Final report for FNC18-1138
Project Information
How many acres: 280
What crops: Corn and Soybeans
Grazing systems: 24 acres of permanent pasture and concrete feedlot with silo and automatic feeder.
How long farm or ranch has been operating: Approximately 20 years
Crop production in Iowa is dominated by short rotations of GM corn and soybean relying on glyphosate (active ingredient of Roundup). Despite claims that glyphosate rapidly dissipates and becomes immobilized in the environment, recent studies report that over 1000 ppb of residues of glyphosate and its decomposition products, were detected in soils under intensive glyphosate use. Based on reports that glyphosate residues affect soil biology coupled with poor weed management due to glyphosate-resistant weeds, many farmers are considering a change toward non-GM cropping systems. However, little information is available on time required for dissipation of glyphosate/AMPA residues or for soil health buildup to guide farmers in assessing the sustainability of management during transition from GM to non-GM crops. We will monitor glyphosate soil residues and soil health indicators during transition to establish baselines for improvement of soil using ecologically sound soil and crop management, which will decrease contamination of surface water with glyphosate in runoff and improve soil biology. Transition may improve profitability as premiums are paid for non-GM grains. Decreasing or eliminating use of glyphosate promotes socially responsible benefits by minimizing exposure of the rural community to drifts of glyphosate spray and providing healthy ecosystems through improved soil health.
- Set up a glyphosate/AMPA monitoring protocol for residues in soils using an intensive soil sampling approach at sites prone to glyphosate drift or runoff and at sites not exposed to glyphosate.
- Concurrently monitor soil health by measuring selected indicators in crop management transition fields without glyphosate. Crop performance (tissue nutrient contents, N response and leaf chlorophyll, and grain yield) will also be measured and related to soil health status.
- Develop a guide for farmers to track soil health improvement after transition from GM cropping.
Research
Progress Report: “Mitigation of Potential Adverse Effects of Transgenic Crop Production for Long-Term Improvement of Soil Health” February 2019
Introduction
The results and preliminary information provided in this report originate from sampling sites on the Osweiler Farm, Delaware County, IA. As a reference the farm map and boundaries are provided in Figure 1. The results as presented are oriented toward sample sites on the farm, which are designated in Figure 1 to accommodate quick reference to details given in the text. NOTE: The current version of this report was updated October 6, 2020 with results from adjusted glyphosate analytical data that was corrected after the analytical laboratory detected errors in the protocol used in analyses some time after the final report was submitted. The adjusted results are reflected in updated text and Table 1 and Figure 5 in the Glyphosate Analysis and Soil Glucosidase sections of the Results and Discussion.
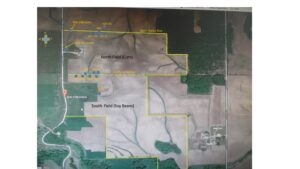
Figure 1. Michael Osweiler Farm, 2336 270thAve, Earlville, Iowa 52041 (Delaware County). Soil sampling sites are shown as blue circles. Note north transect sample sites (3W, 2W, 1W,1E) and south transect sites (1SW, 2SW, 3SW, 4SW); NFR, north fence row; SFR, south fence row; Glyphosate check (NRU), continuous corn with Roundup located in north adjacent field; north field non-glyphosate check at southern edge (1CW, 2CW, 3CW, 4CE). NOTE: North field planted to corn in 2018 and soybean in 2019; South field planted in soybean in 2018 and corn in 2019. Grass waterway on western edge of North field was sampled at inlet at fence row with adjacent field (2GN) and at outlet to the southeast near road ditch (2G); sample collected at outlet of South field near bottom of a NE-SE slope (3G) - this is not developed as a grass waterway and is sparsely covered with weed escapes.
Objective 1a: Monitor glyphosate/AMPA residues in soils.
Glyphosate analyses: Soil samples collected in July 2018 and 2019 along transects established in non-GM corn and soybean fields, adjacent fence rows and one GM corn field with glyphosate (NRU), and grass waterways were analyzed for glyphosate and amino-phosphonic acid (AMPA) residues. The soils were extracted and analyzed for glyphosate and AMPA residues at HRI Labs in Fairfield, IA using liquid chromatography mass spectrometer instrumentation.
