Final report for FNE20-947
Project Information
 This SARE project sought to determine the ideal container depth of nursery containers for propagating taprooted tree species from seed. I raised 625 chestnuts (Castanea mollissima) in 6", 9", 12", 15", and 24" deep containers in 2020, collected growth data, then transplanted the healthiest seedlings within each group outdoors in 2021 to observe how differences observed due to container depth affected field establishment. A second round of data was collected after dormancy in the fall, and analyzed.
This SARE project sought to determine the ideal container depth of nursery containers for propagating taprooted tree species from seed. I raised 625 chestnuts (Castanea mollissima) in 6", 9", 12", 15", and 24" deep containers in 2020, collected growth data, then transplanted the healthiest seedlings within each group outdoors in 2021 to observe how differences observed due to container depth affected field establishment. A second round of data was collected after dormancy in the fall, and analyzed.
It was found that while the 6" and 9" groups produced a significant amount of aerial growth over the other groups in the nursery, this advantage did not extend into the field year. Instead, the 12", 15", and 24" groups demonstrated roughly double the growth rate compared to the 6" and 9" groups. The 15" test group demonstrated the most robust second year growth.
So far, survivorship in the field is at 98.9% with only four deaths - one in all but the 9" experimental group.
It remains to be seen how differences in container depth will affect the chestnut trees long term, but we will be using 15" deep containers when working with taprooting seedlings in our nursery going forward given the initial results. The 24" test group, which aimed to simulate uninhibited taproot growth of direct seeding, does show promise as a competitive propagation method, but needs more years of comparative data, and needs special consideration for care and protection going directly into the field.
I conducted outreach primarily online through farming-focused online groups and forums, with a few email consultations and on-site visits with individuals over the course of the projects. The online outreach cast the widest net, reaching approximately 38,000 individuals with some saying it would affect how they would approach propagating trees this spring, though it is impossible to know how many of these are professional or aspiring farmers versus small landowners and hobbyists. In addition to the online outreach, I wrote an article about the project that will be published in MOFGA's April 2022 newsletter.
This project sought to determine the effects that nursery container depth has on taprooted seedling development in air-pruned nursery pots, and how these differences translate to post-transplant survivorship and initial vigor under contrasting management strategies that simulate real-life application.
In the first year, I grew a total of 625 chestnuts from seed in air-pruning containers of equal diameter within five different soil depths (6", 9", 12", 15", and 24" - 128 seeds each). Our initial intent in the second year was to grow the chestnuts under two management strategies simulating agricultural vs. ecological restoration/forestry applications, having 10 experimental groups with 40 seedlings each, but it became apparent this was too broad in scope to successfully take on. I was concerned about what I could successfully handle while farming full-time, and worried upon consideration that deer browse might compromise a portion of the experiment where tree tubes wouldn't be used. For this reason, the decision was made to focus on growing the trees under agricultural conditions.
In the second year, ~70 of the healthiest trees from each test group were planted at 20' in-row spacing (20' between-row spacing) within five experimental groups of varying container depths: 6", 9", 12", 15", and 24". This aspect of the project sought to draw out if/which depth container produces the healthier and more vigorous seedling. Solid data would help inform purchasing decisions of nursery stock and propagation practices to improve project success rates.
Healthy plant stock is critical for post-transplant success and profitability in business ventures. The challenges of producing healthy root systems in nursery containers are well-documented and are of great importance to nursery growers, restoration ecologists, foresters, and farmers. This is especially true of taprooting perennials: "Tap-rooted plant species are particularly vulnerable to becoming root bound because they initially develop a central leader or taproot during germination, which only later develops lateral roots," (Burkhart 2006). Arizona Cooperative Extension agent Rick Gibson explains that "the taproot is a critical storage organ for water and food supplies," which "quickly finds water and nutrients" when a seed first germinates, and is a reserve during challenging drought years (Gibson 2019). Burkhart, in his paper, goes on to relate research projects that found California chapparal and oak woodland species root systems typically range from 7 to more than 16 feet deep in nature. It being the case that the primary root plays this important role, what are the near- and long-term effects of taproot pruning on saplings transplanted into the field? How should it inform the nursery best practices, and purchasing decisions of tree stock? This project sought to focus on the near-term implications of this question.
One thing to acknowledge is that containerized nursery production will always be an unnatural context for a seedling. It is a human endeavor with inherent constraints. At the same time, we as humans are often planting them into unnatural contexts. A farmers' irrigation, fertilization, staking, and cultivation decisions both have intentional and unintentional consequences to what happens within the soil profile. A plant's root system responds to take advantage of these cultural practices. I hypothesized that the ideal root system depends on the context trees are planted into. Provide a tree intensive care from above as one might see in an agricultural context, and a robust, initial system of lateral feeder roots could provide an edge. On the other hand, if one's intention is to plant a sapling and step away - as might be the case in an ecological restoration or forestry project - planting a tree with an intact taproot may hold the key to its survival and success.
