Final report for ONE19-343
Project Information
Anaerobic soil disinfestation (ASD) is gaining attention in the US and has proven effective against soilborne pathogens in California and Florida where growing seasons are long and warm. This project was the first to evaluate the effectiveness and profitability of ASD to control Rhizoctonia solani on lettuce in Vermont field conditions. In ASD, a carbon source is added to soil, watered heavily, and tarped until soil becomes anaerobic. This project tested three carbon sources that are commonly used by Vermont farmers (compost, poultry manure, and cover crop) compared to an unamended control. We quantified disease incidence and severity, marketable yield, and costs associated with labor and materials. We demonstrated that it is possible to achieve temperature and anaerobic condition requirements of the ASD method in Vermont, where temperatures are relative cool and seasons short. Unfortunately, adequate disease suppression has yet to be observed as a result of the practice in the Northeast. Disease severity and marketable yield were similar for all carbon treatments and unamended controls. Furthermore, none of the treatments tested showed profits that exceeded the costs of implementation (supplies, labor). Higher value crops (such as berries or orchards) and or no-till systems may be better suited for ASD. Our survey of the Northeastern agriculture community showed that 55.5% of the respondents are already utilizing tarping for the purpose of weed control, which makes the practice of ASD more affordable to farmers.
This project tested whether Anaerobic Soil Disinfestation (ASD) can be used to control soilborne pathogen R. solani in Vermont field conditions. There were three components:
- To provide several potential management options, we tested multiple carbon sources (compost, poultry manure, and cover crop) that are available and already used by organic farmers against a non-amended control;
- We assessed efficacy over two years to answer the farmer question “how often do I have to do this?”
- To assess practical and financial viability, a cost benefit analysis was performed at a micro-scale farm and a > 10 acre farm in collaboration with the farm partners.
If the proposed project demonstrates that ASD successfully controls R. solani in field-grown crops over multiple years at multiple farms, it will offer robust evidence to growers that ASD can be used within existing management strategies to control soilborne pathogens. It would provide justification for investigation of ASD in high tunnels and greenhouse soils. Further, it will enable growers to make informed financial cost benefit decisions about the use of this practice on their farm. Finally, it provides a method to manage a persistent soilborne pathogen that meets organic certification regulations.
Rhizoctonia solani is an aggressive pathogen and a saprophyte that can survive as sclerotia under adverse environmental conditions. Up to 70% crop losses on lettuce is caused by R. solani on field-grown lettuce in the US. Control is difficult due to its extremely wide range of plant hosts including Solanaceae, Fabaceae, Asteraceae, and Brassicaceae. Bottom rot of lettuce, caused by R. solani, is a recurring problem for vegetable growers in Vermont, reducing field lettuce yields by 10-50%, and up to 80-90% for lettuce in over-wintered high tunnels. It is a problem exacerbated by increasingly warm and wet fall conditions.
Organic farmers lack proven and effective fungicides to manage the disease and a limited toolbox of management practices such as crop rotation and increasing airflow around plants is relied upon to prevent damage. There is minimal genetic resistance to R. solani and a limited choice of seed varieties that meet the organic certification standards of seed production and pelleting in New York and Vermont.
Market farms on the micro-scale are increasing in popularity. These farms rely on quick succession of high margin leafy crops such as lettuce to be profitable with a limited land base. Crop rotation is difficult because crop groups are limited and space is less likely to be rotated into a non-host crop or fallowed. Small scale growers often extend the season into the early spring and late fall, overwintering greens in tunnels. This combination of factors (limited rotation, extended season, succession of lettuces, higher humidity in tunnels) is prime opportunity for colonization by R. solani, making new growers at this scale a potentially vulnerable population.
Anaerobic soil disinfestation (ASD) is a treatment to reduce R. solani pressure in soil. ASD is implemented through addition of an easily degradable organic matter (labile C) followed by irrigation saturation and covering with an oxygen-impermeable tarp. Soil microbes consume available oxygen and anoxia shifts the balance toward fermentation and facultative anaerobes. Most pathogens die from oxygen deprivation, but others are sensitive to volatile organic compounds produced by facultative or obligate anaerobic microorganisms. To our knowledge, the proposed study will be the first field test of ASD on soilborne pathogens of vegetables crops in the northeastern US. If ASD works as an effective control in northeast growing conditions, it would be a low-cost option that could be easily integrated into existing management strategies that include the practice of tarping.




