which point they were dissected and wet tissue weights recorded. In this experiment, we also recorded the width of the hinge, as measured parallel to the hinge itself to the anterior and posterior margins.
In all shell dimensions (SH, SW, SL, Hinge width - HW), drill position A outperformed all other drill positions. This trend held true for tissue weights as well, with position A demonstrating higher mean wet weights than all other positions for the adductor, gonad, viscera and shell. Due to the relatively low sample sizes for each treatment by the end of this experiment, Table 16 reflects the simple means for each treatment, with all of the three lines combined.
Table 16. Shell growth and final weights for shell and soft tissues, for June 2016 cohort of scallops at the Darling Marine Center.
When expressed as daily growth increment (mm/day) over the length of the study, the four groups were: A = 0.076, B =0.075, C =0.067, D=0.071. At the 2018 statewide average ex-vessel price of $10.54/lb, adductor muscles alone were worth $0.25, $0.23, $0.22, and $0.21 each, respectively.
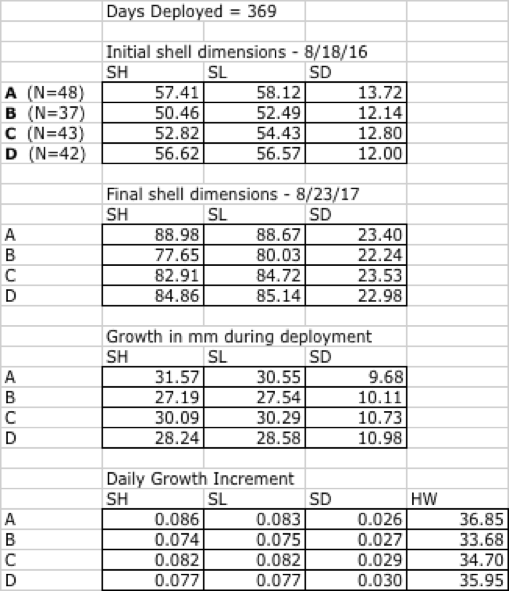
August 18, 2016 cohort - Clark's Cove
In this experiment, scallops were deployed for a total of 369 days. Drill position A showed greatest shell growth in SH, SH and hinge width (HW) than other treatments, although position A returned lowest rates in the SD dimension. Position also had higher survivorship than other treatments, at 80%, or 48 of the original 60 scallops surviving to the end of the experiment. Shell dimension data for the duration of the experiment are given in Table 17.
Table 17 - Shell dimensions and growth increment by drill position, August 2016 cohort, Clark's Cove.
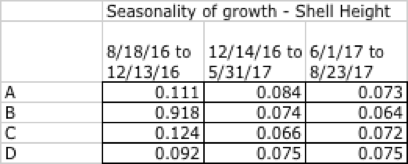
Growth during this experiment displayed both a seasonality and an artefact of high fouling rates. Table 18 gives the daily growth increment for the three intervals between the first and last field visits, using SH as the indicator of growth.
Table 18: Seasonality of growth by shell height, August 2016 cohort, Clark's Cove.
No tissue weights are available for this cohort, as this group was not dissected at the end of the experiment.
Spat Collection
The two seasons of spat collection showed fairly convincingly that OV-7822 mesh performed as well or perhaps even better than Netron in some instances, and for a fraction of the cost. Twenty-one square feet of material was sufficient for good catches of scallops and given that it comes in a flat sheet, was easier and more time-efficient to load into spat collectors than Netron was, and it is much easier to empty and clean. The easier handling of the flat net should translate into less time spent, and therefore lowered labor costs. This experiment led to another one that is underway in the 2019-2020 collection season, which is to evaluate the amount of stuffing in a spat bag, and the effect on number and size of retained scallops.
Ear-hanging: growth and mortality
Growth of scallops via ear-hanging was inconsistent by site and within site, but did show some bright spots. There appears to be a strong seasonal component to growth, which is not unexpected given shellfish physiology in general and the effects of temperature on metabolism, although future work should also include an examination of feed type and availability, over time and depth. Combined with more frequent measurements, such work should yield sound recommendations for site selection.
The size (and speculated value) of adductor meats alone was lower than hoped for. This may be in part a function of the relatively small size attained by the scallops over time, given the rapid increase of scallop meats past approximately 100mm. In addition, the fouling encountered during this study was considerable, and more than anticipated. Such fouling loads could easily have limited growth and condition, including weights of individual adductor muscles.
