Final report for GNE16-134
Project Information
Reducing synthetic chemical inputs and tillage can have numerous benefits in agroecosystems, such as building soil health, promoting biodiversity, and reducing environmental effects of pesticides. In addition to these benefits, low-disturbance cropping systems may also enhance predator communities and the potential for predators to suppress pests. While tillage can kill or disrupt invertebrates, planting a winter cover crop may help sustain invertebrate communities by providing habitat and nutritional resources.
This work complemented an ongoing study investigating soil and cover crop management practices on invertebrates within reduced-tillage organic field crops. We compared four different reduced-tillage cropping systems, focusing on the corn phase of a corn-soybean-spelt rotation. These systems featured two different legume-grass cover crop mixtures preceding corn, termination of these cover crops either by rolling down into a mat or through mowing and incorporation into soil, and interseeding a three-species cover crop mixture into corn mid-season to increase success of establishment and extend the corn growing season. We measured crop damage from invertebrates, characterized invertebrate communities, and measured predation rate of insects in corn plots undergoing four different crop management strategies. Cropping system did not significantly impact the majority of pest and predator metrics we assessed, indicating that the overall influence of reducing tillage and incorporating cover crops through an organic rotation may outweigh more nuanced impacts of specific cover crop mixtures, termination method, and timing of cover crop planting.
We also identified the contributions of key predators within these cropping systems through video surveillance of sentinel predation assays and molecular gut content analysis of common generalist predators.These techniques ended up being much more challenging than anticipated. There is potential value in using these techniques but further refining of methodology is needed to ensure success using them in field crop systems similar to ours in Pennsylvania. The goal of this project was to provide insight into which management practices most effectively enhance predation and suppress damage from pests. We observed that these four cropping systems performed similarly in terms of predation and pest pressure, enabling growers to prioritize soil health, weed management, or other priorities as they decide on cover crop mixtures, establishment timing, and tillage practices.
Objective 1: Characterize and compare pest and beneficial soil-associated invertebrate communities in four organic corn production systems designed to reduce tillage and incorporate cover crops
Objective 2: Assess pest damage to corn in four organic corn production systems designed to reduce tillage and incorporate cover crops
Objective 3: Evaluate biological control potential of predatory invertebrates in four organic corn production systems designed to reduce tillage and incorporate cover crops
Objective 4: Communicate results from this and other research projects focused on reducing tillage and incorporating cover crops through presentations, informational handouts, and presentations at scientific and grower conferences.
The purpose of this project was to investigate the impacts of organic cover crop-based reduced tillage cropping systems on beneficial and pest insect communities and biological control potential. As market demand for organic grain increases in the United States, there is a greater need for approaches that facilitate productivity and profitability (Cavigelli et al. 2013). Crop damage from insects can result in impacts on crop yield estimated at $15.76 billion in the United States (Losey and Vaughan 2006). Biological control of pest insects by predators can lessen this damage by reducing pest populations. In this project, I compared the effects of cover crops and reduced tillage on beneficial predatory insects in an organic corn with the long-term goal of improving the sustainability of agronomic cropping systems. Specifically, I investigated the effects of these practices on insect pests and slugs, their predators, and biological control potential associated with these predators in an organic agronomic cropping system.
Organic growers face tradeoffs when managing their production systems. Reducing tillage has positive impacts on soil health and structure; however, without herbicides, organic growers rely largely on tillage for managing weeds and incorporating fertility inputs (Cavigelli et al. 2013). In addition to considering the tradeoffs between tillage for weed management and soil quality, producers need to consider the effects of their management practices on invertebrate pest management. The high-residue environment created by inclusion of cover crops and approach to tillage may affect the severity of damage from some early season pests, such as seedcorn maggot (Delia platura) (Hammond 1991, Mischler et al. 2010). A typical strategy for managing seedcorn maggot by organic growers is to delay planting until after the “fly-free” period for these flies. However, late planting paired with the restriction on use of transgenic Bt corn in organic production may render organic corn more susceptible to attack by European corn borer (Ostrinia nubilalis). This pest tends to prefer late-planted corn for larval feeding and utilizes ground vegetation as adults (Bohnenblust and Tooker 2010). Thus, understanding how management practices affect pests and the potential for predators to control these pests is needed for growers to maximize productivity and profitability.
Due to the restrictions on synthetic pest management materials in organic production, management of invertebrate pests relies heavily on preventative cultural methods such as strategically adjusting planting date, tillage, crop rotation, and on natural processes such as biological control (USDA NASS 2015). Organic yields tend to be lower than conventional due to lack of chemical treatments (Cavigelli et al. 2013). Biological control of invertebrate pests provided by predators has been valued at $4.5 million per year (Losey and Vaughan 2006). To maximize the benefits of biological control for organic producers, we must understand factors limiting or promoting the establishment of predators, the likelihood that predators in fields feed on prey within those fields, and the relationship between predator abundance and diversity on levels of damage incurred from pests. It is also beneficial to know which predators are the greatest contributors to biological control. Most predatory insects are actually omnivores, and require plant-based resources such as pollen or nectar in their diet. Consequently, although predators may be present in the field, they may not necessarily be feeding on pests, especially if other food resources, such as pollen and nectar, are more abundant (Grieshop et al. 2012). My project will provide valuable information to help formulate recommendations for organic producers on how to maximize predators and predation within their fields to reduce pest damage. Reduction of pest damage through predation will facilitate more reliable organic feed and forage production.
My research investigated practices geared towards good land stewardship and will support NE SARE’s goals. Specifically, we will target two of SARE’s key themes for sustainable agriculture. First, this project will promote the reduction of environmental and health risks in agriculture and improve productivity by providing alternative strategies to pest management without the use of pesticides or other synthetic inputs. Second, the facilitation of biological control of pests and organic production will increase net farm income by suppressing damage and yield loss from insect pests. The Northeast is home to four of the states with highest number of organic farms including New York (#3), Pennsylvania (#5), Vermont (#7), and Maine (#10) (USDA NASS 2015). Pennsylvania (#3) and New York (#7) also rank highly for organic sales (USDA NASS 2015). Thus, improvements for organic productivity will directly benefit farmers in the Northeast. Through the development of extension resources, organic producers and extension educators will have free access to information produced by this research.
