Final report for FNE18-911
Project Information
This project sought to determine the effectiveness of dragging pastures for reduction of fly populations in pastured beef cattle in the mid-atlantic region - specifically targeting face and horn flies that are known to breed in fresh cattle manure. We built 30 traps from cones of window screen with collecting jars on top - these capture adult flies emerging from manure pies 1-3 weeks after eggs are deposited in the fresh manure. In 2018 we compared emergence from undisturbed cow pies versus those that were smeared with a drag unit made from weighted steel bed frames pulled behind a gator - five round of trapping were conducted in 2018. In 2020 we manually smeared cow pies with a hand tool to enable accurate doubling and tripling of cow pie size, completing four rounds of trapping in 2020. After trapping, flies were sorted to species and tallied on a per trap basis. Statistical analysis of the results did not show a clear correlation between our disturbance methods and reduction in reproductive success of face or horn flies - in fact the only statistically significant correlation was an increase in face flies emerging from smeared cow pies in 2020. Some rounds of trapping did show reduced fly numbers in disturbed treatments, so we conclude that pasture dragging may reduce fly populations in the mid-atlantic in some situations although our data do not support recommending this as a cure-all practice. Hot, dry weather and using a more aggressive drag that scatters manure (like a chain harrow or more sophisticated commercial drags) seem likely to produce improved results - farmers are encouraged to apply careful observation to assess whether this practice is worthwhile on their operations.
Outreach for this project included:
"Fly IPM for pastured beef cattle" presentation at 2019 PASA conference - over 25 sustainability-oriented livestock farmers attending with good reviews.
Fly IPM Fact Sheet- summarizing four years of work on non-chemical fly management at Dickinson College Farm.
Article describing the project submitted to Stockman Grass Farmer (submitted May 2021)
Scientific article for publication in academic journal (submitted May 2021)
Our project objective has been to study a promising non-chemical management tactic for fly pests management: pasture dragging to break up manure pies with a light harrow. This practice is sometimes recommended for fly management (e.g., Kaufman, 2011; Rutherford, 2015) because the larvae of horn flies and face flies (together referred to as ‘filth flies’) feed in cow pads; and thus it is conceivable that spreading the manure will reduce fly survival through desiccation. Field trials at the project farm showed anecdotal success in 2017. Our literature search did not find any documented scientific studies of this tactic, so our aim for the 2018-2020 summer field studies was to do just that.
Cattle in pasture settings suffer a reduced quality of life due to a complex of fly pests that feed on their blood and facial secretions. Prominent among these pests are the horn flies (Haematobia irritans), which feed on cow blood 20-30 times per day while resting on backs and sides of cows (Oyarzún et al., 2008). Cows may be afflicted by hundreds or even thousands of horn flies at once and are not immune to the annoyance nor to the loss of blood. Another prominent pest species is the face flies (Musca autunma), which feeds on the mucus secretions of cow’s eyes and noses. These flies are less directly harmful but problematic because they vector pink eye (Pickens and Miller, 1980). In response to fly pressure, cows congregate and swat their tails—behaviors that significantly reduce their feeding and growing efficiencies. This combination of irritation and defensive behaviors in cows have a significant economic cost. In the cattle industry nationwide, horn flies account for estimated losses of 730-876 million US dollars (Watson et al., 2002). Even greater losses occur when the cost of insecticides are considered. Yet insecticides are not always effective. Due to the development of insecticide resistance in horn flies (Watson et al., 2002) as well as the harmful effects of insecticides on non-target organisms, an integrated pest management (IPM) approach is needed that employs diverse tactics to reduce reliance on insecticides (Fincher, 1999; Kogan, 1998; Oyarzún et al., 2008).
Raising beef cattle on pasture is a sustainable alternative to conventional feedlot beef production with many potential benefits to farmers, including reduced energy consumption, sequestration of carbon in pasture soils, and access to high-end niche markets. Improving pastured animal welfare and productivity via reduced fly pressure has potential economic value for livestock producers across the Northeast. This efforts of this project should also be of interest to pasture based dairy farms.
