Final report for FNE19-934
Project Information
Aquacultural Research Corporation in Dennis, Massachusetts was awarded a farmer grant from NE SARE to build a soda bottle upweller system, with a goal to determine the growth potential of the Eastern oyster, Crassostrea virginica, by volumetric comparison. Measurements were done on the volume (Liters/minute) of incoming heated seawater and on the density of the microalgae, Thallasiosira pseudonana. The two combine in a holding tank and feed the juvenile oysters. A TD 700 fluorometer was used to measure chlorophyll. Algae counts were also done in an effort to provide the oysters enough food to optimize growth. The seawater and algae flows going into the tank were adjusted. Adjustments of the flows going through the bottles were also made. The flows through the bottles were visually set ensuring there was enough tumbling motion. Liters/minute were then measured. In 2020 and 2021, numerous experiments were done in effort to identify whether growth was affected by oyster loading densities (by volume). Volume did not affect growth. There was, however, a pattern with growth being affected by the flow going through the bottles. In 2020, bottle flows had a range of 2-5 L /min. flowing through an average of 226ml. oysters loaded into each bottle with an average of 5.94% growth. In 2021, the bottle flows had a range of 3-6 L min. with an average of 284 ml. of oysters/bottle and an average growth of 8.9%. This research has provided insight into the optimal conditions for running the soda bottle upweller system. A scientific poster indicating a general summary on this project will be presented at Northeast Aquaculture Conference and Exposition (NACE) in April of 2022.
The goal of this project was to build a fluidized soda bottle upweller that is space efficient, labor saving and maximizes growth potential of oyster seed. In order to do this, juvenile oysters (less than one mm.) were put away at different population densities in the fluidized soda bottle upwellers. The upweller system was fed increasing amounts of algae and seawater over a period of 7-9 days and oysters were then evaluated. Percent growth was then calculated. No correlation was discovered between population density and growth rate. There was, however, a visual increase in growth with higher bottle flows. We therefore began comparing data on seawater flow rate/one ml of oysters. The objective then became comparing different bottle flows for different densities of oysters.
A primary limiting factor in many aquaculture facilities is physical space. Aquaculturists use a variety of tanks to culture juvenile oysters. Downwellers and upwellers are frequently used but occupy a large area of space. These types of tanks also require daily cleaning which requires labor. A fluidized soda bottle upweller system can house more oysters in a smaller space than a downweller. The movement of the oysters in the bottles help to keep the oysters clean by moving the waste out of the bottle and to the trough. This maintains a cleaner environment for the oysters so that they do not need to be rinsed daily, thus, reducing labor. At Aquacultural Research Corporation, a soda bottle system creates more space to grow an increased population of oysters. Having the soda upweller system is equivalent to running two large downwellers. The twenty bottle upweller system is 158“L X 33“W X 18.5“H and can house 20 million or more small oysters. If one were to attempt raising the same population of oysters in a downweller it would require a downweller tank measuring 256“L X 69“W X 24.25“H. The Soda bottle system therefore takes up 30% of the space that a 20 unit downweller would take up. With this increase in growing space ARC has been able to house more animals in the same footprint. Larger numbers can be produced which has helped to improve fulfilling oyster sales to local fishermen. By determining optimal conditions for the oysters in this system, we have been able to create a plan to maximize production at ARC.
Aquacultural Research Corporation was started in 1960 and is a shellfish hatchery, as well as a farm and wholesale operation. Quahogs, oysters and bay scallops are the primary shellfish that are spawned and reared at ARC. We then sell millions of seed to fisherman. These fishermen, as well as our own field crew grow these animals out so they are ready to be sold for market.