In practice, the solution is unlikely to be black and white. This is an aspect of what this study sought to investigate. Is there a container depth that optimizes balance between depth for taproot growth and lateral root production? The results may have important implications for the ideal nursery stock with a view towards its planting context. How do the root morphologies we encourage in the nursery play out under either intensive and non-intensive management?
I tested this by germinating 625 chestnut seeds using air-pruning containers with the experimental variable being container depth (6", 9", 12", 15", and 24"; 128 seeds per experimental group). Data was collected on initial seed weight, followed by survivorship, taproot length, and caliper at dormancy, as well as photographic documentation. Saplings were overwintered in the farm's walk-in cooler.
In the second season, a selection of the healthiest ~70 seedlings from each experimental group were transplanted at 20' spacing into the field. The field was subsoiled prior to planting. Each tree received irrigation, mycorrhizal inoculant, biochar, staking and a tree tube for animal protection, and weed suppression using a 3'x3' weed mat. At the end of the season, survivorship, tree height, and caliper were measured for all groups.
This experiment was designed to determine the optimum container depth for nursery production of chestnuts, and to hopefully provide guidance that extends to other taprooted tree species. This data will hopefully inform best propagation practices and purchasing decisions for taprooted trees, thereby improving crop productivity by reducing tree replacement and labor costs. Ultimately, we as humans want to provide the optimum conditions for nursery plant growth to produce a transplant that thrives. My hope is that this project will contribute useful data toward this end.
I have been farming for 11 years total. Our own farm is entering into its sixth year of operation, and is a full-time endeavor. We produce strawberries for u-pick and mixed vegetables for wholesale on 4-acres. We are in the process of expanding our u-pick offerings with honeyberries (2-ac.), blueberry (1-ac.), kiwiberry (1-ac.), heartnut (1-ac.), pear (.5-ac.), apple (.75-ac.), pawpaw (.5-ac.), buartnut/butternut (.5-ac.), plum (.5-ac.), hazelnut (.33-acres), and high tunnel blackberry production. This SARE grant project will eventually add 6-acres of chestnut production to the farm. The farm grossed ~$128,000 in 2021.
The SARE grant project utilized several existing pieces of the farm's infrastructure, namely: the farm's JD1050, a single shank subsoiler, a PTO post hole digger, cone spreader, the walk-in cooler, and some of the farm's existing irrigation system.
Cooperators
Research
 2020 Project Changes: The project got underway with the seeding of the chestnuts in the nursery area in the spring of 2020. Each seed was assigned an ID# and weighed prior to being planted into their respective containers.
2020 Project Changes: The project got underway with the seeding of the chestnuts in the nursery area in the spring of 2020. Each seed was assigned an ID# and weighed prior to being planted into their respective containers.
It quickly became evident, though, that the 30" deep containers weren't feasible with the grow tubes. It seemed likely the weight would likely have many coming apart over the course of the experiment. I substituted on the fly with 24" soil depth as the largest container test group, believing that it would still be deep enough not to inhibit taproot growth.
To simplify watering, I set up an overhead system using irrigation materials we had on hand. One issue encountered, however, was that the identifying labels quickly began to fade under UV exposure, so I purchased aluminum tags and re-labeled all the trees. The season went smoothly, needing only occasional weeding.
In November, the chestnuts were removed from their growing containers and stored them in the farm's walk-in cooler for winter storage in contractor bags filled with moistened sawdust. I pulled the trees from storage and collected data on aerial growth, caliper, and taproot length over the winter months in the garage.
Due to Covid-19, I did not pursue any in-person outreach engagements, but posted several social posts to our farm's social media and to farmer forums I participate in online.
Materials and Methods
NURSERY (Year 1):
- 625 Chestnuts were germinated within our greenhouse in air-pruning containers 6.5" in width and of five depths: 6", 9", 12", 15", and 24" at 128 seeds per group. Each container was given an ID#. Initial seed weight was be recorded.
- Fully-ventilated ShellT grow tubes were used as air-pruning containers for their modularity in increasing depth while maintaining a constant container width.
- A 6.5" width was chosen as the pot diameter based on Dr. Carl Whitcomb Ph. D.'s work, who developed the first commercially available air-pruning container. Dr. Whitcomb observed that lateral root promotion tends to only occur ~4" back from an air-pruned root tip (Whitcomb 2003).
- 6" is the minimum container size selected in order to reflect the typical depth of a 1-gallon pot.
- A 9" container size reflects the typical depth of commercially available deep pots.