The Dickinson College Farm operation includes a herd of pastured beef cattle – between 20 and 30 animals are on the farm depending on the time of year. Breeds raised are black Angus and Angus-Herford crosses. The herd includes cow-calf pairs, a bull, young and finishing stock. Direct market freezer beef is the main product sold, along with some young stock sold to other local farms. The cattle are third-party certified grass-fed and Animal Welfare Approved (AWA) by A Greener World. The AWA program provides guidelines for animal husbandry to ensure low stress, humane treatment. The farm practices management intensive rotational grazing, moving the cattle daily through small paddocks on about 45 acres of pasture. In the winter the cattle are fed dry hay on a covered heavy use area. The farm operation also includes 10 acres of certified organic vegetables, grass fed sheep, lambs, and a small flock of pastured laying hens.
Filth fly management has been attempted historically through a range of chemical and biological practices. Chemical management with synthetic pyrethroids, organocholorines or other nerve toxins administered through ear tags, sprays, powders and dip tanks have been employed successfully on short timescales (Oyarzún et al., 2008; Pickens and Miller, 1980). But it is well documented that filth flies develop resistance to these compounds (Oyarzún et al., 2008; Pickens and Miller, 1980). Moreover, this approach is generally contrary to the goals of sustainable agriculture for health and environmental reasons, and is somewhat limited by the stipulations of organic certification.
By contrast, biological and mechanical control methods have potential to be employed sustainably. Biological control in particular is an attractive option in some systems. In dairy barns, for example, parasitoid wasps that lay their eggs in filth fly pupae have been employed quite effectively. Unfortunately, commercially available parasitoids have low dispersal ability and do not favor the pupae of horn flies and face flies, rendering them ineffective in pasture situations (Fincher, 1999; personal communication with Spalding Labs).
Use of free range laying hens to disperse manure pies is another biological control option. Trials of this method at the Dickinson College Farm proved problematic due to chickens wandering out of the cattle pasture and into neighboring vegetable fields. Fencing chickens with portable electric mesh nets was found to be incompatible with the pace of cattle advancing across the farm. While hens did demonstrate an ability to disperse cow pies, the area covered by the chicken flock was much smaller than the area soiled by the faster moving cattle herd. Thus, we have sought to evaluate mechanical pasture dragging as an alternative for diversified farms that do not have appropriately matched cattle and poultry operations.
Yet another biological control option is the use of dung beetles to out-compete filth flies. Scarab beetles such as the beneficial species Onthophagus taurus have been intentionally introduced into to the US because they have the potential to reduce fly larvae success by rapidly dispersing dung pads through their tunneling and burrowing behaviors. In the southern US the populations of O. taurus reach high densities and colonize dung pads by the hundreds, which effectively reduces the manure availability and quality, leaving fragments of manure pads that desiccate quickly (Roth, 1989). Our surveys at the Dickinson Farm as well as a survey from New York (Pimsler, 2007) show that O. taurus and other dung beetles are already present in the northeastern US, but their populations are much smaller: only about a dozen beetles per dung pad at the Dickinson Farm. Thus dung beetles are of limited utility for biological control of filth flies in our region.
In lieu of hoards of dung beetles, we are interested in the practice of mechanically spreading manure to promote desiccation and inhibit filth flies. This strategy is often suggested for filth fly suppression but its efficacy remains in question (Kaufman, 2011) because scientific tests of the practice have not been published. Further, since the mechanism of action is thought to hinge on desiccation, this tactic may be climate- or season-dependent. Hence, testing of the tactic in the northeast throughout the growing season is necessary determine whether farmers in this region would benefit from implementing it.
Farmers at the Dickinson College Farm have anecdotal evidence in support of this tactic. In 2017 farmers in consultation with investigator Dr. Jason Smith trialed several methods for low input fly control on pasture. Methods trialed include feed-through insect growth regulators, construction and regular use of a walk-through fly trap, application of commercial essential oil blends designed to repel flies, use of commercial hanging traps, and dragging. Since pasture dragging requires regular investment of farm labor and machinery use, but we did not yet have data that could show a cause-and-effect relationship, we sought to rigorously evaluate the effectiveness of this practice before promoting it to other farmers in the region.
REFERENCES
Fincher, G., 1999. Biological control of dung-breeding flies affecting pastured cattle, in: Great Plains Beef Cattle Handbook. Iowa State University of Science and Technology, pp. 1–3.