Citations
Bohnenblust, E. and J. Tooker. 2010. European corn borer in field corn. Penn State University Cooperative Extension.
Cavigelli, M.A., S.B. Mirsky, J.R. Teasdale, J.T. Spargo, and J. Doran. 2013. Organic grain cropping systems to enhance ecosystem services. Renewable Agriculture and Food Systems: 28(2); 145–159. doi:10.1017/S1742170512000439
Hammond, R. B. 1990. Influence of cover crops and tillage on seedcorn maggot (Diptera: Anthomyiidae) populations in soybeans. Environmental Entomology 19:510-514.
Hagler, J. R., F. Blackmer, and D. W. Spurgeon. 2015. Accuracy of a prey-specific DNA assay and a generic prey-immunomarking assay for detecting predation. Methods in Ecology and Evolution 6: 1426–1434.
Holland, J.M. and C.J.M. Reynolds. 2003. The impact of soil cultivation on arthropod (Coleoptera and Araneae) emergence on arable land. Pedobiologia 47: 181–191.
Grieshop, M. J., B. Werling, K. Buehrer, J. Perrone, R. Isaacs, and D. Landis. 2012. Big brother is watching: Studying insect predation in the age of digital surveillance. American Entomologist 58: 172-182.
Landis, D.A., S.D. Wratten, and G.M. Gurr. 2000. Habitat management to conserve natural enemies or arthropod pests in agriculture. Annu. Rev. Entomo.45: 175-201.
Losey, J.E. and M. Vaughan. The economic value of ecosystem services provided by insects. 2006. BioScience 56: 311-323.
Mischler, R.A., S.W. Duiker, W.S. Curran, and D. Wilson. 2010. Hairy vetch management for no-till organic corn production. Agron. J. 102: 355–362.
Prasifka, J.R., N.P. Schmidt, K.A. Kohler, M.E. O’Neal, R.L. Hellmich, and J.W. Singer. 2006. Effects of Living Mulches on Predator Abundance and Sentinel Prey in
a Corn–Soybean–Forage Rotation. Environ. Entomol. 35: 1423-1431.
Rivers, A., M Barbercheck, B. Goeverts, and N. Verhulst. 2016. Conservation agriculture affects arthropod community composition in a rainfed maize–wheat system in central Mexico Applied Soil Ecology 100: 81–90.
USDA NASS. 2015. Organic Farming. Results from the 2014 Organic Survey. Accessed from: http://www.agcensus.usda.gov/ Publications/ 2012/ Online_Resources/ Highlights/Organics/2014_Organic_Survey_Highlights.pdf.
White, C., M. Barbercheck, T. DuPont, D. Finney, A. Hamilton, D. Hartman, M. Hautau, J. Hinds, M. Hunter, J. Kaye, J. La Chance. 2015. Making the Most of Mixtures: Considerations for Winter Cover Crops in Temperate Climates. Agroecology in Practice EEO166. Penn State Extension.
Cooperators
Research
Experimental Design:
This experiment has taken place within the context of the ongoing Reduced-Tillage Organic Systems Experiment (ROSE) at the Russel E. Larson Agricultural Research Center near Rock Springs, PA. More information about this ongoing project can be found here. The ROSE has been investigating the effects of cover crop usage, reducing tillage, and manure management on agronomic production, weed management, soil nitrogen dynamics, and invertebrate pests in an annual organic feed grain system. We expanded upon this project by adding a new focus on beneficial invertebrates and biological control potential.
The crop rotation in the ROSE consisted of corn (Zea mays), soybean (Glycine max), and spelt (Triticum spelta) in a 3-year rotation (Fig. 1). We measured pest and beneficial insects, spiders, and slugs during the corn phase of the rotation. Because this was a full-entry experiment, all crops in the rotation were present in each year of the experiment. Each cropping system was replicated four times with plots measuring 9 meters (30 ft)) by 49 meters (160 ft).
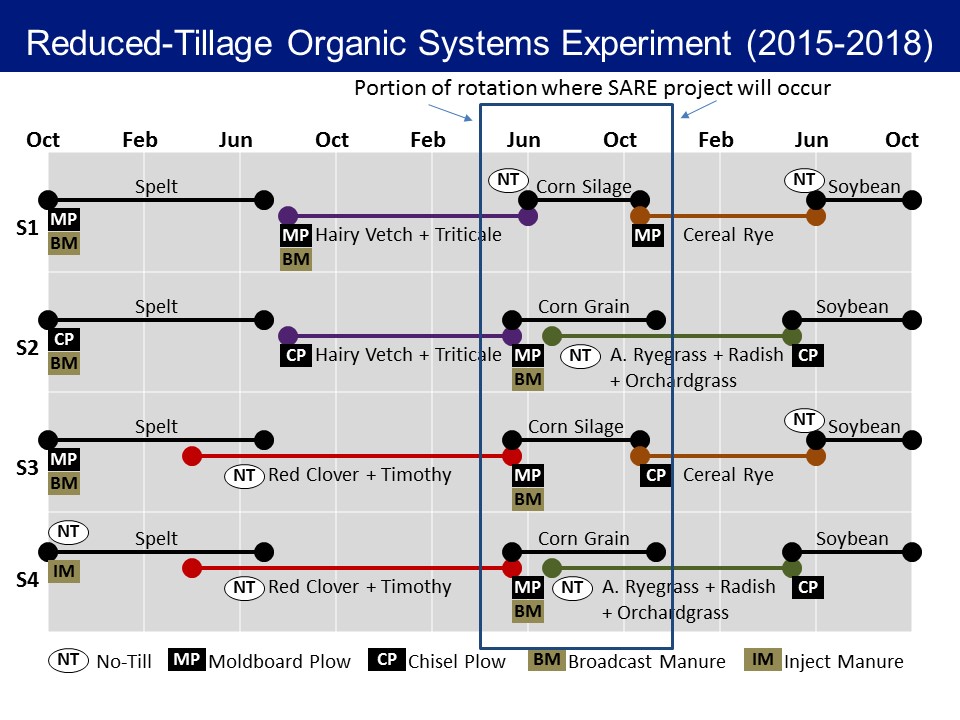
Fig. 1. This SARE project focusing on the corn phase of a three-year field and forage crops rotation with four cropping systems being compared.