- Changes to the Experiment in 2020:
- The choice of a 24" container depth for the maximum size was used to ensure uninhibited taproot growth. I intended initially to use 30" as the maximum size, but was limited by the the capabilities of the grow tubes. 24"-depth was still enough to not inhibit taproot growth.
- I opted to secure the grow tubes using bulb crates purchased locally from Fedco Organic Growers Supply instead of building stands. This was a time efficient and cost effective alternative to building mesh bottom crates on shipping pallets I initially considered. I was able to fit five bulb crates on a pallet, and still achieve the air pruning benefits.
- Seedlings were removed from the containers in the fall, bagged in contractor bags filled with moistened sawdust, and stored in the walk-in cooler as opposed to being heeled in. Practically speaking, this was easier, but it also afforded me the time to collect data over the winter. I measured the aerial growth, caliper, survivorship, and taproot length of each chestnut, and took example photos of the root systems within each group.
-
- I separated out ~70 of the healthiest seedlings within each group to plant into the field 2021 instead of doing it through random selection. This was based subjectively on seedling size, a visual assessment of its root system in comparison with the others within its group, and rogued out seedlings that developed as double leaders from the embryo (having two trunks). I opted for this method because, as a farmer, this is exactly what I would do with any other agricultural crop. You grow more tomato seedlings than you plan to plant, for example, and select the best for the season.
FIELD (Year 2):
- In the spring of 2021, I used mason line to measure an a 20' PVC jig to measure the proper distance between each row.
- The rows were then prepped using a single shank subsoiler to loosen any existing compaction.
- The 20' jig and flags were then used to mark tree spacing in each row. I used a PTO-driven post hole digger to create each digging hole at each flag.
- Based on a recent soil test of the planting location (Front Lenz) I was able to obtain from my neighbor, I applied Solubor 20 and Sulfur 90 to correct a deficiencies, as well as biologicals in the form of compost tea and molasses using the tractor's sprayer.
- On April 23rd, the majority of the chestnut trees were planted into the field randomly with the help of the planting party. They were all soaked in a pre-plant transplant dip of fish hydrolysate with kelp, compost tea amended with a couple commercially-available biological mixes, and a rooting preparation of salicylic acid extracted from willow. Biochar and mycorrhizal fungi were applied to each planting hole. The sides of every hole were scraped with a cultivating hand tool to counteract any sidewall smearing that might have occurred from the post hole digger implement.
- Irrigation was laid out, 3'x3' weed mat and grow tubes with a stake were installed on each tree, and a planting map documenting the ID#'s was created.
- With the landscape fabric holding back weed growth immediately around each tree, I mowed alongside the trees a single time just ahead of my neighbor haying the field, which ensured their visibility. The grass was allowed to grow in-row ummowed.
- Barring slight topographical differences in the orchard, the chestnuts received the same amount of water throughout the growing season. I simplified the experiment to solely look at the effect container size had on field establishment. Our region received a much more regular amount of precipitation than in previous farming seasons.
- In November, at dormancy, I collected aerial height and caliper data for each tree. I compiled this data into a spreadsheet and shared it with a statistician, Michael Sofis, who volunteered his time to analyze the data. He and I met in January over Zoom to discuss the results.
- Changes to the Experiment in 2021:
- I relied on a soil test available from my neighbor who has historically hayed this portion of the field for his dairy operation instead of taking a soil test to send off as previously planned. He was still able to hay the field in 2021, but I mowed close to the chestnuts just prior so they wouldn't be overlooked. In response to the soil test, I spread Solubor 20 at 3#/ac. and Sulfur 90 at 10#/ac.
- I chose not to hire an excavator to dig a test hole to determine if issues of hardpan existed. Instead, I opted to preemptively subsoil all the planting rows. The time I spent subsoiling cost less than what it would have taken to hire an excavator to come to the farm for an afternoon.
- As mentioned, the biggest change was simplifying the overall experiment to focus on their development in an agricultural context. This decision was made for a couple reasons. I realized that the complexity of running two experimental sets with a total ten experimental groups was more than I could take on running the farm's production full-time. Second, I worried that the farm's deer pressure would likely compromise the results of the trees grown in an ecological restoration/forestry context. Overall, it felt more important to collect meaningful results for a portion of the proposed experiment than struggling to succeed overall. I ran five test groups in the agricultural context alone. This had a positive benefit of having greater number of chestnuts within each group.

In this section of the report, I will first lay out all the data collected in both 2020 and 2021. Following that, I plan to discuss how this information relates to my work as a farmer, and how I would consider applying it in future nursery plantings, including economic considerations.