Kaufman, P.E., 2011. Applied management for fly control. University of Florida. http://animal.ifas.ufl.edu/beef_extension/bcsc/2011/pdf/kaufman.pdf (accessed 12.4.17).
Kogan, M., 1998. Integrated pest management: historical perspectives and contemporary developments. Annual Review of Entomology 43, 243–270. doi:10.1146/annurev.ento.43.1.243
Oyarzún, M.P., Quiroz, A., Birkett, M.A., 2008. Insecticide resistance in the horn fly: alternative control strategies. Medical Veterinary Entomology 22, 188–202. doi:10.1111/j.1365-2915.2008.00733.x
Pickens, L.G., Miller, R.W., 1980. Review article: Biology and control of the face fly, Musca autumnalis (Diptera: Muscidae). Medical Entomology 17, 195–210.
Pimsler, M., 2007. A survey of the dung beetles in cattle manure on pastures of an organic and a conventional dairy farm in New York state. Masters Thesis. Cornell University.
Roth, J.P., 1989. Field mortality of the horn fly on unimproved central Texas pasture. Environmental Entomology 18, 98–102.
Rutherford, B. 2015. 7 tools to fight the war against cattle flies. BEEF. http://www.beefmagazine.com/parasites/7-tools-win-war-against-cattle-flies (accessed 12.4.17).
Throne, J.E., Robbins, P.S., Eckenrode, C.J., 1984. An improved screen cone trap for monitoring activity of flying insects. New York’s Food and Life Sciences Bulletin 106, 1-3.
Watson, D.W., Stringham, S.M., Denning, S.S., Washburn, S.P., Poore, M.H., Meier, A., 2002. Managing the horn fly (Diptera: Muscidae) using an electric walk-through fly trap. Economic Entomology 95, 1113–1118. doi:10.1603/0022-0493-95.5.1113
The Dickinson College Farm is an educational program and a working farm, owned by Dickinson College. The farm includes 50 acres of permanent pasture and 15 acres of PCO certified organic vegetable ground. Livestock on the farm include the beef cattle herd (about 25 animals in total including bull, cows, calves, yearling heifers and steers) as well as 15 pastured laying hens and 30-50 sheep producing meat lambs and wool. The livestock operation gained Grass Fed and Animal Welfare Approved certifications by A Greener World in 2017. The farm is managed by three full-time year round managers, five apprentices, and a crew of student workers. Food raised on the farm is sold through the Dickinson College cafeteria, a Campus Supported Agriculture program, farmers market, and the local food bank. The farm is active in PASA and has a significant public outreach mission.
Cooperators
- - Technical Advisor (Researcher)
Research
2018 Field Season:
The 2018 summer field study was completed according to plan. The following is a summary of the steps taken:
In March we (PI Matt Steiman and Researcher Dr. Jason Smith) hired student Cecilie MacPherson to be our summer research assistant. Cecilie grduated Dickinson College in 2020, majoring in environmental biology.
Field work began in June. After an introduction to the cattle, flies, and manure management issues, the following initial efforts proceeded simultaneously:
Steiman built a pasture drag to be used in the study. After shopping around at landscape and equipment suppliers we elected to construct our own drag from materials on the farm. The goal was to make an effective yet inexpensive tool appropriate to the scale of our equipment (for this project we used a John Deere Gator off-road utility vehicle to pull the drag). We settled on a double gang of steel bed frames oriented with the long sides parallel to the direction of travel. The bed frames were chained loosely together to permit floating over obstacles, then weighted down at each end with 6X6 lumber. The whole unit was attached to the rear of the towing vehicle with heavy rope. This configuration effectively distributed cow pies in its path with one pass of the Gator.

Meanwhile Smith, Steiman and MacPherson worked on constructing the fly traps. The inverted cone screen emergence traps were designed to capture flies that mature from pupae on cow pies on the ground surface. Large outer cones were made from rolls of 48″ aluminum screen. A pattern was developed and traced onto flat pieces of screen with a carpenter’s crayon, then cut out using heavy scissors. Cones were formed by rolling the cut screens into a cone shape, then stapling the seam to a 3/4″ strip of wood using a staple gun. A small hole (1-2″) was left open at the top of the large cones.