The four cropping systems include:
1) hairy vetch (Vicia villosa) and triticale (Triticale hexaploide) cover crop mixture – terminated by roller crimper – no-till planted corn – corn harvested for silage
2) hairy vetch and triticale cover crop mixture – terminated by moldboard plowing – corn – interseeded with a forage radish (Raphanus sativus), orchard grass (Dactylis glomerata), and annual ryegrass (Lolium multiflorum) cover crop mixture – corn harvested for grain,
3) red clover (Trifolium pratense) and timothy (Phleum pratense) cover crop mix – terminated by moldboard plowing – corn – corn harvested for silage
4) red clover (Trifolium pratense) and timothy (Phleum pratense) cover crop mix – terminated by moldboard plowing – corn – interseeded with a forage radish (Raphanus sativus), orchard grass (Dactylis glomerata), and annual ryegrass (Lolium multiflorum) cover crop mixture – corn harvested for grain
Cover crops were typically terminated in mid to late May and planting occurred in early June. In System 1, where corn is planted no-till, preceding cover crops were terminated by rolling with a roller-crimper to form a mulch on the soil surface. In systems 2, 3, and 4, we used moldboard plowing to terminate the cover crops and prepare the seedbed for planting. Short planting windows between cash crop harvest in the fall and reduction in temperatures can make establishing cover crops challenging in the fall. The cover crop treatments in ROSE were chosen to overcome this short planting window and improve cover crop benefits. In Systems 2 and 4, we interseeded cover crops at the V5 stage of corn in approximately mid-July, allowing corn to be harvested later for grain instead of silage without compromising our ability to establish a cover crop for that winter. Additionally, in Systems 3 and 4 where corn is preceded by red clover and timothy, the red clover and timothy mix were seeded into the spelt in early spring, allowing it to establish prior to spelt harvest and giving it a longer growing season than the hairy vetch and triticale mixture that is established after spelt harvest in the other two systems. We harvested corn for silage in late September or early October for Systems 1 and 3 and for grain in late October for Systems 2 and 4. Comparison of these four cropping systems allowed us to assess effects of the species and management of preceding cover crops, timing of tillage, and mid-season interseeding of cover crops on pest establishment and damage, predator establishment, and predation by these beneficial predators.
Objective 1 (Completed):
We used multiple sampling methods to characterize pest and beneficial soil-dwelling arthropod communities. We characterized the ground-dwelling arthropod community using pitfall traps. We placed two traps consisting of a plastic deli container buried level with the soil surface in each treatment plot and filled with ethylene glycol as a killing agent. Traps were left open for 72 hours. Arthropods were removed from traps and identified. Pitfall sampling occurred three times per season: two weeks prior to cover crop termination, two weeks after corn emergence, and four weeks after interseeded cover crop emergence. These three time points allowed us to measure effects of the preceding cover crop on arthropods, whether these populations are retained after cover crops are terminated and corn is planted, and whether the addition of interseeded cover crops mid-season influences establishment of predators later in the season.
To assess effect of management system on seedcorn maggot populations, which is a pest of concern to field crop producers in Pennsylvania, emergence traps were placed at the soil surface shortly after corn emergence to capture emerging adult flies (Hammond 1990). Traps consist of a plastic window box (1.0 m by 0.3 m) with a hole cut in the base that is fitted with a quart size mason jar. The traps are placed base side-up so that as flies emerge from the soil, they will move towards sunlight visible through the jar and be trapped by a mesh funnel in the opening in the jar (Hammond 1990). Jars were removed and emptied weekly from corn emergence until true leaf formation (V2). All arthropods caught were returned to the lab and identified.
Objective 2 (Completed):
Early-season pest damage was assessed through visual observations. Areas with missing seeds underneath seedcorn maggot emergence traps were dug up to look for maggot feeding damage. Damage from other early-season pests will be measured on a 5.3 meter (17.5 ft) transect in each plot. Plants were examined for damage from a variety of plant-feeding pests common early in the season including: slugs (Gastropoda), cutworms (Lepidoptera: Noctuidae), billbugs (Sphenophorus aequalis), and other chewing insects. In addition to categorizing damage, the amount of damage per plant was rated on a 0-4 scale with 0 indicating no damage, 1 indicating 1-25% damage, 2 indicating 26-50% damage, 3 indicating 51-75% damage, and 4 indicating greater than 76% of a plant damaged. Plant populations were also recorded along this transect.
There are multiple species of moth that are common to Pennsylvania corn fields and can potentially cause yield-limiting damage during their larval phase. European corn borer (Ostrinia nubilalis) has two flights in central Pennsylvania (Bohnenblust and Tooker 2010), while fall armyworm (Spodoptera frugiperda) and corn earworm (Helicoverpa zea) typically overwinter south of Pennsylvania and infest fields as the growing season progresses. Damage from European corn borer was assessed in early September after the conclusion of the second generation’s flight to provide a cumulative measure of damage over the entire season. Corn earworm damage was assessed simultaneously. Twelve plants per plot were collected and examined for boring tunnels on corn stalks or feeding damage on the corn ear from European corn borer or corn earworm, another common pest of corn in Pennsylvania. Any larvae found in tunnels or on the ear were collected and identified. Additionally, five randomly selected transects of 20 plants each were visually inspected for damage from fall armyworm in mid-August. Any larvae found during either assessment were collected and identified.