Over the course of several days in the winter of '20, I pulled the chestnut seedlings out of the farm's walk-in cooler in batches to collect data of each seedling's aerial height, root length, and trunk caliper. All the data can be found here: SARE Chestnut Data - 2020 Nursery Data
| 2020 Nursery Data | 6" | 9" | 12" | 15" | 24" |
| # of surviving trees (out of 128) | 125 | 119 | 121 | 124 |
121 |
| Avg. Initial Seed Weight | .73 oz. | .71 oz. | .61 oz. | .57 oz. | .53 oz. |
| Avg. Aerial Growth | 21.29" | 22.02" | 17.19" |
15.11" |
15.43" |
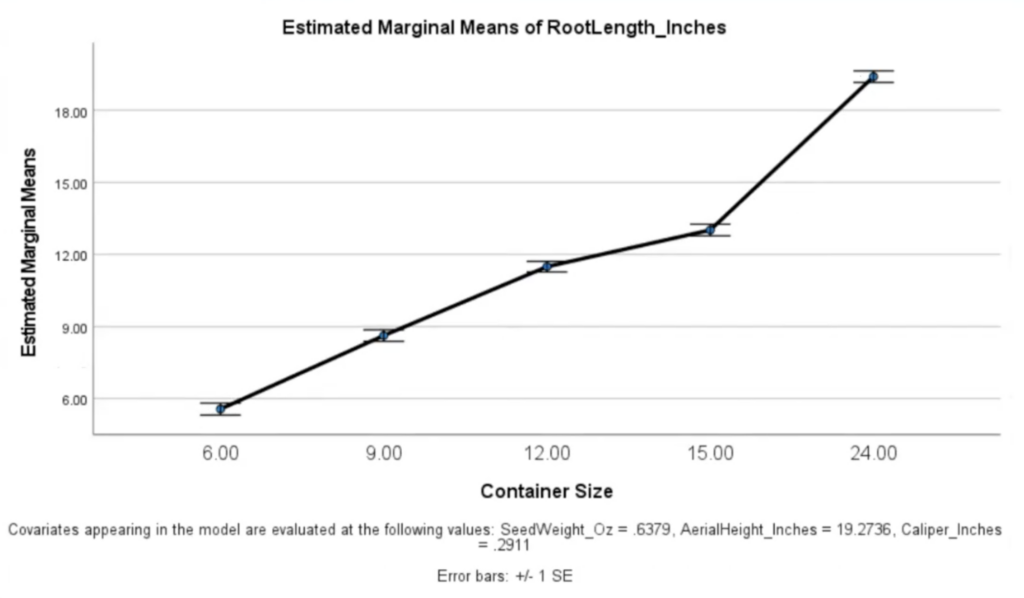
| Avg. Root Length | 5.64" | 8.64" | 11.12" | 12.57" | 16.79" |
| Avg. Caliper | .28" | .28" | .26" |
.25" |
.25" |
Out of 640 seedlings, there was a 95.3% survivorship in nursery stock. Recognizing that the data failed to capture the differences in root morphology I was observing between groups, I also took photos to record these differences. Below are three examples from each group.
Examples of 6" Chestnut Seedling Root Structures: 
Examples of 9" Chestnut Seedling Root Structures:
Examples of 12" Chestnut Seedling Root Structures:
Examples of 15" Chestnut Seedling Root Structures:

Examples of 24" Chestnut Seedling Root Structures:

From what I observed, the 6" and 9" depth containers had multiple smaller feeder roots emerging high up in the root column vs. the 12", 15", and 24" depth containers that more often presented a more robust taproot. At the 24" depth container, the root system was almost spindly as it appears to have been reaching to find its maximum depth. (It should be noted that I initially intended for the largest experimental container to be 30" deep, but I chose not to for logistical reasons. I struggled to lift bulb crates filled with even 24" of soil on my own, and worried they would fall apart with 30" of soil. I believed 24" would still be enough depth to not inhibit plant root growth.)
Following data collection, I selected the healthiest seedlings from each group to plant into the field. I struggled with and questioned if this were the right decision. Should the saplings I plant be selected randomly instead? I decided that any nurseryman/farmer given the choice would always be selecting the very best stock to go into their field, and that this was the proper course to take if the research I was conducting was to be most relevant to farmers themselves. The criteria I used to select seedlings was based on the visual quality of having a vibrant root system, and that the saplings were neither single-stemmed nor stunted in growth.