Cooperators
Research
The experiment was arranged in a randomized complete block design with four blocks at each of two farms with a history of bottom rot of lettuce. Block location within a field was based on grower and researcher observation of disease incidence and severity. Plots were 1.27 x 3.8 m (4.17’ x 12.5’) with a 0.3 m (1’) buffer between plots to prevent interplot interference. Treatments included commercial compost (Black Dirt, Stannard, Vermont), poultry manure, and farm grown chopped cover crop all compared to two types of unamended controls (one with and the other without a tarp). The three amendment treatments were chosen, respectively, to represent the farmer’s current practice, an increasingly popular amendment chosen for its low cost, and a common and inexpensive cover crop used in the region. Application rate was calculated based on carbon content of each material with a target of 4 kg/ha (1.78 tons/acre) (Shennan et al. 2018). Carbon material was incorporated to a depth of 13-25 cm (5 to 10”) with a rototiller followed by irrigation with 5 cm (2”) water. The area was covered with a silage tarp and incubated three weeks.
Some adjustments to treatments were made in response to supply chain disruptions by the COVID-19 pandemic. For example, we were unable to establish the cover crop in 2020, and replaced that treatment with cracked corn animal feed (Blue Seal, Brandon, Vermont), which has a similar carbon to nitrogen ratio to a rye cover crop. In 2021, we incorporated a rye cover crop grown at Riverside farm, and an oat and field pea cover crop grown at Riverberry farm, as originally proposed. Poultry manure was sourced from Giroux (Chazy, New York) in 2020 and a blend of two sources (Espoma Company, Millville, New Jersey, and North Country Organics Bradford, Vermont) in 2021. Lettuce seeds were sown in Fort Light potting soil mix (Vermont Compost Company, Montpelier, VT) and reared in a greenhouse at University of Vermont for 30 days. Lettuce plants were transplanted into the field 1-2 days (2020) and 1 week (2021) after tarp removal. Dataloggers with sensors were installed at each farm to measure soil temperature, water potential, and redox potential every hour from the time of installation until the tarp was removed at the end of the ASD treatment. We lost some sensor data in 2020 due to battery failure but corrected the problem in 2021.
The relative success of these media were measured as disease severity, disease incidence, and the amount of marketable heads of lettuce produced in each treatment. The plants were individually assessed weekly for disease symptoms using a rating scale of 0-6 (0 indicating a healthy plant, 1-5 representing increasingly severe symptoms, and 6 indicating a dead plant). At the time of harvest (5 weeks post-transplanting into the field), each plant was determined to be either marketable or unmarketable based on guidance from collaborating farmers to match the quality required by their purchasers. Data were analyzed using a one-way analysis of variance.
An economic case study was conducted to ascertain the economic viability of the treatments tested. This study examined the additional costs associated with implementing each treatment in comparison to standard operating procedures and costs. The cost of the treatments was calculated based on purchase prices and the quantity of product used (based on manufacturer recommended rates).
In 2021, both sites demonstrated a shift from an aerobic to anaerobic soil state immediately following ASD installation and remained anaerobic for the desired three week period (Fig. 1). Anaerobic conditions were more successful in 2021 than 2020 when we replaced the initial sprinkler irrigation event followed by hand-watering to a drip irrigation system that allowed for pulses of irrigation to be applied throughout the treatment period.
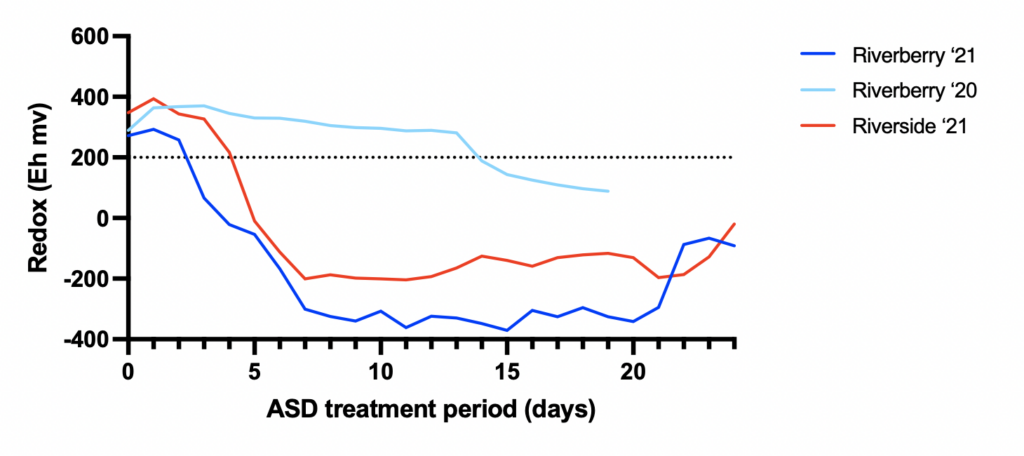
According to the literature, for ASD to successfully control soil pathogens (Verticillium, Fusarium), there needs to be a cumulative level of anaerobic conditions (measured as Eh millivolts (mV) hours below 200 mV) for a minimum of 50,000 hrs during the treatment period. In 2020, Riverberry accumulated 15,829 mV hours during the treatment period, which is insufficient for significant disease control. However, in 2021 both Riverberry and Riverside farms exceeded this minimum threshold to achieve disease control. Riverside accumulated 57,399 mV hours, and Riverberry 116,208 mV hours. The difference between the site’s cumulative mV hours in 2021 is likely due to physical characteristics of the soils at each farm. Riverberry soils (silt loam) had a higher clay content than Riverside soils (sandy loam) which allow for greater water retention contributing to stronger anaerobic soil conditions.
