Progress report for FNC25-1453
Project Information
Much of my life has been devoted to deepening my relationship with the natural world. From an early age, I understood our purpose as stewards of the land: to leave it better than we found it. This guiding principle led me to the fascinating world of fungi, the great decomposers and humus heroes of our ecosystem. My earliest memory of this relationship was playing puffball soccer as a child, dispersing spores with each kick was the goal. What began as a game grew into a lifelong fascination with fungi.
In 2015, my wife brought home a white button mushroom kit from River Valley Mushrooms, sparking my formal exploration into mushroom cultivation. By 2016, I had built a 64-square-foot cultivation space in our basement to experiment with substrates and varieties of gourmet and medicinal mushrooms. These early efforts laid the foundation for my expertise in cultivation.
In 2020, we purchased a 4-acre farm in Pecatonica, Illinois. Over the years, we have raised chickens, rabbits, pigs, sheep, and turkeys alongside annual and perrenial crops, but mushroom production has remained our focus. I have cultivated nearly 20 mushroom varieties, mastering both indoor and outdoor methods, sterile lab work, and community education. Since 2021, I have shared this knowledge through training sessions, original guides, and consultations. My wife, Apolonia Ramon, has also been an essential part of our operations, assisting with mushroom harvesting, packaging, deliveries, marketing, product development, web design, and livestock care.
Our farm operation integrates direct-to-consumer sales, wholesale accounts, and farmers’ markets. We’ve also refined our animal production, focusing on broiler chickens, eggs, and Icelandic sheep for wool and meat. These efforts align with our commitment to sustainability and regenerative practices.
My formal education includes a bachelor’s degree in English - Library Media and an MBA in finance, which provide strong organizational, research, and business management skills. For this project, I will oversee all aspects, including substrate preparation, mushroom cultivation, data collection, and educational outreach.
This project reflects our farm’s sustainable approach, integrating fungi-driven soil regeneration with diversified operations. It serves as a replicable model for improving agricultural outcomes while fostering a deeper connection to the natural world.
Degraded, rocky soils pose significant challenges for sustainable agricultural production, often limiting options for soil restoration and crop viability. Conventional methods rely heavily on tillage and synthetic inputs, which can exacerbate erosion and nutrient loss over time. Small-scale farmers need low-cost, accessible solutions to regenerate soil health while diversifying farm income.
The mushroom cultivation industry, while economically promising, relies predominantly on resource-intensive indoor systems and single-use polypropylene plastics, contributing to significant waste and environmental impact. Outdoor cultivation offers a sustainable alternative but lacks widely adopted, scalable models that integrate fungi into regenerative farming practices.
Additionally, rural communities often lack access to medicinal mushrooms, despite their well-documented health benefits and growing demand. As a result, farmers and ranchers in these communities miss opportunities to incorporate high-value, health-promoting crops into their operations while improving degraded land.
This project addresses these challenges by integrating fungi as decomposers and soil builders into a low-tech, outdoor system. By leveraging waste streams, such as wood pulp and discarded nursery pots, the project minimizes ecological impact while producing a marketable farm product. This solution supports sustainable agriculture by addressing soil degradation, reducing waste, and educating farmers on waste-reducing, no-till mushroom cultivation techniques.
Solution
This project establishes a low-tech, sustainable outdoor system for cultivating Ganoderma lucidum (Reishi) mushrooms while improving soil health on degraded, rocky land in USDA Zone 5b. By utilizing fungi as decomposers and soil builders, the project demonstrates how small-scale farmers can address soil degradation, reduce waste, and diversify income through sustainable agricultural practices.
Reishi mushrooms will be cultivated in nursery pots, sourced from waste streams created by commercial nurseries, and filled with wood pulp substrate derived from tree service byproducts. These pots provide a controlled environment for the substrate while reducing reliance on single-use polypropylene plastics, a common industry practice. Nursery pots are arranged under a high tunnel with shade cloth to regulate light, temperature, and humidity for optimal mushroom growth.