Objective 3 (Ongoing):
Biological control potential was measured in three ways. To measure predation rate, five waxworm larvae (Galleria mellonella) were attached to index cards to serve as sentinel prey. Baited index cards were surrounded by a trap constructed from 19-gauge mesh hardware cloth and covered with a clear plastic lid to allow entry by invertebrate predators but exclude vertebrates, such as mice, and rain. One day prior to each pitfall event, four baited sentinel cards were placed in each plot for 24 hours. After 24 hours, the cards were collected and the proportion of sentinel prey fed upon was determined.
To record the relative contribution of different predator taxa to predation, we placed an infrared camera above each sentinel bait card to record feeding events for the 24 hour-period that cards are in the field each time that sentinel predation assays occur. We have been reviewing video footage from 2017 to identify predators and record the amount of feeding by each predator, allowing us to determine frequency of predation by specific predators and which are likely to make the greatest potential contribution to suppression of pest populations.
To reinforce our understanding of predator activity within the field and connect it to the overall invertebrate community, we marked sentinel caterpillars with a protein mark and test predators for this mark. This was done using diluted egg white sprayed onto waxworms attached to two of the four sentinel bait cards used in each plot. Predators were collected from two additional pitfall traps buried adjacent to the marked cages that had no killing fluid but instead were lined with an adhesive card. Predators were identified, removed from adhesive cards, and analyzed for the protein that we used to mark the caterpillars using enzyme-linked immunosorbent assay (ELISA) techniques. This assay uses antibodies to detect specific proteins, to determine whether predators caught in pitfall traps have fed upon our sentinel prey. This technique was carried out in collaboration with Dr. James Hagler at the USDA Arid Land Agricultural Research Lab in Maricopa, AZ, who is the developer of this method. Predators were brought to Arizona and assayed in Dr. Hagler's lab in 2017 and 2018.
Through this research, we hoped to learn which predator groups make the greatest contribution to predation in each system, depending on time of year and habitat created by those systems. By evaluating the predation occurring within different cropping systems, we aimed to better determine how tillage and cover cropping practices enhance or detract from insect predation. Additionally, by identifying main contributors to predation, we could gear conservation efforts through management practices towards these key predators.
Objective 4 (Ongoing):
To most effectively benefit farmers, the information from this research project must be shared in an accessible fashion and with grower priorities in mind. Through the larger ROSE project, which includes research at the research station and on three organic farms, we have ongoing opportunities to participate in outreach and extension with organic producers and other stakeholders. We have hosted or co-hosted field days at the Penn State research station and on our collaborating growers’ farms, which allowed us four opportunities in each year to interact with producers in a tour-based setting. Events hosted by our project team will include post-event evaluation surveys to obtain directive feedback from participants about knowledge attainment from the event, their intention to implement new practices, and their information content needs, including preferred methods and context for receiving this information. We have also welcomed visitors at the project sites throughout the project's history, allowing us to extend knowledge to farmers that may not be directly related to the project. An ongoing component of this objective will be to author an extension fact sheet on natural enemies in organic field crops, based on the results of Objective 3 as it is completed.
Objective 1:
We measured high activity-density of predators in this project and present here both total numbers of predators, as well as specific common generalist predators. Prior to termination of cover crops in the spring, we measured significantly greater ground beetle (Coleoptera: Carabidae) activity-density in pitfall traps in the two systems planted with hairy vetch and triticale than in the two systems planted with red clover and timothy (Fig. 2). After cover crops were terminated and corn was planted, we observed significantly greater activity-density of spiders in the system planted no-till than in the three systems where tillage was used (Fig. 3). Later in the season, after the interseeded mixture was planted and emerged in Systems 2 and 4, there is no effect of cropping system on any of the common predator taxa (Fig. 4). We do not see any difference in the total number of predators in each cropping system at any of the three sampling dates (Fig. 5). Interestingly, the highest activity-density of total predators was measured at the middle sampling date, shortly after corn emerged. Thus, it appears that the crop management tactics employed in our study do not have substantial impact on natural enemy activity.
We caught significantly more seedcorn maggot in cropping systems where cover crop residue was tilled in than in the system where cover crops were rolled and corn was planted no-till (Fig. 6). Seedcorn maggot is known to be attracted to freshly tilled decomposing plant residue (Hammond 1990, Hammond 1997), so this result is not surprising. Although benefits for predators were less than we had expected, we do not see increased pest pressure of a key pest when no-till practices are adopted in organic systems.
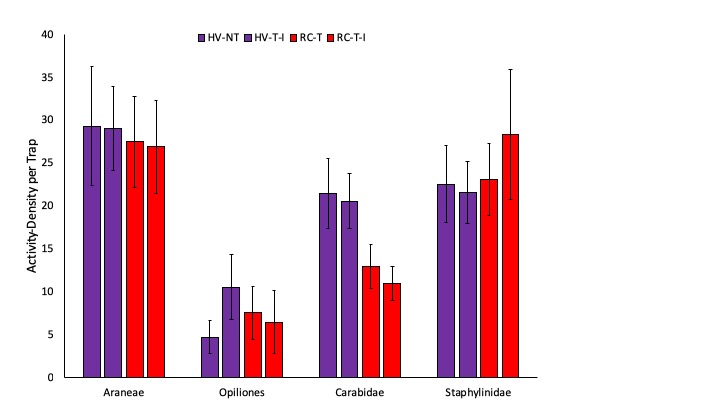
Fig. 2. The most abundant generalist predators including spiders (Araneae), harvestmen (Opiliones), ground beetles (Coleoptera: Carabidae), and rove beetles (Coleoptera: Staphylinidae) are shown prior to cover crop termination. Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). Letters represent means that are significantly different at the 0.05 level of significance.