Analysis of 2020 Data:
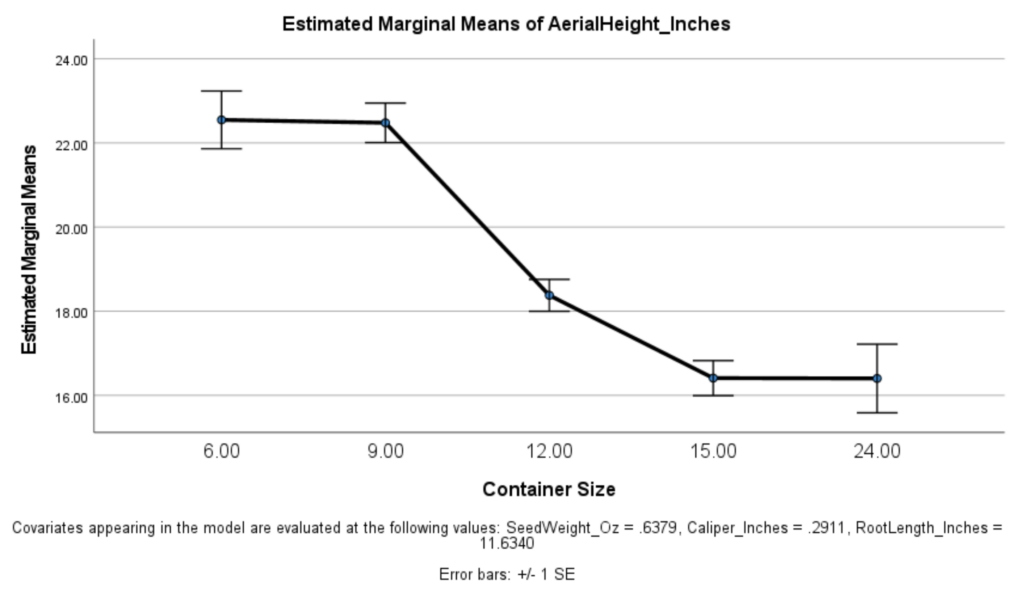
The graph to the left depicts a one-way ANOVA that was used to examine the relationship between container size and aerial height in 2020, when controlling for root length, caliper, and initial seed weight. As shown in the figure, when predicting aerial height in inches in 2020, there was a signficant effect that container size had on aerial height such that the smaller container size corresponded to greater aerial height (p <.001).
It should come without surprise that container size was highly predictive of root length (p <.001, ηp2 .801). Perhaps, having filled out the soil space afforded them, the 6" and 9" pots concentrated their remaining energy on aerial growth, while the 12", 15", and 24" groups put more first year energy into root production.
Seed weight and caliper as covariates in 2020 were also significantly associated with aerial height (p < .05), and root length showed a trend-level effect on aerial height in 2020 (p=.07).
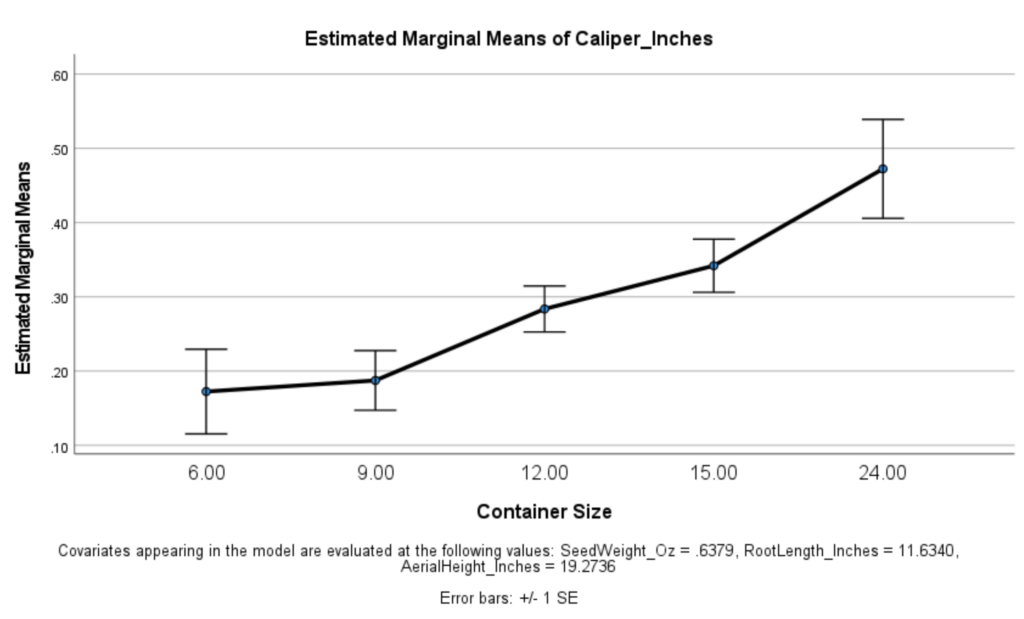
An opposing trend-level finding was found related to caliper size (p <.054). Generally, the greater the container size a chestnut seed grew within, the larger caliper size it developed.