Smaller cones were formed from scraps of screen left after making the large cones. These smaller cones were glued to the inside of lids for wide-mouth pint mason jars using an outdoor rated epoxy. To set the traps, large cones were supported vertically with a plastic or fiberglass fencing stake with the wide end down. Edges of the large cone were tacked to the soil surface with 6″ landscaping staples. The smaller cones with jars attached were set atop the hole at the top of each large cone. Flies emerging from the ground beneath the trap travel upwards along the screen into the jar at the top, where they are trapped and die within a few days. Collection of the flies for analysis was executed by removing jars with flies from the mason jar rings, then fitting them with a cap for transport to the lab. When necessary, flies were euthanized by inserting a cotton swap doused with non-acetone nail polish remover into the jars. Placing jars with flies into a chest freezer was employed for euthanizing as well when suitable.
A video of fly trap installation in 2020 can be viewed here: https://www.youtube.com/watch?v=81YRfUsRe60



In practice the traps proved to be quite effective - we have confidence that the majority of flies emerging from cow pies on the soil below were caught in the trapping jars.
During this phase Macpherson also constructed a cow pie measuring grid from a wooden frame with 1/2" hardware mesh stapled on tightly. The squares of the grid were highlighted with yellow paint in a pattern that made counting covered area relatively easy. This grid was laid over the top of studied cow pies and photographed before and after the dragging treatments to assess the area of each manure pile and the spreading effect of dragging.
The experimental design and time plans were worked out and field checked in early June. Working together we settled on the following program:
Cattle were moved from pasture to pasture daily. Within 1-2 days of cattle leaving a pasture, an area with a good concentration of manure piles was chosen and staked out to protect it against trampling by animals or equipment. 30 cow pies of similar size were identified and flagged - half as controls (no dragging) and half as experimental piles to be dragged. Each pile studied was measured, photographed and flagged with an identification number. Piles were grouped by location into control or experimental treatment categories in order to facilitate practical dragging using the 7 foot wide bed frame assembly. All manure piles studied were located close to one another within an area of less than 1/2 acre. Following pile ID and measurement, the experimental half were disturbed by one pass of the bed frame drag at low speed. The pies were then left alone, uncovered, for 5 to 7 days to allow for fly egg laying, development of larvae, and any predation of larvae or eggs by naturally occurring predators.
After 5-7 days, the cone traps were installed and left in place for three weeks. This length of time was chosen to allow development and emergence of all fly species of concern. During the trapping phase, Macpherson visited the traps daily to note fly emergence and fix any problems with the traps. If any trap appeared to malfunction this was noted in a log book to accompany the fly collection data. A research grade rain gauge and temperature sensing data logger were installed on a portable stake located within the trapping area - Macpherson checked these regularly to account for weather variation in the fly collection results.
A work plan was generated by Dr. Smith that allowed a subsequent set of cow pies to be flagged, measured and dragged near the end of the first trapping period, such that the traps could be redeployed to a following site quickly. This allowed us to run through five rounds of pile dragging and fly collection over the June - September period, in an effort to account for variations caused by temperature, rainfall, daylength or time of year.
Following collection of each group of flies, they were brought to a lab on the Dickinson campus and preserved in sample jars. Dr. Smith trained Macpherson to identify flies to genus using a dissecting microscope. After the first round of flies were collected, Macpherson spent many hours categorizing and counting flies, thus forming the raw data set for the project.
As a side effort, the project team developed a behavior monitoring protocol in an effort to find possible correlation between cattle behavior and fly load. Macpherson spent enough time observing the cattle herd to allow them to become familiar and comfortable with her presence. From this close position, she took photographs of individual animals, then followed these animals for a ten minute period, counting behaviors associated with fly irritation (foot stomping, back biting, tail swishing, and ear flicking). Macpherson later counted the flies in each photo in an effort to correlate easily observable cattle behaviors with actual fly load per animal. The hope of this side project is to link countable behaviors with economic or welfare threshold values for pest flies.

2019 Data Collection Efforts:
Considering that the mechanized manure dragging in 2018 did not reliably reduce fly emergence from cow pads (as explained below in results section), we questioned whether a more thorough dragging process would be capable of reducing fly emergence. To begin answering this question, we developed techniques to manually spread manure pads (using a hand rake) to 100% and 200%of their original size and assess the impacts of this on flies.