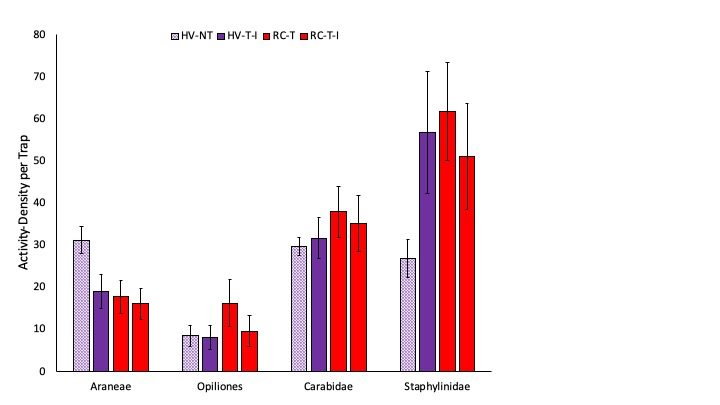
Fig. 3. The most abundant generalist predators including spiders (Araneae), harvestmen (Opiliones), ground beetles (Coleoptera: Carabidae), and rove beetles (Coleoptera: Staphylinidae) are shown after corn emergence. Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while the three tilled systems are shown with solid bars. Letters represent means that are significantly different at the 0.05 level of significance.
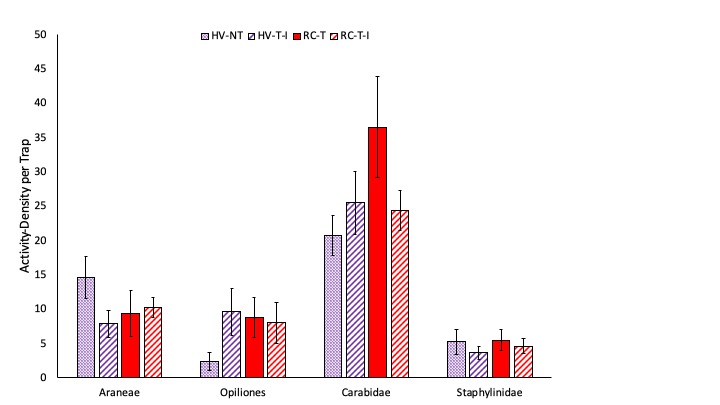
Fig. 4. The most abundant generalist predators including spiders (Araneae), harvestmen (Opiliones), ground beetles (Coleoptera: Carabidae), and rove beetles (Coleoptera: Staphylinidae) are shown after interseeded cover crop emergence. Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while systems where cover crops were interseeded are shown with diagonal lined bars (I). Letters represent means that are significantly different at the 0.05 level of significance.
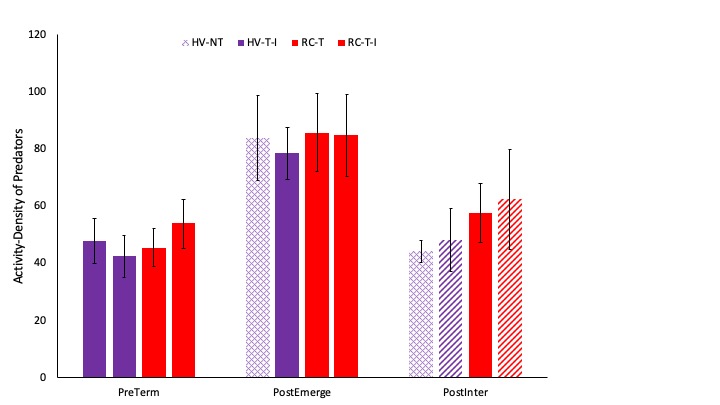
Fig. 5. The total activity-density of predators is shown prior to cover crop termination (PreTerm), after corn emergence (PostEmerge), and after interseeded cover crop emergence (PostInter). Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while systems where cover crops were interseeded are shown with diagonal lined bars (I).
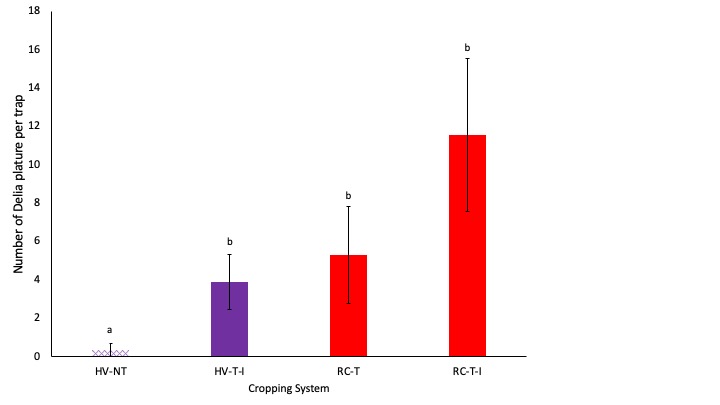
Fig. 6. Abundance of seedcorn maggot per trap is shown. Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while the three tilled systems are shown with solid bars. Letters represent means that are significantly different at the 0.05 level of significance.
Hammond, R. B. 1990. Influence of cover crops and tillage on seedcorn maggot (Diptera: Anthomyiidae) populations in soybeans. Environ. Entomol. 19:510-514.
Hammond, R. B. 1997. Long-term conservation tillage studies: impact of no-till on seedcorn maggot (Diptera: Anthomyiidae). Crop Prot. 16: 221-225.
