Final report for ONE18-318
Project Information
Urine-derived fertilizer (UDF) is a safe, locally-sourced fertilizer that has been shown to be an effective replacement for synthetic nitrogen fertilizers. However, like other ammonium-containing fertilizers such as manure and urea, UDFs have the potential to lose significant amounts of nitrogen to ammonia volatilization. In this project, we sought to quantify potential ammonia losses from UDF under different field application scenarios, identify ways to minimize those losses, and better understand how UDF could be integrated into farmers’ existing fertilizer management strategies. These goals were carried out with two distinct project components: 1) field trials measuring ammonia volatilization, and 2) site visits interviewing farmers about their fertilizer management practices.
The ammonia component consisted of four field trials measuring ammonia volatilization following application to hay fields with different application methods, wind speeds, irrigation/rainfall, and urine pre-treatment methods. Ammonia volatilization losses in all of these scenarios ranged from 5% to 34% of total applied N, and were generally lower than or similar to losses from liquid manure reported in other studies. Our trials identified two important factors for reducing ammonia volatilization losses. Application method had the largest impact on ammonia losses; as with manure, reduced air exposure significantly reduces ammonia volatilization. As a result, we recommend avoiding broadcasting or spraying UDF, and instead applying close to or directly into the ground, which may reduce losses by up to 80%. Our results also showed that irrigation or rainfall during application reduced ammonia losses by up to half.
Based on our results, we have identified two guidelines for minimizing ammonia loss from urine application to hayfields:
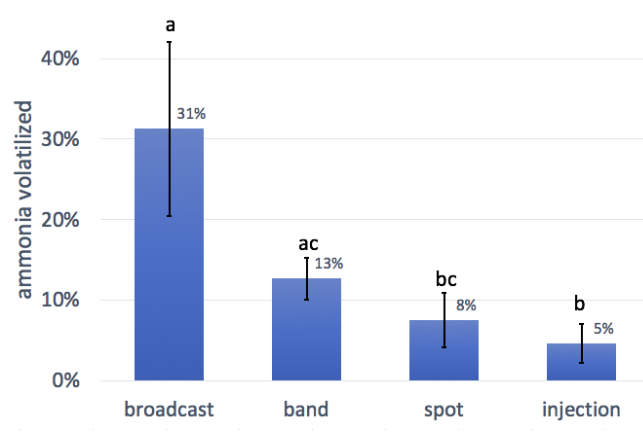
- Avoid broadcasting urine – instead, apply in a stream close to (or into) the ground. Our trials measured the following ammonia loss rates for different application methods: broadcast 31%; band 13%, spot 8%; injection 5%.
- Apply urine to moist soil or during rainfall or irrigation. In our trial, a small amount of irrigation reduced ammonia loss by one half.
Urine pre-treatments that keep nitrogen in urea form, rather than ammonia form, may reduce ammonia losses. In our trial, low-pH-treated urine lost less ammonia than urea pellets.
The goal of our site visits was to strategize with a diverse array of area farmers the practices, tools, techniques, and critical research to make the most effective use of UDF under varying management conditions. We conducted site visits and interviews at ten local farms that raise livestock and grow forage crops, as well as one growing hemp and cut flowers. From this dialogue we learned that small and large farmers, organic and conventional, and those growing a variety of forage crops, were generally open to and interested in the use of urine fertilizer, both for its environmental benefits and its nutrient value. However, they have a number of specific concerns such as the availability of the quantities they would need at the times they need it; availability of appropriate storage and application equipment; and time/labor requirements. None of the farmers currently owned equipment that would meet the recommendations identified in the field trials, but they suggested potential strategies for cooperative ownership or sharing of appropriate equipment. With farmer input, we identified a range of research needs going forward that would help support broader use of UDFs.