A group of livestock interns working for project consultant Jason Smith at Milton Hershey School (MHS) undertook this experiment in 2019 using the emergence traps that were constructed in 2018. They successfully spread manure and monitored insect emergence, but fly captures from manure at MHS were too sparse in all treatments to draw conclusions. We suspect this is due to the history of pesticide treatments of the beef cows at MHS.
2020 Field Season:
Given that our results from 2018 were inconclusive, possibly due to either the unusually wet weather in that grazing season or to the limitations of the custom drag device, we petitioned NE SARE to extend the project timeline to allow for one more season of monitoring using funds remaining in the original grant. This request was granted and the 2020 season proceeded as follows:
Stephanie Levin, a Junior studying biology at Dickinson College, was hired as the 2020 field assistant. Following a Covid quarantine period in late May, Ms. Levin (hereafter “Levin”) arrived at the farm to begin training and monitoring activities in early June.
In 2020 to control for inconsistencies in drag performance, we utilized the manual raking method for spreading cow pies described above under 2019. Following a single day of cattle grazing, animals were excluded from the grazing area and a set of 30 similarly sized fresh cow pies were identified, flagged and numbered. We sought pies that met the following criteria: relatively round or square with little to no extraneous material, greater than 6” diameter, located in a mostly sunny spot (not located in deep shady areas). Using a random number generator, numbered flagged pies were designated for one of the following treatments: control (no smear), 2x (smear to twice the width of the original pie) and 3x (smear to three times the width of the original pie). Numbered pies were measured before and after smearing with the results recorded in a field logbook. Also noted was whether each pie was deposited during daytime or night hours in an effort to analyze whether this impacted fly egg laying behavior.