Objective 2:
Early season damage averaged 1 on my 0-4 scale of severity throughout all four cropping systems, indicating that less than 25% of each plant was damaged. The most common category of damage was slug damage, with more than 80% of plants affected on average (Fig. 7). This is unsurprising, as it was a fairly cool and wet in Pennsylvania throughout the years of this study, which is favorable to slugs. There were no significant differences in damage severity or in prevalence of slug damage between the four cropping systems assessed. There was greater damage from chewing pests in System 1 (45% of plants), which is preceded by hairy vetch and triticale and then planted no-till relative to the three systems which are tilled before planting (about 10% of plants affected in each). Late season caterpillar pressure was fairly low. European corn borer damage (Ostrinia nubilalis) was nearly absent in 2017 with only 1 tunnel found in all of the plants assessed. With the low and highly variable populations, no significant effects of cropping system were observed on damage from these three pests (Fig. 8). Based on these results, there does not appear to be increased pest pressure due to the adoption of cover crop mixtures, no-till planting, or interseeding.
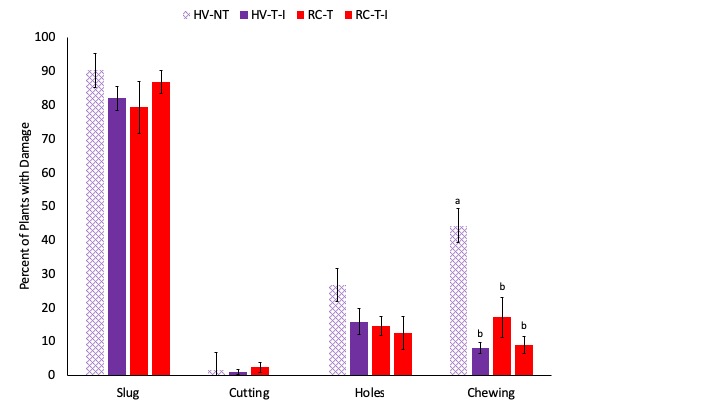
Fig. 7. The percentage of plants in each plot with damage from slugs, cutworms (Cutting) whorl feeding (Holes) and chewing pests are shown. Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while the three tilled systems are shown with solid bars. Letters represent means that are significantly different at the 0.05 level of significance.
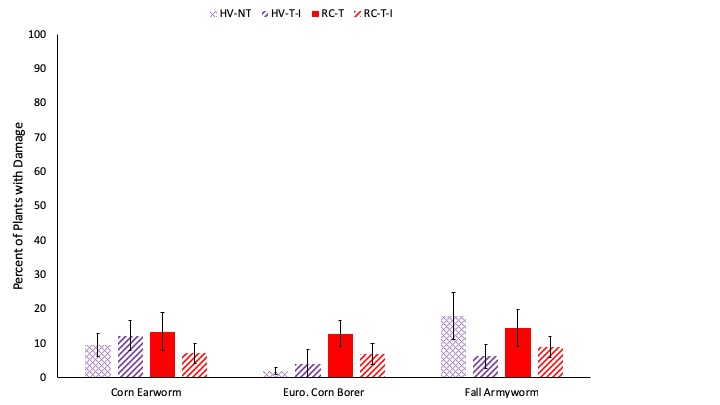
Fig. 8. The percent of plants with feeding damage from corn earworm, European corn borer, and fall armyworm is shown. Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while systems where cover crops were interseeded are shown with diagonal lined bars (I).
Objective 3:
Predation, as measured through sentinel bait cards, increased through the season with the lowest rates occurring before cover crops were terminated and highest rates on the final sampling date after interseeded cover crops emerged (Fig. 9). There were no differences among cropping system at any of the three dates, through the third date was significantly higher than the previous two.
Reviewing footage from infrared cameras of predation on sentinel cards was an incredibly labor-intensive process with multiple technical setbacks. Cameras were successfully employed in the field during summer 2017 (Fig. 10). The resolution of footage was not as high under field conditions and lighting as it was in preliminary trouble-shooting indoors. An additional challenge in achieving high resolution was the need to keep the cages fully enclosed in the field with cameras pointing into the cage, rather than leaving the lid off and giving the camera a clear view of the sentinel cards. We experienced loss of some cards to rodents and birds visiting the cages and found leaving them shut was the best way to ensure enough cards remained in the field to collect data. We also encountered difficulty keeping vegetation from moving in front of the camera's view due to wind or humidity. It appears that the community visiting these cards is dominated by ground beetles (Coloeptera: Carabidae), particularly late in the season at the sampling date after interseeded cover crops have emerged. We do see visits to cards by wolf spiders (Arachnida: Lycosidae) and harvestmen (Opiliones), but not as frequently as these taxa are caught in traps. Although visits to cards by ants (Hymenoptera: Formicidae) were common, they were not always predation events that led to prey mortality, but were instead feeding visits to waxworms that had already been killed by previous visitors. During the sampling date after interseeding, we also observed feeding visits by crickets (Orthopera: Gryllidae) despite catching very few crickets in pitfalls. Due to the labor-intensive nature of this data collection, processing of video footage is still in progress and final conclusions on key contributors to predation can't be made. However, the footage reviewed thus far indicates that there is a difference between the community composition determined through pitfall trapping and the community that visits sentinel cards and contributes to prey mortality. The scope of this objective that we attempted by placing 64 cameras in the field limited its usage as a practical and affordable method for studying predator behavior for our goals. We had hoped to gain more insight in variation across our experimental site and achieve sufficient replication to understand predator contribution, but have found this number of cameras to be impractical. We would recommend this tactic on a much smaller scale for asking questions about specific taxa and their role in predation, such as beetles or ants, rather than the entire ground-dwelling predator community.
Additionally, gut contents of predators that fed on protein-marked sentinel prey were assessed using ELISA. Of 540 arthropods assessed, only 3 yielded positive marks. All three of these were ground beetles (Coloeptera: Carabidae). Unfortunately, the low number of positive marks limits conclusions that can be drawn. Re-evaluation of methodology may be necessary to enable successful use of these techniques to study predation in the field.