Soil analyses:
Soil test properties were determined for each sample site along the transects plus for samples from fence rows, adjacent GM corn field and waterways. Measurements included pH, soil C (soil organic matter), total N, macronutrients and micronutrients. The soil health indicator, active carbon, was analyzed using the permanganate oxidizable C (POXC) assay, an estimate of labile soil C.
Objective 2a: Monitor crop performance (tissue nutrient contents, N response and leaf chlorophyll, and grain yield).
Plant tissue analyses:
Nutrient analyses were performed on leaves collected during active growth of corn (ear leaf at early tassel or VT) and soybean (newly expanded trifoliate leaves at full bloom or R2) after drying and grinding to a fine powder for chemical extraction and analyses on an ICP.
Leaf chlorophyll analyses:
A SPAD chlorophyll meter that detects differences in far red wavelengths related to leaf chlorophyll concentration was used to non-destructively measure the chlorophyll content of leaves of corn and soybean in July 2018 and 2019 at same growth stage as those collected for nutrient analysis. Measurements are expressed in dimensionless “SPAD units” ranging from 0 to 80. SPAD units are highly correlated with chlorophyll content per unit leaf area and also indicate leaf N status, leaf senescence, overall plant health, and responses to environmental stresses. Our intent was to detect effects of soil residual glyphosate and possibly glyphosate drift on leaf chlorophyll content. Ten readings were taken for each entry and the average values are presented in Figures 3 and 4 and summarized over years in Table 5.
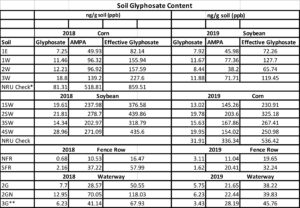
Glyphosate analyses. Soil samples collected in July 2018 along transects established in non-GM corn and soybean fields, adjacent fence rows and one GM corn field (with glyphosate applications), and waterways were analyzed for glyphosate and amino-phosphonic acid (AMPA) residues. The effective glyphosate (glyphosate x 1.5 + AMPA; HRI Labs) contents were highest for soils cropped to soybean; glyphosate content also varied among samples within crop fields in 2018 ranging from 82 to 227 ng/g soil (ppb) for corn and 318 to 440 ng/g for soybean (Figure 2). Glyphosate was last applied to these fields in 2015 thus showing long-term persistence (3 years+) and a highly variable distribution within small sub-areas of the fields. Some reports suggest that GM-corn may accumulate glyphosate in roots and release into soil, however this does not explain the higher glyphosate content detected in soybean soils as the previous GM corn was planted in 2015. More background investigation will be required to understand the variable glyphosate accumulation noted for corn and soybean.
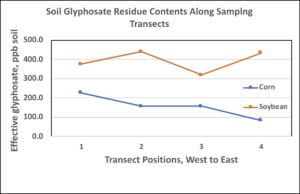
Figure 2. Effective glyphosate residues detected in soils collected along two 400-ft transects established in 2018 corn and soybean fields. Samples collected at 100-ft intervals along each transect and position locations are numbered beginning with western (1) through eastern points (4). The corn transect was 50 ft from an adjacent GM corn field that received Roundup applications in 2018; the soybean transect was adjacent to the non-GM corn field.