At the end of the growing season, spent mushroom substrate will be combined with other organic waste materials, such as straw, manure, and wood chips, to create nutrient-rich compost. This compost will be applied as a top-dressing on the grow site, building organic matter, enhancing microbial activity, and improving soil fertility without tillage. By using fungi-driven decomposition, the project promotes a regenerative cycle of nutrient replenishment that restores degraded soils over time.
Following the first harvest, chickens will be introduced to one side of the high tunnel. They will graze and naturally fertilize the soil with nitrogen-rich manure, while their scratching behavior will aid in substrate breakdown and aeration. The other side of the high tunnel will serve as a control site to assess the chickens’ impact on soil health.
The project also minimizes ecological impact by repurposing agricultural waste streams, such as discarded nursery pots and wood pulp, to reduce landfill contributions and reliance on non-renewable inputs. This approach aligns with regenerative agriculture principles, addressing soil degradation while promoting sustainability and economic viability for small-scale farmers.
Education and outreach are integral to the project. Workshops and field days will be held to demonstrate every stage of the process, from substrate preparation and mushroom cultivation to composting and poultry integration. Instructional guides, videos, and detailed data summaries on yields, soil health, and economic performance will be shared online and at community events to maximize the project’s reach and replicability.
This system exemplifies how fungi can be harnessed to regenerate soil, create value from waste, and provide access to high-value medicinal mushrooms for rural communities. By integrating no-till composting, poultry systems, and waste stream utilization, the project offers a replicable, low-cost model for improving soil health and sustainability while promoting income diversification and health benefits.
Objectives
- Develop a sustainable outdoor Ganoderma lucidum cultivation system using nursery pots and wood pulp substrate.
- Build soil health and organic matter on degraded land through fungi-driven composting and no-till practices.
- Utilize waste streams, such as nursery pots and wood pulp, to minimize waste and reduce inputs.
- Integrate chickens to enhance nutrient cycling and decomposition.
- Produce a marketable medicinal mushroom to support farm income.
- Educate 100 farmers and community members through workshops, field days, and online materials.
- Promote awareness and access to medicinal mushrooms, supporting health and wellness in rural communities.
Research
Materials and Methods (Process + Logic Behind Decisions)
This project tested a low-tech, outdoor method for cultivating Ganoderma lucidum (Reishi) on a small farm in USDA Zone 5b while improving degraded, rocky soils through no-till organic matter building. The system was designed to be replicable for other farmers by prioritizing low-cost infrastructure, locally available waste streams, and simple management practices.
1) Site Preparation and Growing Environment
We selected a caterpillar tunnel as the primary growing structure because it is more affordable than a conventional high tunnel and provides a semi-controlled environment for mushroom production. Because the site had little to no natural tree cover, we planned to regulate light and heat using 70% shade cloth.
To suppress weeds and avoid tillage, we laid down donated cardboard across the growing space and top-dressed it with approximately 4” of wood chips sourced from a local waste stream. This created a mulch layer that helped retain moisture, reduced weed pressure, and provided a biologically active surface where spent substrate could later be broken down and incorporated naturally over time.
2) Substrate Sourcing and Preparation
Our cultivation method relied on woody substrate materials that could be sourced locally as waste. We initially attempted to source hardwood sawdust from woodworking shops and sawmills, but supply constraints required us to use hardwood fuel pellets for roughly half of the season’s substrate production. Later, we secured a delivery of oak sawdust from a contractor and tested it using the same formulation.
To support stronger yields and healthier mycelium, we supplemented woody substrate with bran (as a nitrogen source) and gypsum (as a calcium and sulfur source). Substrate was mixed in a cement mixer to reduce labor and improve consistency. We found best results when mixing dry ingredients first, then slowly hydrating to the desired moisture level. Hydration was verified using a moisture meter, typically reading 35–40%.
Substrate was loaded into compostable mushroom grow bags at approximately 5 lb per bag. Bags were folded and prepared for pasteurization.