ASD effectiveness appears to be associated with sustaining temperatures between 20°-25°C (68°-77°F) beneath the tarp during an ASD treatment to suppress soilborne fungal pathogens including Rhizoctonia solani, Fusarium oxysporum and Verticillium dahliae. Mean soil temperatures at Riverberry Farm were 27°C and 26°C in 2020 and 2021, respectively. In 2021, Riverside’s mean soil temperatures were 23.5°C (Riverside data from 2020 was excluded due to datalogger equipment malfunction). Attaining these temperatures was a critical component of this trial, as ASD has never been implemented in the Northeastern United States in a field setting partly because of the cool climate, especially when compared to states like Florida and California where ASD is more frequently utilized that have much warmer temperatures. This trial demonstrates that summer conditions are warm enough in Vermont to achieve the necessary temperatures associated with suppressing soilborne plant pathogenic fungi.
Mean disease severity was greatest for the cover crop and least for the poultry manure treatment but not statistically significant (P = 0.7515). Numbers of marketable heads of lettuce were inversely associated with disease severity and not statistically significant among treatments (P = 0.9598).
Economic analysis revealed that it would cost the grower an additional $91.42 above standard operating costs to use a compost-based ASD treatment for a 200-square foot area (0.005 acre, equivalent area needed to grow 144 lettuce plants). Similarly it would cost the grower an additional $95.23 for ASD with poultry manure, $76.36 for ASD with cover crop, and $82.04 for an ASD treatment that was only balanced for nitrogen content. Irrigation costs included only drip tapes and the fuel costs to run the water pump. Other irrigation supplies/costs (disc filters, emitters, pressure regulators, etc.) were omitted based on the assumption that a commercial grower would likely already own these items. Cover crop estimates of seed based on 60-lbs/acre rye broadcast rates (Schnitkey, 2018), and poultry manure prices were based on poultry manure produced by North Country Organics. Labor rates include (as needed by treatment): spreading carbon sources, irrigation set up (installing water pump, drip tape install), tarp application and removal post-treatment. Labor hours were calculated as the mean amount of time between the farms to both install the treatment and remove the tarp upon completion.

Literature cited
Shennan, C, et al. “Anaerobic Soil Disinfestation Is an Alternative to Soil Fumigation for Control of Some Soilborne Pathogens in Strawberry Production.” Plant Pathology, vol. 67, no. 1, 2018, pp. 51–66.
Schnitkey, G., K. Swanson, J. Coppess and S. Armstrong. "Managing the Economics of Planting Cereal Rye as a Cover Crop." farmdoc daily (8):151, Department of Agricultural and Consumer Economics, University of Illinois at Urbana-Champaign, August 14, 2018
Both farms in our study were able to achieve soil temperatures necessary to effectively control soilborne diseases as described by the literature. Both farms were also able to exceed the threshold of anaerobic soil conditions (measured as cumulative Eh millivolt hours) that correlate with the significant reductions in the amount of soilborne fungal pathogen sclerotia in the soil observed in successful ASD treatments. However, despite the apparent success of achieving the metrics understood to suppress soilborne fungal pathogens like Rhizoctonia solani, these effects were not reflected in the disease and yield data. This study shows limited treatment effects and a relatively high cost of implementation compared to untreated control treatments, suggesting that even though the Northeast can attain the conditions associated with “successful” ASD implementation, current market prices and control effects may not be profitable to a commercial grower. Nonetheless, because this was the first instance of implementing ASD in a field setting in Vermont and considering the very limited range of options for soilborne pathogen control on organic farms, ASD may still be worth further investigation. For example, alternative carbon sources, different (higher value) crops, and different cropping systems (greenhouses, hoop houses) may contribute to more effective ASD-mediated disease control and result in a net profit. Until further research is conducted, this study serves as an initial case of implementing ASD in a field setting on organic Vermont vegetable farms for soil pathogen control.