Results summarized over the two-year project shows that soil glyphosate residues decreased somewhat in 2019 compared with 2018 (Table 1). Glyphosate continued to be variable across the landscape within short distances with contents reduced from 12 to 58% in soils from transect sites in both fields and by 38% in the glyphosate check (NRU). The NRU site, within a field under continuous GM corn and receiving multiple Roundup applications, showed the highest effective glyphosate contents in soil each year. Decreased glyphosate contents in the grass waterway soils ranged from a 24% decrease for the outlet site to 66% at the inlet (see Figure 7, Photographs page) where we suspect glyphosate entered from the adjacent field in runoff and sediment. We suggest that the overall decreases within four years after last Roundup application to the crop fields were due to changes in management during transition away from transgenic crops and termination of any glyphosate applications in the future. Based on the high dissipation in the waterway, establishment of perennial vegetation or rotation to include cover crops during the off season likely supports a diverse and highly functional microbiome capable of relatively fast glyphosate degradation. The variable decreases in residual glyphosate content noted within the fields and across the corn-soybean rotation suggests that the extent of biodegradation is also variable likely due to a variable distribution of functional microbial communities across the field landscape, which may be a result of different soil properties and microclimatic factors. For example, site 2GN (grass waterway inlet at north field fence row) had highest values for soil organic matter (8.9%, Table 2), soil active carbon (890 mg/kg, Table 5), and total microbial biomass (186,890 pg/kg, Table 6), which contribute to high functional microbial diversity, including herbicide biodegradation potential, that likely contributed to the 70% decrease in effective glyphosate content in 2019.
Table 1. Glyphosate and AMPA contents detected in soils during 2018 -2019. *Site NRU from field adjacent to North field that is in continuous GM corn receiving recommended applications and timings for Roundup.**Site 3G is situated near bottom of NE-SE slope in south field with drainage outlet into adjacent pasture. This site is not developed as a grass waterway and is sparsely covered with scattered weed escapes.
Soil analyses: Soil test properties were determined for each sample site along the transects as well as for samples from fence rows, adjacent GM corn field and waterways (Table 2). Most nutrient levels were within sufficient ranges for both corn and soybean crop performance standards except for insufficient levels for boron, copper, and molybdenum in nearly all samples. Potassium was rated insufficient for soybean in two of four samples. Evidence for persistence of soil glyphosate contents based on relationships with soil contents of specific nutrients was not consistent. The lower phosphorus (P) contents at soybean sites (highlighted data in Table 1) suggest that glyphosate may be concentrated since it shares same soil adsorption sites as P. Also, lower soil pH in some samples (all soybean and some corn) may contribute less reactive glyphosate as the compound carries less charge under these conditions, may be less adsorbed, occur in soil water, and more available for plant uptake. This may be the case for the corn field receiving 2018 glyphosate applications (‘With RU’) on soil with a pH of 5.06, which likely led to the highest soil glyphosate content, and likely contributed to high glyphosate content at the entrance to the grass waterway (NW-2GN) in the north field via erosion and/or runoff. Higher soil organic carbon (SOC) at some sites may hold (adsorb) glyphosate, accounting for higher glyphosate content relative to samples with lower SOC. Overall, most of the nutrient content in soils were similar suggesting adsorption of glyphosate by cation nutrients as a means of glyphosate retention in soil was likely similar in both fields.
Table 2. Soil analyses for 2018 production fields at Osweiler Farm for SARE Project.
**SOC = Soil organic carbon expressed as %; SOC X 1.75 will approximate % Soil Organic Matter content; Waterways: NW-2GN, grass waterway in northwest corner of north field near county road, sample position at fence line with neighbor’s corn field on the north; NW-2G, grass waterway in northwest corner of north field near county road, sample position near “outlet” at southern end near drainage into road ditch; S-3G, drainage from southwest slope in south field near gate into adjacent pasture, sample position near outlet at edge of field.
Plant tissue analyses: Nutrient analyses of leaves during active growth of corn and soybean revealed some insufficient nutrient levels that may have been affected by glyphosate soil residues (Table 3). However, the effect on nutrient level in corn was not consistent across samples likely due to variability in nutrient distribution across the field, even in the localized area of the transect. Nevertheless, low levels of potassium (K), zinc (Zn), and boron (B) might be affected by immobilization in soil by glyphosate thereby making these nutrients less available for uptake through corn roots. Molybdenum (Mo) was insufficient or deficient, however, based on past nutrient analyses records, these soils appear to be deficient in this micronutrient regardless of glyphosate application. The less than sufficient level of nitrogen (N) in corn is likely a consequence of immobilization of micronutrients (and insufficient Mo) required for N metabolism in corn thereby affecting accumulation within the plant. Interestingly, the corn sampled at the site most distant (‘No exposure’ or ‘Check’) from glyphosate-treated GM-corn showed sufficiency levels for all nutrients tested. Although soil glyphosate was not measured, results suggest that glyphosate drift from adjacent fields (as is the case for the other corn samples along the north transect) might intensify effects on nutrient uptake and metabolism in corn. Soybean was similarly affected by apparent nutrient immobilization by soil residual glyphosate, with more consistent affects across all samples for magnesium (Mg) and Cu as well as two of four samples for K.