It was encouraging that the results of the drill position experiments were not quite as drastic as perhaps expected, and that with careful placement, drilling through both shells of the scallop is a possible option. This may apply particularly to sites that are more exposed or where tides are stronger, and where longlines would consequently experience greater vertical and horizontal motion. That said, drop-off of scallops and mortality from all sources will certainly need more attention, and future studies should keep close track of the reasons for mortality, and their relationships to husbandry interventions, or the biophysical factors at play.
Long line siting, deployment and management will need to improve over time, and if ear-hanging lines are deployed to raft platforms, then good care will need to be paid to spacing. In both instances, scallops were lost when lines crossed, in particular with long line setups. This issue seems likely to resolve over time, as growers learn more about how ear-hanging lines perform on their sites.
Recommendations:
Some observations and recommendations from this study are as follows, given in no particular order of importance.
- Ear hanging lines may need a small amount of weight attached to the bottom of the line; perhaps 1 kg. This should limit crossing of lines and loss of product.
- Excessively hot or cold days should be avoided for husbandry actions, especially if it's windy.
- Where possible, keep scallops in the water; when drilling, measuring or cleaning, try to create a work flow where scallops are exposed to air as little as possible.
- Fouling management will be a major component of successful ear-hanging. Type of fouling, timing of settlement, and evaluating the negative impacts of each fouling species will be helpful in strategizing for most effective treatment.
- In spat collectors, it is frequent to find many small scallops at the bottom of the collector bag; from 1-3mm. These are worth saving; they will grow once given adequate space and feed. Conversely, releasing these to the bottom may also have a benefit to the wild stock. In either case, spat collectors are worth cleaning fully, and removing all possible catch.
Literature Cited:
Anonymous, 2018. Maine Department of Marine Resources Landings Statistics. https://www.maine.gov/dmr/commercial-fishing/landings/index.html
Hamada, T., N. Yamashita, T. Watanabe and S. Natsume. 2001. Drilling position of the ear affects growth and mortality of scallop (Patinopecten yessoensis, Jay) in ear-hanging culture. Aquaculture 193, 249-256.
Kuenstner, S. 1996. Polyculture of sea scallops suspended from salmon net pens. Final report to NOAA Saltonstall-Kennedy Program, Northeast region, Award #NA46FD0327. New England Fisheries Development Association, 91p.
Pottle, TJ., and M. Hastings. 2001. Sea Scallop Demonstration Project, Final Programmatic Report. Submitted to National Fish and Wildlife Foundation. 10p, with appendices.
Smolowitz, R. 1999. Sea scallop enhancement and sustainable harvesting. Final report to the Saltonstall-Kennedy Program, Northeast Region, Award #NA66FD0027. Westport Scalloping Corporation, 490 p.
Additional Reading:
Beal, B.F., M. Hastings, W. Hopkins, S. Inches, D. Morse, K. Porter, T. Pottle, S. Rappaport, L. Taylor, J. Trenholm. 1999. The culture of Japanese scallops (Patinopecten yessoensis). Report of the Maine delegation to Aomori Prefecture, Japan, May 14-21, 1999. The Maine Aquaculture Innovation Center, 49p.
Couturier, C., Dabinett, P. and Lanteigne, M. 1995. Scallop Culture in Atlantic Canada. In Cold-Water Aquaculture in Atlantic Canada. Edited by A. Boghen. Print Atlantic, Moncton. pp. 297-340.
Davidson, L-A., R. Nowlan and M. Niles. 2014. Open-ocean sea scallop (Placopecten magellanicus) culture trials in Chaleur Bay, Canada: comparing culture gears and husbandry practices. Canadian Technical Report of Fisheries and Aquatic Sciences #3121. 26p.
Stewart, P.L. and S.H. Arnold. 1994. Environmental requirements of the sea scallop (Placopecten magellanicus) in eastern Canada and its response to human impacts. Can. Tech. Rep. Fish. Aquat. Sci. 2005: 1-36.
Shumway, S. Editor. Volume 40. Scallops: Biology, Ecology, Aquaculture and Fisheries. Elsevier, 1214p.