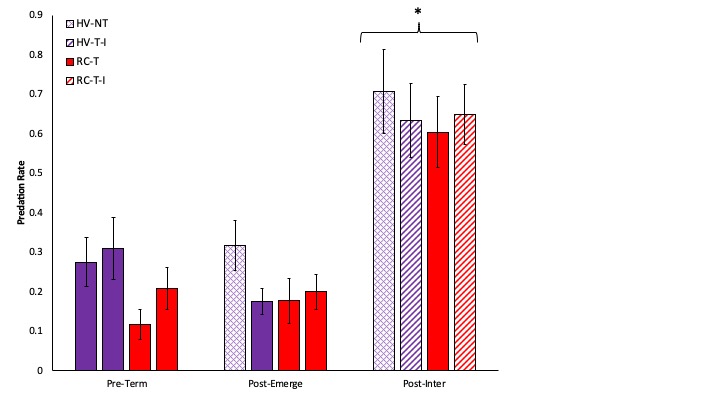
Fig. 9. The percentage of waxworms with feeding damage prior to cover crop termination (PreTerm), after corn emergence (PostEmerge), and after interseeded cover crop emergence (PostInter). Systems shown in purple were planted with hairy vetch and triticale (HV) while systems shown in red were planted with red clover and timothy (RC). The no-till system is shown with cross hatched bars (NT) while systems where cover crops were interseeded are shown with diagonal lined bars (I).


Fig. 10. Shown here are photos of cameras set up to record feeding events at sentinel bait cards in plots. The environment around each camera ranged from dense living cover crop vegetation or mostly bare soil, depending on sampling date and cropping system.
Objective 4:
I have summarized information learned from this project on the benefits and trade-offs in cover crop management and approach to tillage reduction on predator conservation and biological control. This information has been disseminated to both technical and stakeholder audiences. I have presented my research at the national Entomological Society of America meetings in Denver, CO in November 2017 and in Vancouver, BC in November 2018, as well as the Eastern Branch meeting of ESA in Newport, RI in March 2017 and in Annapolis, MD in March 2018. I have given three invited seminar presentations featuring this research, as well as five poster presentations. I have also shared this information with farmers, extension educators, and industry representatives associated at seven extension events in the past two years. I was also able to share information on arthropods in organic agriculture through a poster session for organic agriculture research at the Pennsylvania Association of Sustainable Agriculture’s annual conference in February 2018, extending our reach to a wider audience and allowing us to benefit more producers interested in sustainable agriculture. For all presentations, NESARE was acknowledged for providing funding to support the research.
Through this project investigating pest pressure, predator populations, and biological control potential in cover crop-based reduced-tillage cropping systems, we were able to identify potential concerns that may require additional scouting but found relatively little effect on natural enemies. For early season pests, there may be few benefits to using tillage as a pest control tactic. We observed more prevalent damage from chewing pests in systems planted no-till than in tilled systems, but there was not a corresponding increase in damage severity. Seedcorn maggot, which is a particularly concerning pest for Pennsylvania growers, was less abundant in no-till corn than in tilled corn. Slugs, another key pest in Pennsylvania, were unaffected by preceding cover crop mixture or by tillage. Later in the season, there was no impact of cropping system on three common moth pests (corn earworm, European corn borer, and fall armyworm). Thus, it appears that growers can benefit from incorporating interseeding as a tactic to improve cover crop establishment without any increase in pest pressure.
The abundance of common natural enemy taxa differed by cropping system before cover crops were terminated and after corn had emerged, but these differences did not persist throughout the season. Predator populations and biological control were not enhanced by cover crop mixtures, no-till, or by interseeding. These tactics may still hold other benefits for producers in weed management or soil health, but do not appear to provide the additional benefit of increasing biological control.
Education & outreach activities and participation summary
Participation summary:
I assisted with two separate workshops related to identification and conservation of predatory arthropods in farm fields. The first occurred at the Penn State Student Farm to educate local residents and gardeners on improving the health of their soils and conserving beneficial arthropods, including soil-dwelling arthropods. The second was a workshop focused on soil health presented to over 100 farmers, industry representatives, and extension educators attending the annual Penn State Agronomy Diagnostic Clinic. My portion of the workshop focused on predators living in or on the soil and how soil health practices affect them. Both workshops relied on preliminary lessons learned through my SARE research on soil-dwelling pests and predators.
I presented a description of research I was conducting at the Penn State Research Farm, including my SARE research, to a medium-sized audience (40) of extension educators working in field and forage crops at Penn State, to a group of visiting farmers and agricultural educators from South America (15 people), and a group of farmers from Kazakstan (10 people). Most information was presented orally but I used the following poster as a backdrop for in-field presentations: ExtensionPoster_27June2017
I gave a tour of our research site and a description of my research to a visiting farmer and his grandson from Massachusetts.
I presented research findings at five poster symposia and four oral symposia at research conferences, as well as giving three invited seminar presentations. The posters I used can be viewed here: Regan_poster_SCSS17 and here: Regan_PASA2018poster_V3
I gave a presentation to the local community on the importance of insect predators in agriculture and the ways in which crop management impact pests and predators through an event organized by the Penn State Science Policy Society.
I helped to complete an online fact-sheet on seedcorn maggot biology and management that addresses risk factors associated with cover crops and tillage, which can be accessed here: Seedcorn Maggot FactSheet_final
Invited Presentations
February 2019. “Arthropod Communities within a Reduced-Tillage Organic Systems Experiment” K. Regan. Entomology Department, New Jersey Agricultural Research Station, Rutgers University, Bridgeton, NJ.
January 2019. “Arthropod Communities within a Reduced-Tillage Organic Systems Experiment” K. Regan. Entomology Department, Cornell AgriTech, Geneva, NY.
April 2018. “Arthropod Response to Cover Crop-Based Organic Cropping Systems” K. Regan. Plant-Insect Group Seminar Series, Entomology Department, Cornell University, Ithaca, NY.