After smearing, cow pies were left undisturbed for 5-7 days before deployment of cone traps. Trapping, fly collection, identification and sorting were as described above in the 2018 field season.


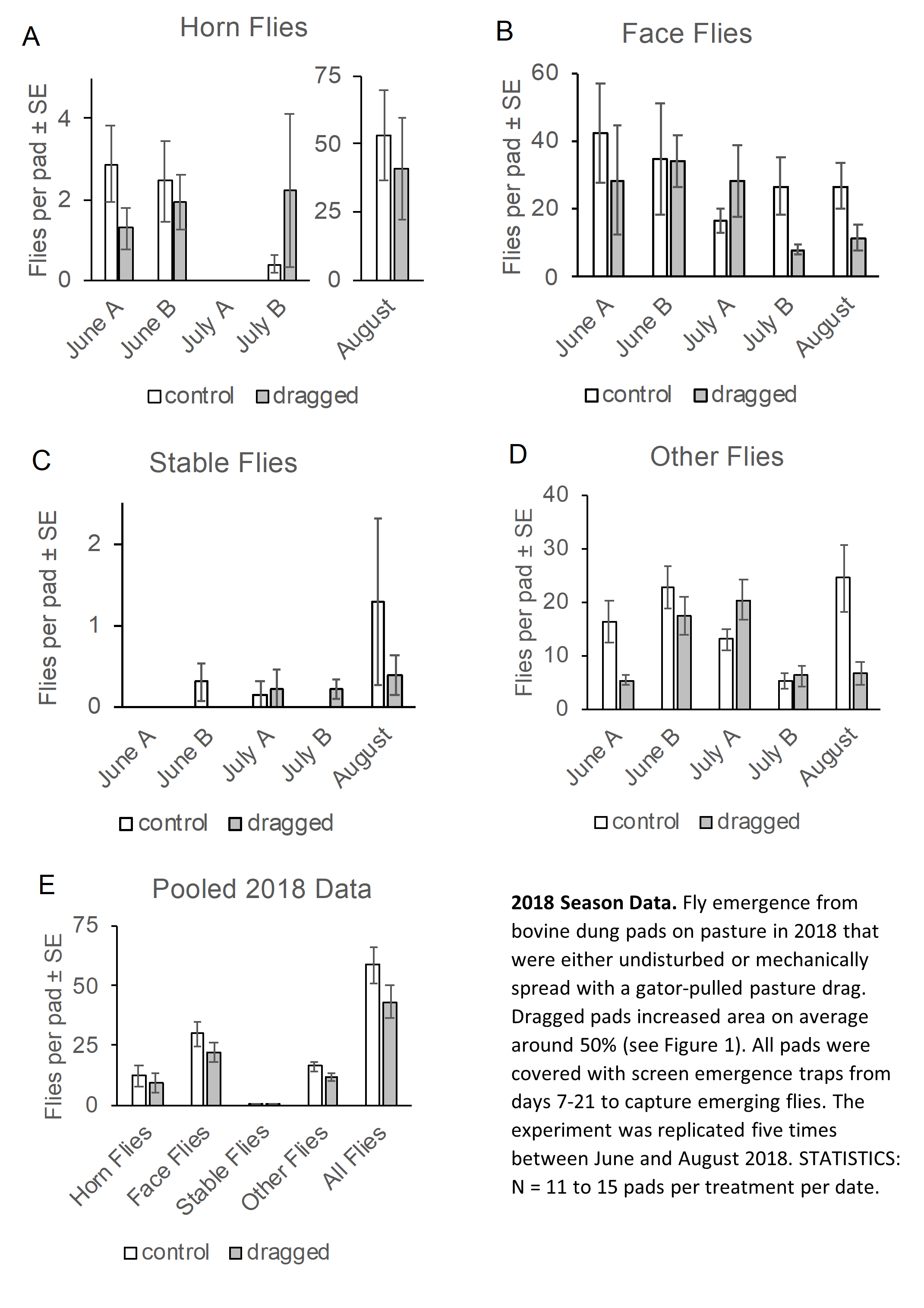
Flies collected from the traps were tallied throughout the fall of 2018. Dr. Smith conducted statistical data analysis in January of 2019 in preparation for the PASA conference in early February. Initial analysis shows that while dragging did have an apparent effect of reducing face and horn flies in some rounds of trapping, in other cases the opposite was true. Thus on a season wide basis (all five rounds of collection) there was not a statistically significant effect of the mechanized drag on horn flies or face flies in 2018.
Our 2018 efforts were hampered by the unusually wet summer weather in Pennsylvania throughout most of July, August and September, and interestingly the precipitation was a significant predictor of horn fly and face fly capture. This makes sense since the objective of pasture dragging is to spread cow pies so they desiccate faster and become less suitable habitat for fly pupation. In 2018 there were several weeks where drying of anything exposed to the weather was difficult.
A second round of data collection attempted during the 2019 fly season at the Milton Hershey School farm did not yield meaningful results because horn and face flies did not emerge from the manure pads. This is most likely due to historical use of synthetic antiparasitic treatments on that farm’s cow herd.
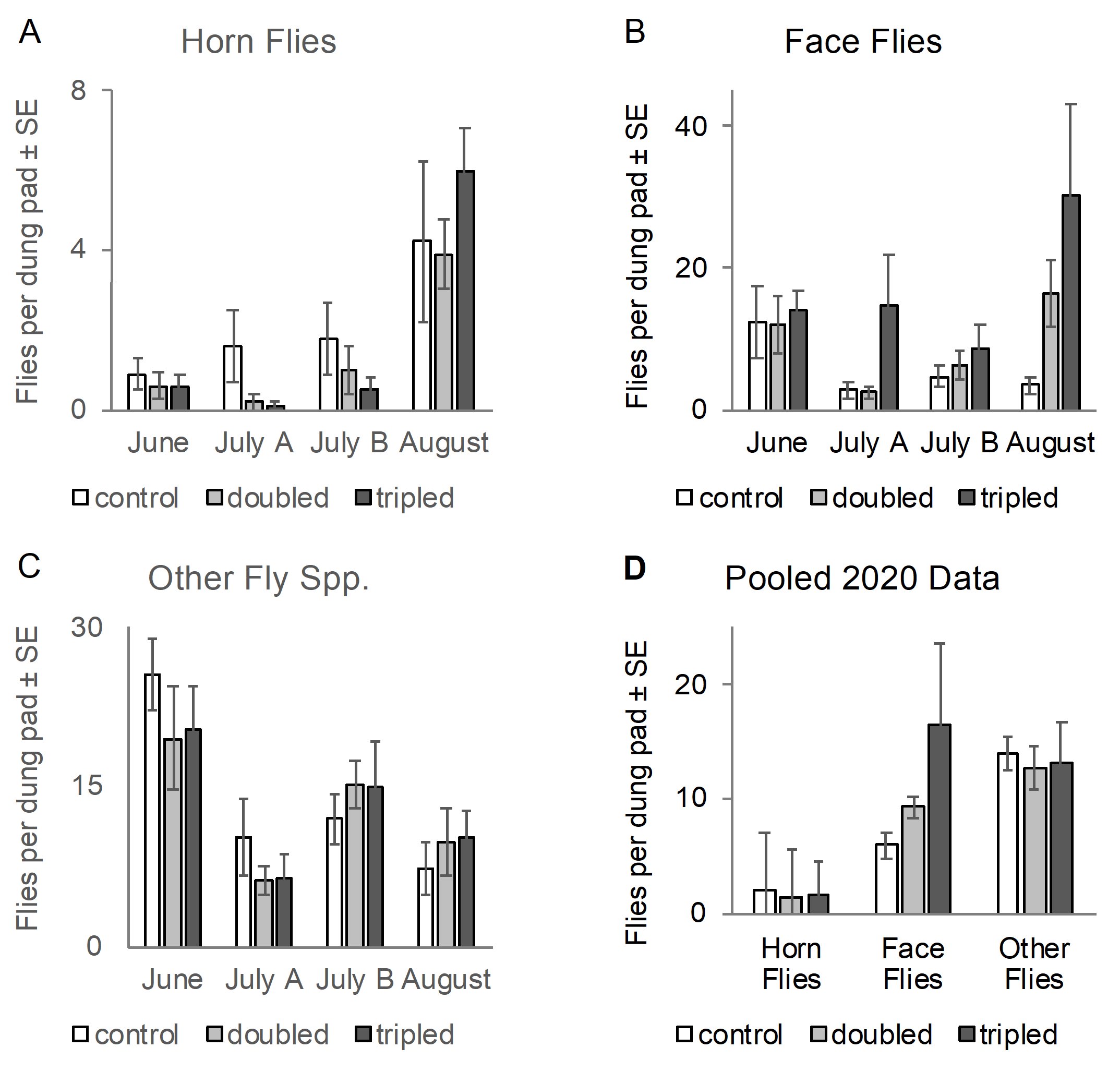
In 2020 we conducted four more rounds of the experiment using a manual drag technique (a hand tool) that ensured consistent spreading of manure to either one- (as a control), two- or three-times the original area. This practice was more labor-intensive than the tow-behind drag from 2018, but it allowed us test the effect of a thorough manure-spreading treatment without re-engineering the mechanized pasture drag.