Table 3. Plant Tissue (Leaf) Analysis - 2018 Osweiler Farm - SARE Project

For 2019, summarization of both soil and plant tissue analyses (Table 4) reveals similar trends as discussed above for 2018 results. In contrast to soils from North and South fields in transition to non-transgenic crops, the glyphosate control (NRU) shows a lower soil pH and reduced contents of Ca and Mg, possibly a consequence of repeated use of Roundup allowing glyphosate to interfere with the cation nutrients as well lower pH. This is also reflected in reduced tissue content of Ca and Mg. The insufficient levels of Boron (B) and Molybdenum (Mo) at nearly all sites is of concern as these are critical micronutrients required for a number of important metabolic functions in both plants and microorganisms. It appears both corn and soybean are able to extract sufficient B, however, most plant tissue was deficient in Mo. These results confirm the need to monitor soil micronutrients and make appropriate amendment either to soils or as foliar applications when necessary. The impact of perennial vegetation on soil fertility and health is shown in samples from waterways and fence rows, especially in the considerably higher levels of soil organic matter (SOM) and total N (Table 4).
Table 4. Soil and plant tissue analyses summary for 2019.
Leaf chlorophyll analyses:
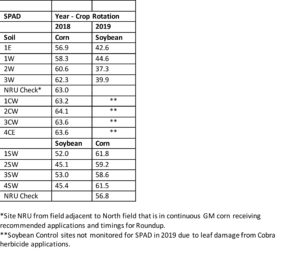
Field measurements taken on corn ear leaves (stage VT) showed an indirect relationship of residual glyphosate content on SPAD measurement in non-GM corn sampled along the transect (Figure 3). Interestingly, GM corn sprayed with glyphosate revealed higher SPAD, however, this may reflect selection for photosynthetic ability of a transgenic hybrid in presence of glyphosate. The non-GM corn showing highest SPAD readings under presumably lowest glyphosate exposure suggests that glyphosate detrimentally affects photosynthesis in this hybrid.
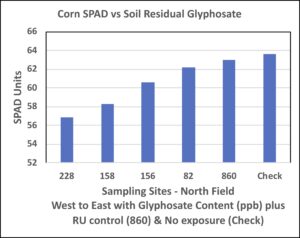
Figure 3. SPAD units of leaf area of the earleaf of corn measured on plants along the sampling transect in the north field and those in GM corn receiving glyphosate and in non-GM corn not exposed to glyphosate. Leaves were monitored at VT corn growth developmental stage (tasseling) during the 2018 growing season. Samples collected at 100-ft intervals along each transect, and position locations are numbered beginning with western (1 or 228 ppb) through eastern points (4 or 82 ppb).
Field measurements taken on trifoliate leaves at bloom stage (R2) similarly showed an indirect relationship of residual glyphosate content on SPAD measurement in soybean sampled along the transect (Figure 4). As observed for corn, soybean showing highest SPAD readings under presumably lowest glyphosate exposure in soil suggests that glyphosate detrimentally affected photosynthesis in this particular variety.
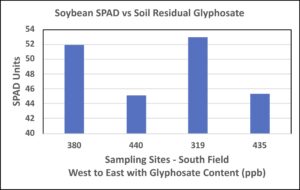
Figure 4. SPAD units of leaf area of the newest expanded trifoliate leaves of soybean measured on plants along the sampling transect in the south field. Leaves were monitored at R2 soybean growth developmental stage (flowering) during the 2018 growing season. Samples collected at 100-ft intervals along each transect, and position locations are numbered beginning with western (1 or 380 ppb) through eastern points (4 or 435 ppb) (See Figure 1 for the south field transect sample locations).