Cooperators
- - Technical Advisor
Research
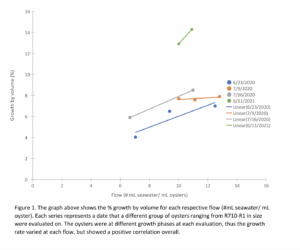
In order to conduct experiments, the fluidized soda bottle upweller had to be built. This process included sourcing and ordering parts, construction and troubleshooting of the system. The design of the system included 20 bottles in which oyster seed would be fluidized, with a mixture of seawater and algae flowing through each bottle. Algae density, seawater flow going into the main soda bottle tank and seawater temperature were also carefully monitored. Seawater flow going into the main soda bottle tank was adjusted based on the population of animals in the system. For example, as much as 25L/min. seawater flow per 11 bottles was required in order to maintain a pH of 7.8 or higher. A goal for algae density equal to 2,000 cells/ml. and a temperature of 25-26 degrees Celsius was set. A cleaning schedule was also implemented. Keeping the system clean is an essential step in rearing healthy animals. This cleaning schedule was less labor intensive than a large sized downweller which held an equal population of animals. In ARC’s 2020 season, experiments started with comparing percentage growth with initial population density. Initial volumes of 150ml. to 600ml. of oysters (less than 1mm) were placed in several bottles. Feed numbers for the total volumes of oysters put away then have to be calculated. ARC has guidelines set for feeding oysters. When the oysters are first measured and put into the bottles a standard feed number is calculated. For example, at an average cell density (2,000,000 algae cells/L) , 300L of algae will be added to the algae holding tank for 600 ml. total of R600 oysters. This tank will then be diluted with seawater so that the total volume of the algae holding tank is approximately 1,000 L. A flow rate of approximately .9 Liters/minute will be added to the soda bottle holding tank. This flow will be set in the afternoon. These measurements of food and flows may vary depending on the density of the algae. The next morning a sample of the soda bottle seawater tank will be taken. The remaining algae density in the tank will be measured. If the a.m. remaining food is less than 30,000 Thalassiosira cells/ml. we add 15% onto the initial feed number. If there is too much remaining food in the morning, for example, 40,000 thalassiosira cells /ml., we can feed the same amount as the day before. On average we are increasing by 5-20% each day of the thalassiosira pseudonana (based on the remaining a.m. algae) In doing this, we provide readily available food in the water column to the oysters at feeding time. The seawater was also increased going into the soda bottle tank. A starting seawater flow was set on the first day the oysters were put into the soda bottles. For 600 ml. of R600 for example, a seawater flow of 4L/min. was set. Ph’s were done of the water in the soda bottle tank in the morning. If the PH was 7.8 or less we would increase the seawater flow rate by 1 L/minute. The goal was to maintain a PH of 7.8 or greater. Each day that feedings were increased seawater flows were often increased due to an increase in waste After 7-9 days of giving increased food and seawater, volumes were done on the oysters and percentage growth was calculated. After several growth vs. density experiments were done, it was determined that there were no conclusive patterns with population density and growth. The project therefore started comparing the bottle flow rates ((#ml. seawater and algae mix/min. bottle flow) / 1 ml. of oysters) to growth. In 2020 and 2021, ¾” elbows and ½” i.d. tubing was attached to the bottle outflow on each bottle in order to measure the flow (Liters/min.) going through the bottles. The flows were first set by adjusting the flow of seawater through the bottles until the fluidization of the oysters was maximized without losing oysters out the outflow. When a maximum flow of seawater was moving through the oysters, bottle flows (L/min.) were measured. After 7-9 days of running the system at these bottle flows with steadily increasing amounts of Thallasiosira pseudonana and seawater final observations were made. Microscopic observation was performed on the different groups of oysters which assessed their overall health. Percentage growth was calculated using the equation: %Vol= [((final volume/initial volume)^1/n)-1] x100



The initial goal was to determine the ideal population of oysters (less than 1 mm) in size to place in a fluidized soda bottle that would yield the highest growth rate. There was no correlation between population density and growth rate. There was a positive correlation between bottle flow rates and growth (figure 1 & figure 2).
With increased flow there is likely a higher food availability. Since each spawn can grow at different rates it is expected that the percentage growth will vary for the same flow rate, but still maintain a positive correlation. When each spawn was evaluated, the spawns were at different ages and therefore had different growth rates. Some of the animals were at the top of the spawn where the fastest growers are found, while others were in the middle of the spawn where animals exhibit an average growth rate. This helps to explain why the growth rates varies at each flow rate, but the averages show a positive correlation. Overall, the growth in 2020 was lower due to lower flow rates as the system was still new to us and we were at the beginning of designing our experiments.