3) Pasteurization Method (Low-Tech Atmospheric Steam)
To avoid the high cost and complexity of sterilization systems, we used atmospheric steam pasteurization. Our setup consisted of a 170-gallon stock tank and a 55-gallon boiler fashioned from a drum. Substrates were steamed until the internal temperature reached 200°F, then held at that temperature for at least four hours. A digital probe was used to verify internal temperatures during each run.
Although the system was originally expected to process more than 150 bags per cycle, we found that practical capacity was closer to 80 bags per run to ensure adequate steam penetration. The full cycle required significant time to heat the mass of substrate and multiple days for cooling before inoculation, which ultimately influenced production timing.
This pasteurization method proved effective: after processing 436 substrate bags, contamination remained low and manageable for an outdoor system.
4) Spawn Expansion and Inoculation Strategy
We planned the grow site for a capacity of 400 substrate bags and selected a 10% inoculation rate to support strong colonization and reduce contamination risk. Bags were inoculated in front of a laminar flow hood once cooled sufficiently, because spawn mycelium is heat sensitive and can be damaged by warm substrate.
One challenge was the cooling period after pasteurization—blocks often required four days to cool enough for inoculation. This slowed production and spread inoculation across multiple batches, which later contributed to staggered fruiting and difficulty aligning ideal weather conditions with the fruiting window.
5) Fruiting Induction and Bag Management Trials
Once colonized, most bags began forming primordia inside the sealed bag. We intentionally allowed primordia to initiate before opening bags because Ganoderma mycelium tends to climb toward the filter patch seeking fresh air, which can increase contamination risk and interfere with gas exchange.
We tested four fruiting induction techniques to determine what was most suitable outdoors:
-
small corner cut for gas exchange
-
horizontal incision below the seal
-
removing the bag above the substrate surface (common indoor method)
-
trimming excess structures and wrapping the bag around the strongest fruiting body, securing it with a rubber band
The most effective technique was the wrapped-bag method, because it minimized exposed substrate surface area while supporting healthy conk development. The least effective technique was removing the top of the bag, which exposed too much substrate to drying and contamination risk in outdoor conditions.
6) Humidity, Irrigation, CO₂, and Temperature Management
Humidity and temperature were monitored using sensors, and multiple irrigation strategies were tested:
-
Overhead misting increased bacterial risk on fruiting bodies and was discontinued.
-
Soaker hoses did not meaningfully raise relative humidity.
-
A hose-and-sprayer approach that thoroughly wetted the mulch surface raised RH effectively while avoiding direct soaking of fruit bodies, and became the preferred method.
CO₂ levels were generally within acceptable ranges for fruiting (<2,000 ppm). However, temperature management became the most limiting factor. During summer heat, the tunnel environment ran significantly hotter than ambient temperatures, and substrate surface temperatures exceeded ideal growth ranges. When this occurred, we moved blocks into the basement of an old dairy barn for recovery and continued incubation.
These observations led to a key conclusion for other farmers: outdoor Ganoderma production is highly sensitive to timing, and success depends on aligning colonization and early fruiting with the narrow seasonal window when temperatures are within the preferred ranges.
7) Chicken Integration (Nutrient Cycling Trial)
After the first harvest period, chickens were introduced to one side of the tunnel to support nutrient cycling and substrate breakdown, while the other side served as a control area. Chickens foraged actively in the mulch and manured heavily, improving fertility inputs. CO₂ levels increased while chickens were present, but ventilation through rolling up tunnel sides kept levels manageable.
However, chicken presence also increased fly activity and pest pressure, especially around freshly cut mushroom tissue. Based on this, future cycles will avoid overlapping chickens and active mushroom fruiting whenever possible by starting the mushroom cycle earlier and introducing poultry only after harvest and substrate dispersal.
8) Harvest, Post-Harvest Handling, and Soil Building
Reishi was harvested using pruning shears and processed immediately while fresh to avoid excessive woody hardness. Fruits were chopped and dehydrated. Spent substrate was removed from bags, broken apart with a shovel, and dispersed across the wood chip mulch surface as a no-till top dressing. Straw was applied over the broken substrate to support decomposition and increase organic matter.