Growth increment conversions: millimeters to inches
Education & outreach activities and participation summary
Participation summary:
The work outlined in this report has been an exceptionally productive method to get producers, students and others involved; for training, student projects, internships, etc. Education, training and outreach efforts associated with this project are listed below.
A project web page has been created for this project, at http://www.seagrant.umaine.edu/aquaculture/resources-for-shellfish-growers/species/scallop/ear-hanging . Please note that all the pages within the 'Resources for Shellfish Growers' rubric are currently unavailable on the MESG website; they are in the process of being updated and will be available when updates are complete.
Presentations
Presentations/workshops about this project occurred at:
2017 Northeast Aquaculture Conference and Expo (NACE)
Providence, RI. Attendance ~ 45
2017 Maine Fishermen's Forum
Rockport, ME. Attendance - 65
2017 National Shellfisheries Association Annual Meeting
Knoxville, TN. Attendance ~ 45
2017 Aquaculture in Shared Waters, aquaculture training program. Scallop Farming.
Ellsworth, ME. 15 students
2017 International Pectinid Workshop, Portland, ME. 170 participants.
2018 Maine Fishermen's Forum
Rockport, ME. Attendance ~ 80
2018 United States-Japan Panel on Natural Resources (UJNR), Aquaculture Section.
Mystic CT. Attendance ~ 50
2018 Aquaculture in Shared Waters – Scallop Farming. Bath, ME (25 students) and Machias, ME (15 students)
2018 National Shellfisheries Association Annual Meeting
Seattle, WA. Attendance ~ 55
2019 Maine Fishermen's Forum
Rockport, ME. Attendance ~ 70
2019 Aquaculture in Shared Waters – Scallop Farming. Brunswick, ME. 35 students
2019 Northeast Aquaculture Conference and Expo (NACE)
Boston, MA. Attendance ~ 50
2019 Pacific Coast Shellfish Growers Association Annual Meeting
Portland, OR. Attendance ~ 70
2020 Maine Fishermen’s Forum (planned)
Rockport, ME
2020 National Shellfisheries Association Annual Meeting (planned)
Baltimore, MD.
2020 Aquaculture in Shared Waters (planned) – Scallop Farming. Brunswick ME and remotely at Hutchinson Center, Belfast ME. 55 students.
There are a number of informal opportunities that arose out of this project, to introduce growers, students and others to the project, and to the processes of scallop culture that we are investigating. In many cases, these became hands-on learning opportunities; measuring and dissecting scallops, drilling and deploying ear-hanging lines, etc. Some examples include:
2016 Governor Angus King visit to Damariscotta River; tour of scallops at Pemaquid Mussel Farm aquaculture site.
- Mr. H. Sugiyama, inventor of scallop farming equipment and founder of Mutsu Kaden Tokki Co., visits Damariscotta River, and tours Pemaquid Mussel Farm site, where scallops from this project are being grown.
2016, November 2. Local growers visit DMC to learn about scallop farming; review drilling and the production lines in this study.
2016, May 2. Ken Beebe of Hog Cove Oyster Farms, assisting with scallop farming: thinning, grading, ear-hanging
2018 Essex High School at Darling Marine Center. 18 students: measuring, drilling scallops, aquaculture discussion.
2018, D. Morse participates in a SARE workshop at Wolfe's Neck Farm, Freeport ME, for SARE staff and other investigators. Methods and findings of spat collection segment are presented, approximately 50 attendees.
2018, July 1. Scallops from the project given as side-project to Dillon Shaw, of Dewey’s Shellfish, Damariscotta River.
2019 Essex High School at Darling Marine Center. 15 students: measuring, drilling scallops, aquaculture discussion.
There were two interns who participated in this study. In 2017, Melissa Britsch was heavily involved in fieldwork and data analysis/presentation. Her work resulted in a poster, and a presentation at the Darling Center Science Symposium (attached files). In addition, she took the lead in developing an update document, that summarized spat collection work to date; this was sent to all scallop producers in the state, in 2017. Ms. Britsch is now a dual-degree Master’s student at the University of Maine.
In 2018, Mitchel Stewart interned at the Darling Center and was also involved in the SARE scallop project. He assisted with spat collection fieldwork and data summarization, as well as some field visits on the ear-hanging trials. Mr. Stewart graduated the University of Maine in 2019.