A summary of SPAD readings over the two-year period show fairly consistent values for corn and soybean over the rotation phase (Table 5). A decrease for the glyphosate check (NRU) may indicate cumulative effects of glyphosate residues in soil, however more replicate samples are needed to make a firm conclusion. Soybean following corn in the North field in 2019 show an overall reduced SPAD relative to soybean in South field in 2018, suggesting reduced activity to possible glyphosate drift from the field adjacent to the North field, as shown for corn in 2018.
Table 5. SPAD results summarized for corn and soybean during 2018-2019.
Objective 2b. Concurrently monitor soil health in crop management transition without glyphosate.
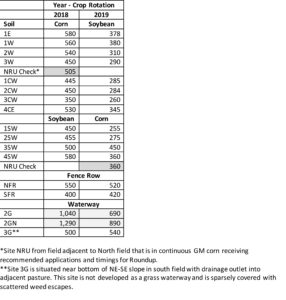
Active Carbon: Active C was analyzed using the weak extractant POX as it represents the most sensitive soil organic C fraction to management changes and is therefore a sensitive biological indicator of soil health. Active C also indicates the level of microbiological activity present in soil. Active C contents were higher for all soils in 2018 relative to 2019. Values were similar across all cropped soils (including NRU), fencerow, and the non-grass drainage sites (Table 6). This is likely related to the lower SOM values for all sites (see Tables 2 and 4), suggesting overall lower microbial activity. Highest active C in both years was associated with the grass waterway sites, related to vegetation and higher SOM, and suggesting that management incorporating either perennial vegetation or consistent presence of living plants throughout the year will improve biological soil health.
Table 6. Active carbon contents in soils at selected sites during 2018 and 2019.
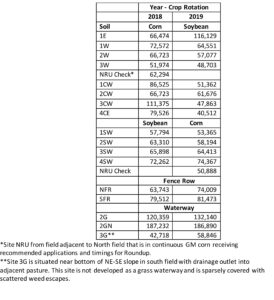
Soil Microbiome: The soil microbiome ("microbial community") was assessed using the PLFA biochemical marker to distinguish broad groups including mycorrhizae, saprophytic fungi, Gram-negative and Gram-positive bacteria, actinobacteria, and protists. We found very little differences among all soils in the abundance of microorganisms in each of these groups (data not reported). It is not known whether the somewhat homogenous distribution of similar abundance is related to effects of management such that the microbiome was resistant to changes and remained similar to non-managed fencerow and waterway sites or if abiotic climatic factors had an impact. It should be noted that 2018 and 2019 experienced above normal precipitation during the growing seasons (27 and 30 inches during April - September, respectively), which may have reduced impacts of management. Interestingly, total PLFA content, an indicator of total soil microbial biomass, varied among the sites, with the glyphosate check (NRU) among the lowest suggesting an effect of cumulative soil glyphosate residue (Table 7). With two exceptions, highest total PLFA were associated with fencerow and waterway soils. Residual glyphosate may affect microbial abundance and biomass in the cultivated soils. More importantly, the type of soil ecosystem provided by undisturbed, perennial vegetation enhanced microbial abundance, which is important to soil health for providing diverse biological function (nutrient cycling, plant growth regulators, pathogen suppression, etc.) and a source of nutrients in biomass are recycled for plant availability.
Table 7. Soil microbiome analyzed as phospholipid fatty acid content (pmoles/g dry soil) at selected sites within two fields at Osweiler Farm under corn-soybean rotation, 2018-2019.
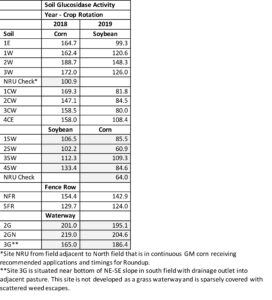
Soil Glucosidase Activity: Soil glucosidase is a microbial enzyme that represents C cycling ability by the microbiome and therefore is an indicator of soil microbial activity and considered a biological soil health indicator. Soil glucosidase appeared to be more sensitive to soil glyphosate residues than other biological indicators in our study (Table 8). Although activity values were lower in 2019 likely due to dry soil conditions at sampling, the trend relative to glyphosate content was consistent across years. The lowest activity associated with the highest soil glyphosate content (treatment NRU) illustrates this trend.