This step completed the regenerative loop of the project: mushrooms produced a marketable crop while the spent substrate became a soil-building amendment for degraded ground.
9) Culture Selection and Adaptation Planning
We sourced Ganoderma lucidum culture from Terrestrial Fungi due to their strong reputation and genetics quality. Thank you for all of your hard work and dedication, Ryan! We collected spores from this season’s fruits with the intention of selecting or breeding strains better adapted to outdoor conditions in Zone 5b. For the next season, we also plan to trial Ganoderma sessile (a vigorous Midwest strain) alongside G. lucidum to compare performance under the same outdoor system.
In 2025, we successfully developed and tested a low-tech, outdoor Reishi cultivation system using pasteurized woody substrate blocks in compostable grow bags, managed within a caterpillar tunnel and later in a cooler barn space due to heat stress. The season produced measurable outcomes in four major categories: (1) substrate throughput and contamination rates, (2) mushroom fruiting success and yield, (3) environmental management performance, and (4) soil-building and nutrient cycling outcomes through substrate dispersal and poultry integration.
1) Production Results: Substrate Processing, Fruiting Success, and Contamination
Substrates produced and evaluated
We processed a total of 436 substrate blocks during the 2025 season. Each block was prepared as a ~5 lb hydrated substrate bag, pasteurized using atmospheric steam, and inoculated with sawdust spawn.
We tracked block performance through three measurable outcomes:
-
whether the block reached at least primordia formation
-
whether it reached mature conk development
-
whether it showed visible contamination
Fruiting performance
Out of the 436 total blocks:
-
232 blocks entered production and developed primordia at minimum (many reached mature conks)
-
178 blocks did not have adequate time to reach primordia and enter production before conditions became unfavorable
-
26 blocks were contaminated in vitro (in-bag)
This indicates that 53% (232/436) of the blocks made it into productive fruiting stages during the season, while 41% (178/436) remained viable but were delayed by timing and temperature limitations.
Contamination rate
We observed contamination in 26 blocks, which equals a contamination rate of:
26 / 436 = 5.96% contamination
For an outdoor, low-tech system using pasteurization (not sterilization), this contamination rate is considered commercially acceptable and supports the feasibility of atmospheric steam pasteurization for farmers without indoor lab infrastructure.
Key observation: once blocks were moved out of the tunnel during extreme heat and stored in the barn basement, contamination became minimal, suggesting that heat stress was a major contributor to contamination events.
2) Yield Results and Biological Efficiency (BE)
Harvest measurements
We measured yield using:
-
fresh harvest weight (lbs)
-
dry yield weight (grams)
-
estimated biological efficiency (BE)
In 2025 we harvested:
-
14.5 lbs fresh Reishi fruiting bodies
After dehydration, we processed:
-
2,194 grams dried Reishi
We observed that Reishi lost approximately 68% of its fresh weight during drying, which is lower shrink loss than many culinary mushrooms (often closer to ~90%).
Biological Efficiency (BE)
BE is calculated as:
fresh mushroom weight / dry substrate weight
Using our production data:
-
fresh harvest = 14.5 lbs
-
dry substrate weight estimate = 535.2 lbs
(232 productive blocks × ~2.31 lbs dry substrate per block)
BE = 14.5 / 535.2 = 2.7%
Discussion of yield
This yield was significantly lower than expected. A commonly referenced BE range for Ganoderma is 80–120%, though those figures often assume controlled indoor conditions, optimized genetics, and stable environmental parameters. Our season was impacted by two major limiting factors:
-
Temperature stress under the tunnel during peak summer
-
Late-season timing, leading to slowed growth and freezing exposure before many blocks could mature
Because 178 blocks did not fruit in time, we believe the season’s total yield is not fully represented by the 2025 harvest weight alone. These blocks are being stored in a cool environment and will be brought into production in spring 2026 to attempt recovery yield.