Aerial height and root length both significantly related to greater caliper diameter in 2020. Based on the data, initial seed weight appears to be predictive of aerial height (p <.040), but not of caliper (p <.926).
It's important with all these graphs to understand that they are adjusted with consideration of covariates in mind (listed below each figure). That is, variables like initial seed weight that might affect outcomes are factored in the analysis.
2021
The chestnut trees were planted in April of 2021 over the course of several days, and with the help of one planting party. I altered the planting plan slightly, giving thought to easier maintenance by planting in 11 long rows. Seedlings were planted at 20'x20' spacing. 360 trees were planted in total with (69 in 6" group, 78 in 9" group, 74 in 12" group, 70 in 15" group, and 73 in the 24" group). The difference in population size between groups was unintentional.
Field year data was collected in the fall following dormancy, and can be found in full here: SARE Chestnut Data - 2021 Field Data
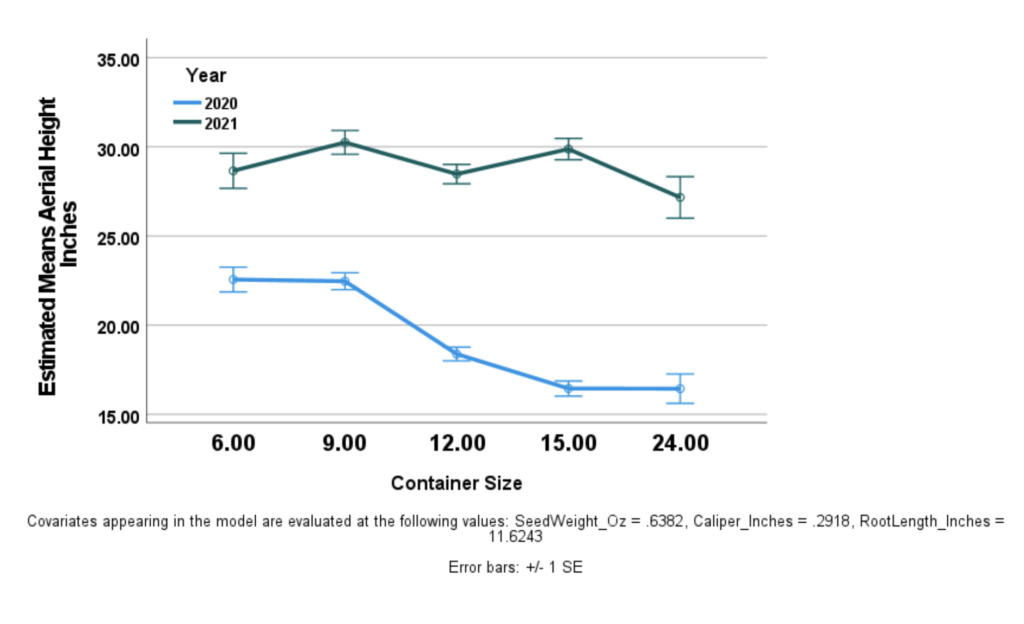
In the graph to the left the blue line represents the mean aerial growth collected in 2020 (controlling for initial seed weight and 2020 root length, and 2020 caliper), while the teal line is the aerial growth data collected in 2021.
A one-way repeated measures ANOVA controlling for initial seed weight, 2020 root length, and 2020 caliper was used to analyze the effect container depth had on 2021 aerial height in inches.
We can see a few things here. As depicted in the figure, a timepoint x container size significant interaction was found (p <.05) such that while smaller container size was generally associated with greater aerial height in 2020, this was not the case in 2021.
In other words, the correlative effect noticed in the 2020 data did not maintain in the field in 2021. Robust growth in 12", 15", and 24" experimental groups had these groups catch up in aerial height to the 6" and 9" containers that grew taller in 2020. The 24" group lagged slightly behind, and demonstrated a larger standard deviation within its group. Final average height between the 15" and the 9" groups was the same, but the growth of the 15" group was roughly double the 9" in the second year.
Sadly, I failed to satisfactorily collect data on caliper growth in the field this past fall to provide good analysis. Data was collected, but as can be seen within the document (SARE Chestnut Data - 2021 Field Data) many if not most of the values I recorded ended up smaller than those recorded in the nursery data (SARE Chestnut Data - 2020 Nursery Data). The average difference in caliper measurements from 2020 and 2021 between all groups -.025". I factor this to human error in measurement with the challenges of doing so in the field. I cannot claim it with any certainty, but perhaps such a negligible difference suggests little to no caliper growth between the nursery year and the field year. If I were to repeat the experiment a second time, I would consider marking where I initially measured the caliper of each tree with a paint marker, so I could be sure I was measuring the same spot between years.