After running the system, numerous improvements to the fluidized soda bottle upweller were made. In the beginning, we had a 3 “½ pipe to catch the bottle outflow which overflowed. We therefore made a larger 3 “¾ pipe which held all of the outflow from the bottles. In addition, we started out trying acrylic marbles that were less dense than the oysters. We therefore replaced them with 1” glass marbles. The weight of the glass marbles kept the marbles at the bottom of the bottle. As a result, it kept the oysters in the bottles when the bottle system was on and off. Additionally, we found that hydrogen sulfide was forming in bottle # 20 (bottle at the end, farthest to the right). Hydrogen sulfide was forming due to a lack of flow, which caused a lack of oxygen. In order to prevent this an elbow and a valve were installed at the end of the pipe that feeds the bottles seawater. This creates a constant flow through the pipe that feeds the bottles. The flowing oxygenated water will prevent hydrogen sulfide from building up in bottle #20. Another improvement we started was measuring flow rates. We did this by connecting an elbow to the bottle outflow. Tygon tubing was then connected to the other end of the elbow to divert the water from the halfpipe to a flow meter.
In conclusion, our experiments yielded valuable insight to the design and operations of the fluidized soda bottle upweller. We determined that bottle flow is more important to growth rate than population density. Aquaculture facilities will likely benefit from the knowledge of our soda bottle system design as well as the experiments performed. Many Aquaculturists have worked with soda bottle systems and measure the flow of the bottles by eye only. By setting the bottle flows by eye and then measuring the actual Liters/minute bottle flow we have provided useful information as standards for running the system. For example, different volumes of oysters grow best at different flows.
The comparison of the space required for a commonly used 20 unit downweller vs. a 20 unit bottle system is also valuable information. A 20 unit downweller occupies 70% more space to house the same amount juvenile oysters. This is very useful information to an aquaculturists building a facility or trying to maximize its juvenile oyster capacity. ARC’s bottle system is also an indoor system with an algae holding tank. Many aquaculture facilities use unfiltered seawater and natural algae, instead of cultured algae.
Education & outreach activities and participation summary
Participation summary:
Upon completion of this project, we will present the findings at the Northeast Aquaculture Conference and Exposition in April of 2022 by showing a scientific poster of the bottle system and the experimental information.
Tours were offered of ARC hatchery which included the larval and juvenile rearing of the Eastern oyster and how the soda bottle system is space efficient. These included:
- Cape Cod Cooperative extension - Introduction to Shellfishing Course
- 3rd grade Cape Cod Shellfish Education Program for Monomoy School District
- International Shellfish Buyers as part of New England Seafood Exposition
Learning Outcomes
The 10 farmers/staff of the hatchery has improved upon the original design and gained knowledge in how to operate the system.
The bottle flow is more important to growth rate than the population density.
Project Outcomes
The fluidized soda bottle upweller has allowed ARC to be more space efficient and reduce labor. On average, it saves 30 minutes/day of physical labor and 1 ½ hours / day of time filling tanks with seawater. Therefore, it saves 2 hours / day of paid time. It has also given ARC more space to house more oysters thus contributing to an increase in production. It is not known at this time how much of an increase in production this soda bottle upweller system has provided. Each year a spawn schedule is created in order to provide oyster seed to be sold. A noticeable ease has taken place when creating the schedule since the bottle system was built. Having more space to put the oysters is apparent, it however, cannot be put into a percentage at this time. It will take a minimum of two years in order to determine the percent increase in production the soda bottle system has provided.
In the future, we have a few more problems that need to be addressed. One problem is that air gets into the system and bubbles attach to the oysters, causing them to float into the trough. We will attempt to fix this by adding an air bleed off to the system. This bleed off will be plumbed before bottles with a piece of PVC pipe and a valve which will be placed above the inflow to the bottles. This system is also limited by the ¾” i.d. diameter bottle outflow. Enlarging the diameter of the ¾” i.d. outflow to 1”-1.25” will support an increased flow through the bottles. We also suspect that having longer bottles will also enable us to house a larger volume of oysters per bottle. The bottles in this system are 24” high. If they were 12” higher there would be more room for more oysters.
Further experiments also need to be designed in order to set standards to optimize the growth of the oysters. The ideal and practical bottle flows for maximum growth need to be established. The oyster size and volume will be considered when setting these standards.
ARC plans on to continue to use this soda bottle upweller. In the future ARC may build another soda bottle system along with expanding the seawater and microalgal production. The growth in the soda bottle system appears to be comparable to the downweller growth. ARC will also compare the % growth performance of the two systems in the future.
With the support of SARE , ARC has been able to provide valuable insight to the troubleshooting of building this system and growth experiments. Aquaculture facilities will likely benefit from this information.