3) Environmental Monitoring Results (Temperature, RH, CO₂)
Temperature (primary constraint)
We measured temperature in two ways:
-
ambient daily lows/highs
-
substrate surface temperatures using an infrared laser thermometer
Between 7/9/25 and 7/21/25, we recorded:
-
ambient average low: 61.9°F
-
ambient average high: 81.45°F
During the same period, substrate surface temperatures under the tunnel averaged:
-
low: 86°F
-
high: 93°F
On 7/22/25, substrate surface temperature peaked at 113°F, while:
-
ambient high was 86°F
-
tunnel air temperature reached 101°F
This confirmed that the tunnel created a heat-amplifying effect that exceeded Reishi’s ideal fruiting temperature range and likely caused physiological stress, reduced primordia formation, and contributed to low BE.
Relative Humidity (RH) management
We tested three irrigation methods and measured RH using a sensor:
-
overhead misting (caused bacterial issues on fruit bodies)
-
soaker hoses (no meaningful RH increase)
-
hose + sprayer wetting mulch surface (successful)
Example RH response on 10/11/25:
-
before watering: 70°F / 70% RH
-
after thoroughly wetting mulch: 70°F / 85% RH within ~1 hour
This demonstrated that wetting the mulch surface directly was the most effective low-tech method for raising RH without waterlogging fruit bodies.
CO₂ monitoring
CO₂ remained generally below 2,000 ppm, aligning with recommended levels for fruiting development. After introducing chickens, CO₂ occasionally increased (peak 2,470 ppm) but was managed through ventilation (rolling tunnel sides up). CO₂ was not a primary limiting factor for fruiting success.
4) Poultry Integration Outcomes (Nutrient Cycling + Added Farm Yield)
Chickens were integrated to test nutrient cycling and decomposition support. Chickens:
-
foraged heavily in the mulch layer
-
contributed manure across the surface
-
increased fly/pest pressure during overlap with active mushroom fruiting
A production outcome from poultry integration was measurable:
-
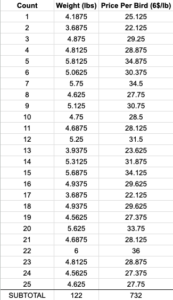
25 birds raised, 25 survived
-
122 lbs dressed meat
-
valued at $732 at $6/lb
-
average dressed bird weight: 4.9 lbs
While poultry integration improved fertility cycling and added farm revenue, it also introduced complications (CO₂ spikes, flies). In future cycles, we plan to separate mushroom fruiting from poultry grazing to reduce pest pressure and contamination risk.
5) Comparison to Conventional Systems / Previous Methods
Compared to conventional indoor Reishi cultivation
Conventional systems typically rely on:
-
sterile indoor environments
-
controlled temperature and humidity
-
single-use polypropylene grow bags
-
higher consistency and higher BE potential
In contrast, our outdoor system:
-
achieved a manageable contamination rate (5.96%) using pasteurization rather than sterilization
-
reduced reliance on controlled indoor infrastructure
-
demonstrated that low-tech farmers can produce fruiting bodies outdoors, but timing and temperature control are critical
The largest disadvantage compared to conventional systems was yield consistency and BE, which were strongly impacted by seasonal heat and late start timing.
Compared to typical mushroom production standards
Indoor cultivation often expects contamination rates near 0–2% under optimized sterile practices. Our ~6% contamination rate is higher than indoor standards but acceptable given the outdoor, low-tech design and was low enough to continue production without major failure.
Conclusion
The 2025 season confirmed that atmospheric steam pasteurization and outdoor Reishi cultivation can be successfully implemented with low contamination and minimal technology, but temperature management and seasonal timing are the primary determinants of yield and biological efficiency. The project also demonstrated that spent substrate can be returned directly to the grow site as a no-till soil-building amendment, supporting the project’s regenerative soil health goals. In 2026, we will begin production earlier, refine shading and cooling strategies, and trial additional genetics to improve yield outcomes and better match the outdoor growing window in Zone 5b.