Last things to note: survivorship in the field year was at 98.9% with only four deaths - one in all but the 9" experimental group; initial seed weight provided a trend-level effect on aerial height in general for 2021 (p <.061).
Discussion
I believe that the root system developed in the deeper container sizes in 2020 allowed them to take better advantage of resources once in the field. Alternatively, perhaps the reason the data shows less aerial growth in the 6" and 9" groups is that those saplings were simply expending more energy in 2021 to develop more robust root systems that they weren’t afforded in the nursery.
The current data suggests that the 15”-depth containers may lead to the best outcomes. While the 6” and 9” containers are the most economical to produce, I wasn’t impressed by the root systems produced. Meanwhile, the other groups showed nearly double the aerial growth in the field year.
Comparing the 12" and 15" groups, we see that the 15"-group averaged 2.96" more aerial height. If this growth trend continues, the 15" group might average one foot taller than the 12"-group by their first year of bearing (and this is considering it simplistically as linear vs. exponential growth). As seen in the figure below, the difference in cost for an additional 3" in aerial growth is only $.20/plant, or $21.60/acre (@ 108 trees per acre), and the benefit that soil may offer is potentially substantial.
| 2 ShellT Grow Tubes 6.5" diameter | Cost of ShellT Grow Tubes ($1.19/tube) | Cost of Soil @$145/cu.yd. | Total Cost |
| 6" (78.43 cu. in.) | $2.38 | $.48 | $2.86 |
| 9" (109.25 cu. in.) | $2.38 | $.68 | $3.06 |
| 12" (140.07 cu. in.) | $2.38 | $.88 | $3.26 |
| 15" (170.89 cu. in.) | $2.38 | $1.06 | $3.44 |
| 24" (263.35 cu. in.) | $4.76 | $1.64 | $6.04 |
Growing chestnuts in the 24"-deep containers demonstrated similar results to the 12" and 15" groups, but is far from a practical nursery practice. The directing seeding this group sought to mimic may have positive benefits that will only bear out in the coming years. Propagating taprooting perennials in this way just requires consideration to irrigation, to rodent and deer pressure, and for roguing out and replacing seedlings that are undesirable or that don't survive.
If it is shown that direct seeding produces the superior chestnut, you'll likely find me in the field sowing seeds in the future. For the time being, the benefits a nursery offers in providing ideal conditions in a manageable way means our farm will likely produce seedlings in 15"-deep containers going forward.
Conclusive results remain to be seen, of course. Perhaps the trees in the 6” and 9” groups took their first year in the field to focus energy on expanding their limited root systems, and will now keep track with the other groups this season. Perhaps the 24” group will gain an advantage over time by virtue of a deeper root system. Time is needed to answer important questions related to survivorship, years to bearing, and overall health. Presently, our field planting is at 98.3% survival with only a single tree in each group dying. How will those numbers look after their first winter? How will they look if they experience drought in the next years?
I intend to apply for a three-year SARE grant this fall to continue inquiry into this research project. Timing will allow me to collect 2022 data in the early spring of '23 before dormancy break, the continue with data collection for subsequent years in each fall. By 2025, at 5-years-old, it's possible that we may be able to capture data for when the first nuts bear from these trees.
Changes to the Experiment
- The biggest change to the experiment is that I limited my field research in year two to strictly the intensive/agricultural setup. I made the decision to reduce the overall complexity that I would still be able to come out with meaningful results at the project's end. Deer browse and weed competition in the field would likely have compromised the ecological restoration setup. Instead, all tree received protective tree guards, weed mats, irrigation, and biological preps.
- For the biggest experimental group, I reduced the container depth size from 30" to 24". It was exceedingly to successfully fill the containers with 30" of soil without running into issues of the grow tubes coming apart, and I was unable to lift the bulb crates to move them. 24" is what I settled on as still a safe bet for not inhibiting taproot growth and seems to have worked successfully based on the root length data collected from this group in 2020. There are areas within the study, particularly the ID numbers, that suggest a 30" planting depth. Readers should be aware that these refer to the 24" experimental group.
- I was able to obtain a recent soil test from my neighbor who has used the field for haying: Soil Test: Front Lenk
- I did not collect taproot diameter measurement in 2020. The smaller experimental groups did not have a discernible taproot to make meaningful comparisons across the experimental groups. As well, selecting a consistent place to measure the taproot on each tree would have led to wildly varying data points.
- I did not measure caliper at breast height in 2021. None of the saplings reached breast height by fall of '21. Instead, diameter at base height was measured, though these measurements proved unreliable.
- Outreach and coordination with advisors was limited due to Covid-19.
- Each experimental group had 70-78 trees per group, not 40 as initially outlined.