We regard the interns' participation in the project as an important part of their exposure to applied, collaborative research, and in gaining valuable experience in data collection, summarization and analysis, reporting and presentation of the results.
Instructional document(s)
The principal outreach documents from this project are two. First, H. Cowperthwaite and D. Morse created an iBook chapter, that covers the scallop farming process in comprehensive fashion, and which has been delivered twice, through the Aquaculture in Shared Waters program. A PDF of the document is attached here. Secondly, a fact sheet has been prepared, to be delivered in electronic or printed formats. This 16-page document is fully written and only awaits layout and posting to the Maine Sea Grant website. Draft fact sheet is attached here.
Note: All materials related to this report we be posted to the web page devoted to the topic, at: seagrant.umaine.edu/extension/northeast-sustainable-agriculture-research-and-education-scallop-project
As of the time of submission for this report (January, 2020) all pages on shellfish aquaculture are temporarily off-line, while they are updated. This project is included in that group, but the page will become publicly viewable when updates are complete.
Learning Outcomes
This project took place within the context of other work, all aimed at improving our understanding and productivity of spat collection and aquaculture production of sea scallops. Within that context, it's a bit hard to draw out what learning can be attributed specifically to this project, although there are a few things. For example:
- The location of the hole used in ear-hanging is relevant and important. We demonstrate better survival and growth with alternate locations that we expected at the outset, but still note that the recommended method - drilling inside the byssal notch - provides best result. Growers using the ear-hanging method in Maine recognize this.
- Some commercially-available agricultural nettings will function as well as the industry standard, and at considerably less cost. Some growers are already using this material, although since the standard material is already owned by some farmers, they will probably continue to use that until it is no longer serviceable, and will then switch to the ag netting.
- This project was enormously helpful in getting students involved in the research, and perhaps more to the point, getting other researchers involved. This work formed part of the basis for subsequent grant proposals, one in particular which was very successful, and has been vital in the development of several young scientists, helping them see the methods and benefits of applied research, and getting them important exposure to industry members and their lives and work.
- Overall, farmers are much more knowledgeable about ear-hanging now, than they were at the beginning of the project. Again, it's hard to say that this is due exclusively to the current project, but this project certainly added to the general knowledge of scallop producers.
- Farmers have better knowledge now of growth rates and influence of seasonality and production type on the yield of scallops, meaning shell growth and the weight of the soft tissues. These numbers will be very important as producers plan their harvests, as they will want to maximize weight harvested.
Project Outcomes
This project had quite an impact; well over its funding level. Farmers are more informed about the practices and successful methods for spat collection and ear-hanging, and there are many individuals who have gotten exposure to the work, via opportunities in the field, or through outreach activities. In general, the project added to the strong relationships involved in collaborative research on scallop production in Maine.
In retrospect, there was a lot to consider in this project. The spat collection work involved two seasons instead of one, and that both made our results more robust, but allowed us to continue to explore new materials and to deepen this area of inquiry. As a result - and not reported here - is that in the 2019-2020 spat collection season, D. Morse and N. Perry continue to work together, to examine the influence of the amount of substrate in a collector on the resulting catch. The current project has paid off in helping us continue to optimize spat collection, which will accrue to the benefit of producers and scientists statewide and elsewhere.
The trials of ear-hanging turned out to be somewhat haphazard at Clark's Cove, and somewhat limited at industry sites. In the former case, the haphazardness arose from taking advantage of opportunities when students or industry members visited the Darling Center, and we took it upon ourselves to set out more ear-hanging lines. In the latter case, it seems wise to weight ear-hanging lines, so that they do not tangle. In addition, it's clear that it will take some dedicated space by industry partners to set experimental lines aside from their production areas, and that also it's simply a matter of learning; we are all learning how to set and operate longlines and ear-hanging droppers more effectively.
More frequent visits are recommended for data collection and for cleaning. This will get at the influences of seasonality much more effectively, and as we saw, biofouling can be a significant and severe obstacle. More frequent observation and cleaning should yield higher growth rates than we observed.
The project was a standout in its attraction for students and visitors. The hands-on nature of the work, and the fact that the scallop industry is so new, was a real plus in terms of getting others involved. All those working with scallops in Maine are using this dynamic to broaden the participation from students, other industry members, scientists and members of the interested public. It's a real win.