Table 8. Soil glucosidase activity, as amount of product produced per g of soil (ug PNP/g dry soil), at selected sites within two fields under corn- soybean rotation, 2018-2019.
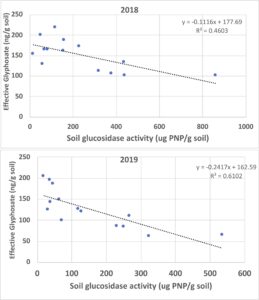
Relationship of Glyphosate Soil Residues with Soil Microbial Activity: Several plant and microbiological parameters were analyzed for any correlation with soil glyphosate contents to detect potential impacts of continuous use of glyphosate in transgenic crop production on soil health. The most robust correlations found were the indirect relationship for glyphosate and glucosidase activity in both years, showing decreasing activity with increasing glyphosate content (Figure 5). A moderate correlation (correlation coefficient, R = 0.46) of decreasing soil glucosidase activity with increasing effective glyphosate content in the soil surface was found in 2018; this correlation was even stronger (R= 0.61) in 2019, suggesting that persistent glyphosate residues in soils are biologically available and interfere with microbial activity. The high content of soil residual glyphosate in the control soil (NRU) likely contributed to lowest glucosidase activity detected in those soil samples, which was consistent for both years. It is also possible that additional herbicide treatment to control glyphosate-resistant weeds or different weather patterns in 2019 led to values for fencerow and waterway soils to remain stable resulting in a consistency at about the same levels for both years. This suggests that favorable conditions in these sites not only provide for higher microbial activity but maintains the same level of activity across seasons. Subsequent evaluation of glucosidase activity in other soils containing glyphosate residues may reveal similar indirect correlations, suggesting that soil glucosidase may be a sensitive indicator for detecting potential effects of glyphosate that would be useful to other farmers considering management changes for transitioning away from transgenic crop production.
Figure 5. Relationship between soil effective glyphosate content with soil biological activity measured as soil glucosidase enzyme activity for both years of project.
Objective 3. Develop a guide for farmers to track soil health improvement after transition from GM cropping. We are in the process of assembling this guide now that the results have been summarized. Our plan is to publish a simple brochure to guide users to a website that will be accessible to provide all results collected for this project, include management tips learned from the project, including photographs; and information on soils and soil health.
The general outline for the brochure (2-sided color printed on card stock):
Side 1: Title - "Soil Health Restoration After Transgenic Crop Production" - include a few selected photos
Online resources: Experimental data; Photos; Soil health methods
Short statement: "for transitioning to a sustainable cropping system, healthy soils, improve environmental quality, profitable farming
Side 2: Cropping transition outreach online
- for farmers, educators, agricultural professionals, youth
- how to improve: soil health and productivity; protect environment (effective waterway design); cropping efficiency (non-GM varieties, etc.)
- Resources available (provide list)
Questions/suggestions: Contact person information; website address (currently under development)
Major Findings to Date:
- Soil glyphosate residues are variably distributed across a given field.
- Glyphosate residues persist in soil more than four years after last application.
- Glyphosate seems to be concentrated in soils under soybean production.
- Glyphosate movement offsite from adjacent GM-corn field occurred based on detection in waterway in north field.
- Glyphosate appears to be dissipated (degraded) quickly in grass waterway due to perennial vegetation supporting diverse and abundant soil microbiome and high microbial activity.
- Glyphosate persistence in soil is complex involving the soil properties SOC, pH, and several cation nutrients; however, the properties vary among soil samples making interpretation difficult.
- Glyphosate appears to affect nutrient content and sufficiency in both corn and soybean likely due to effects of soil residues on nutrient immobilization and decreased plant uptake.
- Glyphosate residues appear to detrimentally affect photosynthesis in corn and soybean based on SPAD measurements.
- Glyphosate drift may be an additive factor in reducing nutrient metabolism and photosynthesis in corn.
- Maintaining sufficient levels of soil micronutrients is critical for both plant nutrition and soil microbial activity during transition period.