Research Presentations
November 2018. “Effects of Reduced-Tillage Organic Cropping Systems on Athropod Predators” K. Regan, C. Voortman, and M. Barbercheck. Student Paper Competition, Entomological Society of America, Vancouver, BC.
May 2018. “Insect Response to Organic Cover Crop-Based Reduced Tillage Cropping Systems.” K. Regan, C. Mullen, and M. Barbercheck. Poster Presentation, Penn State University Life Science Symposium, State College, PA
March 2018. “Effects of Organic Reduced-Tillage Cropping Systems on Epigeal Predators.” K. Regan, C. Mullen, and M. Barbercheck. Student Paper Competition, Eastern Branch of the Entomological Society of America, Annapolis, MD.
February 2018. “Insect Response to Organic Cover Crop-Based Reduced Tillage Cropping Systems.” K. Regan, C. Mullen, and M. Barbercheck. Poster Presentation. Pennsylvania Association for Sustainable Agriculture Annual Conference, State College, PA.
November 2017. “Arthropod Response to Cover Crop-Based Reduced Tillage Organic Cropping Systems. K. Regan, C. Mullen, and M. Barbercheck. Student Paper Competition, Entomological Society of America, Denver, CO.
June 2017. “Arthropod Response to Cover Crop-Based Reduced Tillage Organic Cropping Systems.” K. Regan, C. Mullen, and M. Barbercheck. Poster Presentation, Penn State University Life Science Symposium, State College, PA
March 2017. “Arthropod Response to Cover Crop-Based Reduced Tillage Organic Cropping Systems.” K. Regan, C. Mullen, and M. Barbercheck. Poster Presentation, Sustainable Cropping Systems Symposium, University Park, PA.
March 2017. “Arthropod Response to Cover Crop-Based Reduced Tillage Organic Cropping Systems.” K. Regan, C. Mullen, and M. Barbercheck. Poster Presentation, Behavioral Ecology and Experimental Methods Workshop, University Park, PA.
March 2017. “Arthropod Response to Cover Crop-Based Reduced Tillage Organic Cropping Systems.” K. Regan, C. Mullen, and M. Barbercheck. Student Paper Competition, Eastern Branch of the Entomological Society of America, Newport, RI.
Extension Presentations
October 2018. “Who’s in a corn field?” Invited speaker for Science Brew, geared toward the general public and hosted monthly by the Penn State Science Policy Society, Bellefonte, PA
September 2018. “Insect Management in a Reduced-Tillage Organic Systems Experiment.” ROSE Field Tour for visiting Khazak farmers and agricultural consultants, Russell E. Larson Agricultural Research Center, Rock Springs, PA
May 2018. “Who’s in a corn field?” Rock Springs Annual Orientation to the Farm. Rock Springs, PA
August 2017. Soil Health and IPM Workshop, Penn State Student Farm, State College, PA
June 2017. “Pest Management Strategies in Organic Row Crops: how do tillage and cover crops play a role?” PSU Field and Forage Extension Team In-Service Day, Rock Springs, PA
June 2017. “Arthropod Response to Reduced-Tillage Organic Systems.” ROSE Field Tour for visiting Argentinian farmers and agricultural consultants, Russell E. Larson Agricultural Research Center, Rock Springs, PA
July 2017. Soil Biology and Health Workshop, Annual Agronomy Diagnostic Clinic, Rock Springs, PA
May 2017. “Arthropod Sampling Methods in Organic Row Crops” Rock Springs Annual Orientation to the Farm. Rock Springs, PA
Project Outcomes
This project has indicated that decisions around cover crop species mixtures, adoption of no-till practices, and interseeding cover crops to improve their establishment can all work without concerns of increased pest pressure. Many of the growers I interacted with were interested in the efforts we were making to reduce tillage and improve cover crop establishment, but face greater problems from weed and nutrient management than from insect management. The low pest pressure and high predator activity-density we observed appears to align with the experience of growers throughout Pennsylvania, as well. By troubleshooting these management practices and learning that pest pressure does not increase, we have been able to provide pest management insight as farmers make decisions about adopting new practices to improve soil health in organic systems.
I was interviewed by Entomology Today, which is the blog run by Entomological Society of America in regards to my experience conducting extension in Pennsylvania and grower interest in reducing tillage and using cover crops.
Remaining output that is ongoing includes publishing five research manuscripts outlining my research and preparing an additional fact sheet on natural enemies in organic field crops.
Media Coverage: Kraft, L. J. “Extension Unplugged: Communicating entomology to Amish and Mennonite communities in Pennsylvania.” Entomology Today 18 Dec 2018 Accessed from: https://entomologytoday.org/2018/12/18/extension-unplugged-communicating-entomology-amish-mennonite-plain-communities-pennsylvania/
My advisor and I's knowledge of sustainable agriculture was enhanced by this project because we learned that effects of management on pests and predators may not occur at as fine of a scale as we had hypothesized when developing this project. We had expected cover crops species mixtures, tillage, and interseeding of cover crops to have measurable impacts on early and late season pest pressure, predator activity-density, and biological control. In most cases, we did not observe impacts of crop management on pests and predators. Thus, within organic systems that are reducing tillage and using cover crops, pest pressure appears to be low in most cases while predator activity-density is fairly high across the board.
My future career will continue to involve pest management and extension. I look forward to continuing to learn about and work towards increasing adoption of practices that will support sustainable agriculture. Since this grant was funded, I have applied for and received two additional grants to continue the video surveillance work outlined in Objective 3 of this project, identify key predators in organic systems, and enhance my understanding of predator behavior. I have developed a collaboration with a colleague at Penn State to further address the effects of tillage on pests and predators in agriculture. The video surveillance and ELISA components of this project, as well as my work in reduced-tillage agriculture have helped to start discussions and establish networking relationships with other entomologists, ultimately leading to a position as a post-doctoral research associate, beginning in June 2019 where I will continue to research pest management and biological control in agriculture.