On average, the numbers of horn flies emerging from doubled, tripled and untouched manure pads was very similar. Statistically there was not a significant effect of manure spreading on horn fly emergence, although there was a significant effect of precipitation.
By contrast, the drag treatment did statistically impact face fly emergence from dung pads on some of the dates. Surprisingly, the dragged pads produced more flies than the control pads. There was a statistical interaction between the Drag effect and Precipitation, meaning that the boost in fly survival following manure dragging might depend on the weather. One possible reason for the increase in face fly emergence could be that manure spreading removes the crusty layer from the manure pad, exposing a moist layer that could attract another round of oviposition.
2020 Season Data. Fly emergence from bovine dung pads on pasture in 2020 that were either undisturbed or spread with a hand-held scraper. Non-control pads were manually spread on day two after deposition to test effects of spreading on fly emergence. All pads were covered with screen emergence traps from days 7-21 to capture emerging flies. (Statistics: N = 9 or 10 pats per treatment per date).
Two seasons of field data did not support our hypothesis that mechanical disturbance of cow pies would reduce filth fly reproductive success and thus reduce fly loads on untreated pastured beef cattle. Remarkably, the only statistically significant effect of dragging was to increase face fly emergence on some dates of the experiment.
Based on these data we cannot recommend that farmers in south-central PA and regions with similar climates use pasture dragging to control face flies or horn flies. (It is an open question whether this practice can reduce fly emergence in more arid conditions where dessiccation or overheating could happen more rapidly).