Educational & Outreach Activities
Learning Outcomes
This grant project demonstrated that outdoor Reishi cultivation can be integrated into a small farm system in a way that supports both soil-building goals and income diversification, while also reducing reliance on conventional indoor mushroom infrastructure. The project produced clear lessons related to production feasibility, environmental limitations, waste stream realities, and operational timing.
What we learned through the project
We learned that low-tech atmospheric steam pasteurization is a viable method for preparing substrate for outdoor Reishi production. After processing 436 substrate blocks, contamination remained manageable, with an overall in-bag contamination rate of 5.96%. This confirmed that sterilization is not required to run a functional cultivation system at this scale, and that pasteurization can be an accessible approach for small farms without dedicated lab infrastructure.
We also learned that timing and temperature management are the primary factors determining success in outdoor Reishi production in Zone 5b. Although the tunnel provided physical structure and humidity potential, it also amplified heat during the peak of summer, which reduced primordia initiation and slowed or halted development. These conditions contributed to a low overall biological efficiency (2.7%) and resulted in a significant portion of blocks (178) not reaching primordia before the season ended.
Another major lesson was that substrate sourcing is a real constraint even when a material is considered a “waste stream.” Sawdust was more difficult to secure than expected due to competing demand, and the project required adapting by using hardwood fuel pellets for a large portion of the season. When raw oak sawdust became available later, we observed differences in substrate performance and suspected tannins affected mycelial vigor and colonization speed. This reinforced that not all woody waste streams perform equally, and the chemistry of the material can influence outcomes.
We also learned that outdoor fruiting strategies must be adapted to reduce exposure of substrate to open air. Bag management methods that minimized exposed surface area supported healthier conk development and reduced contamination during fruiting compared to methods that exposed the entire block.
Finally, we learned that integrating poultry into the system has benefits for nutrient cycling, but introduces complications when overlapping with active mushroom fruiting. Chickens increased pest pressure and raised CO₂ levels at times, even though both were manageable. The overlap between chickens and fruiting mushrooms also increased food safety considerations and required additional separation measures.
How this affected our farm operation
This project strengthened our farm’s capacity to produce medicinal mushrooms using scalable, low-tech systems and confirmed that we can operate without relying exclusively on indoor production. It also advanced our broader soil-building goals by producing a large volume of spent substrate that could be broken up and applied as organic matter across the grow site without tillage. In addition, the project supported our farm’s diversification through value-added processing: the dried Reishi produced in 2025 was sufficient to formulate a tincture yield with meaningful retail potential.
The project also directly informed our farm scheduling and infrastructure decisions moving forward. We now understand that outdoor Reishi production must be aligned tightly with seasonal temperature windows, and that production pacing is dictated not only by labor but also by pasteurization capacity and cooling time.
Did we overcome the identified barrier? If so, how?
The primary barrier identified in this project was developing a sustainable, low-tech outdoor cultivation system that reduces waste and can function on degraded land without heavy infrastructure. We overcame this barrier in part by successfully establishing a working system that produced fruiting bodies, maintained acceptable contamination rates, and generated usable spent substrate for soil improvement.
However, we did not fully overcome the barrier of achieving strong yields comparable to conventional cultivation systems. Environmental stress, particularly excessive heat in the tunnel during summer and the late start to the season, limited biological efficiency and reduced total harvest. The project clarified that the system is feasible, but that success at commercial yield levels will require better alignment with seasonal conditions and additional refinement in strain selection and site climate moderation.
Advantages of implementing a project like this
A key advantage is that the system can be implemented with relatively low-cost equipment and materials compared to indoor mushroom cultivation. Pasteurization can be performed using atmospheric steam without sterilizers, and the growing space can be created using affordable tunnel structures. The project also demonstrated that contamination rates can remain manageable even in outdoor conditions, making the approach realistic for farms without advanced sterile facilities.
Another advantage is that the system produces both a marketable crop and a soil-building amendment. Spent substrate can be applied directly as organic matter, supporting long-term improvement of degraded soils. This creates a regenerative loop where mushroom production contributes to land restoration rather than generating a waste product.