Education & outreach activities and participation summary
Participation summary:
Outreach methods changed due to impacts and restrictions from COVID-19. Outreach occurred digitally in lieu of presenting results at in-person conferences, as originally proposed pre-pandemic. Trial results were distributed in the form of an online anonymous survey. A link to the survey was shared through several influential agricultural organizations in the Northeast, including the University of Vermont Extension newsletter, Deep Root Organic Co-op newsletter, Northeastern Organic Farming Association (NOFA) newsletter, and posted on the Vermont Vegetable and Berry Growers Association (VVGBA) listserv forum. The survey was active for three months in which time 18 responses were collected.
The survey posed questions about whether a farmer was currently struggling to control soilborne pathogens, management practices used (e.g., incorporating compost, growing cover crops, utilizing tarps for weed control/solarization). Due to the high cost of treatment application and poor yield performance observed in the ASD treatments, we assumed most growers would not consider practicing the methods tested and, thus, did not include any query about participants’ “likelihood of adoption”. At the conclusion of the trial, results were shared with the grower collaborators who then participated in an interview inquiring about their perceptions of the drawbacks, benefits, and overall observations from the ASD trial. The survey and grower interviews served as community outreach and education efforts due to the unforeseen challenges caused by the COVID-19 pandemic.
The two farmers who collaborated on this project were interviewed at the end of the trial (summer 2021) for feedback based on their perceptions of the relative successes of the treatments, benefits/challenges, what could be improved, etc. The interviews were analyzed using a similar method as the surveys, in which the responses were compiled, categorized, and sorted.
Learning Outcomes
The anonymous survey showed that the majority of respondents have issues relating to soilborne pathogens (61%). Although this was not feedback specifically pertaining to the pathogen in this study (R. solani), it exemplifies the pressing need to find effective means of control on organic farms in the Northeast. The majority of the survey respondents also report practicing tarping for weed control (55.6%). Interestingly, neither of the collaborating farmers utilized tarping for any reason on their farms prior to this study, but one noted that they have been using the tarp leftover from the trial on their farm for weed control and protective coverings for their compost pile. A majority of survey respondents also reported incorporating compost (55.6%) and incorporating cover crops (72.2%) as a part of their farming practices; both grower collaborators implement these methods well. This input suggests that if an efficacious ASD method using tarping, cover crops, and compost could be ascertained, it could be easily integrated in the farming practices of the Northeastern agricultural community.
Project Outcomes
Based on the survey findings, farmers are unlikely to adopt the methods tested in this study. This experiment may serve as an example for farmers to reference before deciding to implement ASD in a field setting on a commercial vegetable farm in Vermont, therefore potentially saving them time and money due to the lackluster disease control results and expensive costs of implementation.
This study serves as an initial case study of implementing and demonstrate that the ASD method is achievable in a field setting on Vermont vegetable farms. Although the practice was not reflected in the disease and yield data, it is still worth investigating possible use of ASD in Vermont. For example, alternative carbon sources and/or different (higher value) crops may contribute to more effective ASD-mediated disease control and result in a net profit.
Achieving sufficient anaerobic conditions (eliminate oxygen), it was necessary to install drip tape to irrigate under the tarp regularly to keep moisture conditions near saturation. Although farmers often own tarps for weed control, considerable labor is necessary to establish ASD treatments. For example, we discovered an unanticipated problem with cracked corn. Upon removal of the silage tarp at the end of the ASD treatment, we witnessed a plume of Aspergillus spores arising from the soil, a fungus capable of inciting a respiratory ailment called aspergillosis. A subsequent controlled experiment confirmed the fungus was originating from the corn itself which affirms our intent to disband use of cracked corn and revert to our original plan of using a cover crop in 2021.
Until appropriate equipment and carbon source costs can be affordable to the Northeastern community, ASD may be a practice reserved for greenhouse and hoop house settings more for pathogen to control, especially in organic production systems where pathogen control is of critical importance, but few viable options currently exist. Greenhouse conditions are both more consistent than field conditions and are more likely to already have drip irrigation systems installed which, based on our research, proved to be a critical component of successful ASD.