However, we remind readers that pasture dragging can be a helpful strategy for breaking up long-lasting pads that suppress pasture regrowth and tie up nutrients. To preclude the unwanted effect of boosting face fly reproduction (as we saw in 2020), it might be advisable to allow pads to dry for several days before dragging them so that the exposed interior does not attract face fly oviposition.
A future study could explore whether a spring-tooth harrow or similar drag device that dissipates manure pads into small, separated clumps would more rapidly dry the manure and reduce fly survival. The tools used in our 2018 and 2020 field studies had more of a smearing effect which apparently left enough manure volume intact to allow face and horn fly reproduction. A flinging or scratching field tool might accumulate grass and other debris when in use, but farmer ingenuity can likely overcome this barrier through trial and error.
Farmers wishing to improve cattle welfare and performance without chemical inputs are encouraged to incorporate multiple fly reduction strategies, including minimizing water spillage & mud holes, managing spilled feed and hay accumulations, application of natural repellents, and use of walk through traps where appropriate. For further details on an integrated approach to fly management please download the "Fly IPM" fact sheet included as a product with this report.
Education & outreach activities and participation summary
Participation summary:
On February 7th, 2019 Dr. Smith (researcher) and Matt Steiman (Farmer PI) co-presented a 90 minute talk entitled “Pastured Beef Cattle Fly IPM” at the annual conference of the Pennsylvania Association for Sustainable Agriculture. This slideshow and talk included background on the farm’s beef operation, an overview of the different fly species and their life cycles, non-chemical control measures trialed at Dickinson, and detailed results and discussion from 2017 and 2018 field studies. Northeast SARE was credited as a supporter of the work. About 25-30 people attended – the room was full. Evaluations from PASA indicate a positive review of the talk (3.6 out of 4.0 rating) 18% of respondents indicated a relative increase in knowledge on the subject.
A Fly IPM for Pastured Cattle Fact Sheet was developed to summarize our work on non-chemical fly management for the past four years. Fly-IPM-Fact-Sheet-Dickinson-2021
Dr. Smith has completed a scientific paper on this study and will be submitting it for publication in the Journal of Veterinary and Medical Entomology in May of 2021. PI Steiman leads the effort for sharing through Stockman Grass Farmer, extension bulletins and similar publications. An article on the results of our fly management work was published in Stockman Grass Farmer October 2021. Dragging Pastures for Fly Control, Stockman OCT21
Learning Outcomes
Evaluations from the 2019 PASA conference (ten respondents) show self reported increased knowledge among 18% of the participants. Average rating for the talk was 3.6 out ot 4.
The public articles and fact sheet are just being released at the time of the final report for FNE18-911.
Project Outcomes
Given the results of this study, our farm has stopped using pasture dragging with our existing tools as a regular practice, since our data indicate it does not reliably provide fly management in our microlimate. We have adopted several of the other techniques described in the Fly IPM Fact Sheet. It remains to be seen how many farmers will respond to the acticle in Stockman Grass Farmer and our fact sheets by changing their practices.
In retrospect we could have benefitted by investing in a more sophisticated dragging device for this study. Instead of buiding our own drag for the project we could have purchased a commercial one such as those depicted here:https://www.youtube.com/watch?v=eQ3owUmyW8o&t=14s and here. https://www.youtube.com/watch?v=9YvczwPGd7s
Our farm-made drag appeared to work very well at disturbing the cowpies but did not result in the effect we were looking for. This provides a lesson that other farmers can benefit from - despite looking like beneficial practice, in terms of fly control this may only be effective some of the time (i.e. when conditions are hot and dry). Using an inexpensive but ineffective drag may be costing farmers more in the long run than a commercial pasture harrow designed for the job.
A follow-up study using similar methodology with an improved dragging device would help validate or debunk the value of such tools for fly control in the northeast. We feel confident in the effectiveness of our trapping method. One potential improvement would be addition of hoops, spreaders or more stakes to the cone traps (we used two stakes per trap). More spreading devices would ensure a consistent opening for fly capture across all traps.
This study should be of interest to pastured beef and dairy producers in the northeast region, as well as extension and ag support professionals in regions with similar climate. This information is being shared widely for the benefit of more cattle producers in the northeast region via our fact sheet, scientific article and trade article submitted to Stockman Grass Farmer.