The project also creates opportunities for diversified revenue streams through value-added processing and education, and it increases rural access to medicinal mushrooms by producing them locally.
Disadvantages of implementing a project like this
The primary disadvantage is that outdoor production is highly sensitive to weather and seasonal timing. Temperature extremes can disrupt colonization and fruiting, and growers have less control than in indoor systems. This can lead to inconsistent yields, delayed fruiting, or lost production windows.
The system also depends on reliable sourcing of substrate inputs. Even when materials are framed as waste streams, supply may be inconsistent due to competition, transportation constraints, or quality variation.
In addition, scaling production is limited by the physical throughput of pasteurization and cooling cycles. This affects scheduling and can spread inoculation and fruiting across a longer period than desired. Poultry integration, while beneficial for nutrient cycling, can also complicate pest pressure and food safety separation requirements when performed simultaneously with fruiting.
What we would tell other farmers or ranchers if asked for more information
We would tell other farmers that outdoor Reishi production can be done successfully with low-tech pasteurization and simple infrastructure, and that contamination can remain within a workable range even without sterilization. We would also emphasize that environmental timing is the central factor in success: aligning inoculation, colonization, and fruiting with the appropriate temperature window is more important than maximizing the number of blocks produced.
We would recommend approaching the system as both a crop and a soil-building practice. Even when yields are lower than expected, the spent substrate contributes measurable organic matter to degraded sites and supports regenerative outcomes that are valuable beyond immediate mushroom harvest weight.
Finally, we would share that genetics and substrate quality matter. Culture selection, regional adaptation, and substrate sourcing influence colonization speed, vigor, and yield, and should be treated as major decision points when adopting a similar model.
Project Outcomes
This project confirmed that low-tech outdoor Reishi cultivation can be implemented on a small farm using atmospheric steam pasteurization and minimal infrastructure, while also producing a valuable soil-building amendment for degraded land. The work also highlighted several areas where additional study and refinement would improve replicability and performance for other farmers.
A primary recommendation for future study is identifying the most reliable seasonal production window for outdoor Ganoderma cultivation in northern climates. In Zone 5b, temperature proved to be the dominant limiting factor, and yields were strongly affected by summer heat under tunnel conditions and late-season cold exposure. A multi-year dataset comparing start dates, incubation timelines, and fruiting success would help establish clearer best-practice timing for farms in similar regions.
We also recommend further research into low-tech temperature and humidity regulation strategies for outdoor tunnel systems. While the tunnel provided structure and moisture retention potential, it also amplified heat beyond ideal fruiting conditions. Understanding how shade cloth density, tunnel orientation, mulch type, airflow, and evaporative cooling influence substrate temperature and relative humidity would help refine this model for broader adoption.
Another valuable area of study is strain selection and regional adaptation. Culture performance appears to play a major role in outdoor success, and future work comparing Ganoderma lucidum with more locally adapted varieties such as Ganoderma sessile may improve yield stability and biological efficiency in Midwest growing conditions. Continued spore collection and selection from fruits produced outdoors may also support the development of strains better suited to farm-scale outdoor systems.
We recommend further investigation into substrate sourcing and material chemistry, particularly when using raw sawdust from different tree species. Our observations suggest that tannins in oak sawdust may reduce vigor and slow colonization, and that simple pre-treatment steps may improve performance. Standardizing how waste-stream substrates are prepared would strengthen consistency for farmers replicating this approach.
Finally, we recommend additional evaluation of integrated poultry systems in relation to mushroom production timing. Poultry integration supported nutrient cycling and soil fertility, but overlapping chickens with active fruiting introduced pest pressure and increased complexity. Future work comparing sequential versus overlapping integration would help determine the most practical and lowest-risk approach for diversified farms.
Overall, we believe this project supports SARE’s goals by demonstrating that fungi can be used as a practical tool for soil regeneration and farm diversification, and that low-tech systems can be accessible to small-scale producers when paired with realistic timing and infrastructure expectations.