- Soil glucosidase activity seems to negatively correlate moderately well with soil glyphosate content, yielding a potential assay for residual glyphosate impacts on soil health and crop productivity in the transition period
- The restoration of microbial activity and abundance in soils may require several years and/or incorporation of perennial crops or cover crops during transition from transgenic to non-transgenic cropping systems
Educational & Outreach Activities
1. Informal discussions with visitors to farm involving description of the project and the potential for extension of the measured parameters to include pollinator refuge as protection against possible damage from surrounding fields of transgenic crops receiving glyphosate herbicide applications.
2. Visited with NRCS personnel to keep the agency informed on soil health assessment being conducted on the farm relative to other farms in Delaware County.
Participation summary:
We communicated with visitors and NRCS personnel in follow-up email messages regarding soil health assessments and preliminary results of the project. We also discussed the possibility of setting up pollinator surveys on the farm either during the project or as a follow-up after project completion. A brochure is under development highlighting the main findings and guiding users to a website, as described for Objective 3. Dr. Kremer has used some of the glyphosate residue results for illustrating carryover in soils as part of some presentations to farmer groups with full acknowledgment to SARE. We plan to further communicate the results in a website devoted to the project to be set up by April 15, 2020; as part of a presentation on soil glyphosate residue distribution and impacts on soil health planned for the Soil Science Society of America meeting in November 2020; and as part of a scientific article to be prepared for an appropriate journal such as Agrosystems, Geosciences, and Environment .
Learning Outcomes
Advantages included conducting a project that was probably one of a kind and provided novel and useful information to farmers concerned about the effects of continuous use of Roundup on crop productivity and on soil health. At project termination we collected a sizable amount of data that provides at least a preliminary information basis to use in making recommendations for transitioning from transgenic cropping systems regarding potential glyphosate carryover effects on crop performance and soil health, especially soil biology.
However, we recognize this was a limited study due to the funding level and the amount of time and labor required to conduct a replicated study that would provide reproducible and confident results. The cost of glyphosate analyses severely limited the number of soil samples that were submitted for analyses. In the future, more intensive assessments with replicate samples at various sites within fields under study will be necessary to validate the information basis.
When conducting a farm-scale study such as in this project, more attention to activities and management systems on adjacent farms is required. We found that glyphosate can be readily dissipated onto our research sites through spray drift and in runoff and on sediment suspended in runoff. This provided an opportunity to document this dissipation onto the crops in our study and to demonstrate the efficiency of the constructed grass waterway that collected runoff from the adjacent field. However, it is critical to include samples from the adjacent field to validate that herbicide was accumulating in soils and provide more robust data than relying on the one sample we collected for the current project. The lack of additional funding to analyze these soils for glyphosate was a major limiting factor in pursuing this approach.
It is critical to have a consistent management plan in place when undertaking a transition project such as this. Farmers and landowners need to be aware that herbicide-resistant weeds likely developed after years of continuous Roundup use. As occurred on this farm, the crop managers (tenants) did not take prompt and effective action to implement management to control such weeds within the proper timeframe (i.e., early season control of marestail and control of waterhemp species before four-inch height) resulting in numerous post-emergence applications in attempt to manage the weeds during the mid- to late crop growing season. The additional herbicide applications and trips across the field confounded results collected especially in the second year (2019). The soil degradation and crop damage along with input of herbicides into the soil may have affected soil biology as well as crop productivity. These are factors that farmers attempting to change management on their fields must be aware of.
Project Outcomes
Interest about effects of glyphosate on pollinators has continued through informal discussion and email messages, which may lead to another project on the same sites to evaluate beneficial arthropods exposed to glyphosate.
NRCS personnel continue to express interest in the general soil health assessment activity that may be applicable for other farms or areas within their region.
The co-director of HRI Labs in Fairfield, IA has expressed interest in our results as they are one of few laboratories that offer this type of analytical service in the nation. Continued discussions may lead to collaborative projects in which HRI would provide the analytical service in exchange for using results as part of a database or as collaborative authors on seminal papers addressing the topic of glyphosate persistence in the environment.