Miscellaneous Notes
If someone reading this report seeks to use this information to start their own nursery plants and is considering utilizing the ShellT grow tubes as I did, my recommendation would be to use the ventilated tubes with slits instead of the fully-ventilated tubes as I did for this experiment. Based on my experience, I believe that the slitted tubes offer fine root pruning, but are easier to water properly and don't dry out as quickly. As well, I believe you will get fine results using a single ShellT tube per seed. It is slightly small in diameter at 3.25" - I would prefer 4" - but I seeded extra chestnut seeds into single slitted grow tubes in 2020 and was quite impressed with root systems they produced. This is probably the route I would go in the future.
The research demonstrates that plant container depth does indeed have an impact on taprooted tree crop growth - to root morphology, caliper diameter, and aerial growth, but it revealed interesting results in the trees' establishment year. In the nursery year, the greatest aerial growth was exhibited by the 6" and 9" experimental groups. Caliper growth and root length, on the other hand, are significantly correlated to a larger container size. From a subjective visual perspective, the 15" and 12" root systems looked healthiest. The 6" and 9" test groups' roots seemed stunted, especially in comparison to their larger aerial growth, while the 24" group's root systems delicate and spindly as they appear to have concentrated energy producing a deep taproot into the soil.
In the establishment year, the aerial height advantage the 6" and 9" container groups exhibited disappeared. The 12"-24" group all grew just under double their counterparts. It's unclear if this is due to the 12"-24" groups being able to take advantage of resources with their deeper root systems, or whether the 6" and 9" groups were merely devoting more energy on developing a root system now that they had the space. If the trend continues in future years, it suggests that container depths of greater than a foot should be the starting point of consideration for producing robust taprooting tree seedlings, but that -at least with the experimental groups used in this study - a 15"-deep container is best. A modest $.20 increase in cost per tree produced a great root system that ultimately led to ~3" greater aerial growth compared to the 12" group. This small difference could provide dividends for a farmer down the line.
I hope to re-apply to SARE for continued research to see if the growth trend persists in the experimental groups, but going forward, based on the present data, I would err on starting seedlings for our farm's expanding u-pick in 15"-deep containers.
Education & outreach activities and participation summary
A secondary but smaller way I have conducted outreach is speaking with customers coming to the farm for the month of June for our strawberry u-pick. I'm unsure the number, but it's provided the opportunity to speak with people, some farmers, about SARE and our particular project.
Participation summary:
Conducting outreach amidst the pandemic has been the greatest challenge faced in executing this grant. Though vaccinated, my household continues to avoid large public gatherings and indoor events. What I have been able to do is make social media posts to raise awareness, speak with countless u-pick customers at the welcoming tent about the project, and give small tours of 1-4 people at a time to speak with them about the work and the experiment.
Additionally, I have provided consultation to five farmers over the course of this project by phone, by email, and - limitedly - outdoors in-person. This has led to two of those farmers to establish their own small chestnut orchards (20 and 50 trees), two who are still in the early planning/land prep stages, and one who has decided to pursue planting other perennials as part of their operation. A sixth reached out to me last week, and hopes to meet in-person in the spring when the trees have leafed out.
I have written an article for the MOFGA Newsletter on the project that will be published in April.
In February, I conducted a day's worth of outreach via social media on Reddit, Facebook, Instagram, GrowingFruit.org, Permies.com, and MOFGA's Journeyperson listserv. Reddit proved to be the most far-reaching way to get information out to others about the project results, being viewed by over 35.7k people within 24 hours.
Learning Outcomes
This project has provided initial data that is already affecting how others are starting perennials. I only know one other person to be a confirmed farmer due to doing most of my outreach by social media, but comments on the forums I posted demonstrate that others will be using the information whether on farms, homesteads, or backyard plantings.
Project Outcomes
This project has informed how we approach propagating taprooting perennials going forward. Based on the data results, we are using a 15" container depth for starting new seeds in the nursery this spring. Additionally, though not a taprooting plant, we have begun to apply deeper containers for our tomato seedling production.
The project results has been received positively through outreach on Reddit, Facebook, Instagram, GrowingFruit.org, Permies.com, and the MOFGA JP listserv. Based solely through the comments, it appears nine others are taking physical steps to apply the results found in the research. I know two farmers who are considering applying the practice to their work. Outreach for the project on social media reached ~38k individuals, so it may be that more will apply the research than I am aware of.
I intend to apply for a second SARE grant this fall to continue project research. The results have provided insight into the effect container depth has on the initial development of chestnut seedlings - with applicability to other taprooting tree species - but the long-term implications remain to be seen. The question of how container depth relates to survivorship over time, tree health and vigor, years to bearing, and yield is what is on my mind, and what I believe will have greatest relevant for farmers.