Following the completion of both projects, we organized a farm field day at which we reported results of both the ammonia volatilization study and the farmer site visits. Attendees included a number of area farmers and agricultural educators. Three farmers currently using sanitized urine supplied by Rich Earth Institute on hay shared their experiences and answered questions. Overall, farmers participating in this study and at the field day were supportive of moving implementation of agricultural use of UDF forward. The support for UDF among farmer participants, combined with manageable levels of ammonia loss in our field trials, indicate that UDF is a viable option for perennial grass if the additional research questions identified can be answered to farmers’ satisfaction.
Field Trials: Our objective was to determine relative rates of ammonia loss when urine-derived fertilizer (UDF) is applied to perennial grass using different fertilizer preparation and application methods, and under varying soil moisture and wind speed regimes. We wanted to establish a range of conditions where UDF can be applied with acceptable ammonia volatilization rates in order to enhance nitrogen retention in soils. By identifying surface application methods and conditions that minimize ammonia volatilization, our goal was to reduce nutrient pollution and increase the efficacy and economic benefit to grass farmers using UDF.
Site Visits: In dialogue with farmers from a diverse array of livestock farms, we planned to explore the best strategies and opportunities for using current or slightly modified equipment to use UDF effectively on their farms for hay, corn, and other feed production while minimizing ammonia loss through tillage or judicious surface application. The documentation and dialogue addressed fertility management practices (including the use of liquid manures and digestates); equipment used; timing and weather conditions of application; and different soil management practices. The goal was to strategize with farmers the practices, tools, techniques, and critical research to make the most effective use of UDF under varying management conditions.
Our project addresses two problems: 1) nutrient pollution of waterways, and 2) the unsustainability of synthetic fertilizer. Our approach to solving both problems is to divert human urine out of the wastewater stream, reclaiming dissolved NPK and trace plant nutrients for use as agricultural fertilizer.
Ponds, lakes, and coastal estuaries throughout the Northeast are heavily impacted by nitrogen and phosphorus pollution caused by human activity. Much is made of nutrient pollution from agricultural sources, but wastewater is also a major contributor, and is the principal source of nutrient pollution in some heavily populated watersheds in the Northeast. Human urine contributes 80% of the nitrogen and 65% of the phosphorus in wastewater (Larsen et al. 2013). Most of these nutrients pass through treatment facilities and into surface waters where they cause pollution.
Fertilizer is expensive for farmers, and prices are unpredictable, threatening farms’ financial viability. Nitrogen is tied to the volatile price of unsustainable fossil fuels. Phosphate is a finite resource, and the Global Phosphorus Research Initiative predicts a shortage of quality rock phosphate within 40 years. Diverting urine from the wastewater stream and recycling it into fertilizer would reduce nutrient pollution from waterways, and provide a renewable and reliable supply of domestically produced fertilizer
Previous SARE-funded research by the Rich Earth Institute, (which operates the nation’s first community-scale urine recycling program in Brattleboro, Vermont) has demonstrated in multi-year trials that sanitized urine can effectively replace synthetic fertilizer in hay production when applied under appropriate conditions. However, nearly all the nitrogen in urine rapidly converts into ammonia, which is prone to loss by evaporation under certain conditions. Although research exists on ammonia loss from animal manures, there are currently no research-based guidelines on acceptable methods and conditions for surface application of urine-derived fertilizer (UDF) on hay.
Our farmer partners, who have seen dramatic yield improvements as a result of UDF, are enthusiastic about continuing to use it. They have expressed strong interest in obtaining data on nitrogen retention in soil over time and best management practices for maximizing the benefits of UDF. Most grass farmers responding to an earlier survey (described in final report SARE ONE14-218) reacted positively to the idea of using UDF. Seventy-five percent said they wanted more information about fertilizer benefit and 58% wanted to know more about the equipment required to use this fertilizer. In this study we aimed to further contextualize farmer perspectives concerning effective use of UDFs in their specific situations.
In addition to high-ammonia, sanitized urine, the Institute has also developed several forms of stabilized urine products that can now be tested to determine optimal fertilizer formulation, application method, timing, weather and soil conditions that are best suited to minimize loss of nitrogen to the atmosphere. In this research project we:
1) conducted field trials to quantify ammonia loss from urine-derived fertilizers applied to perennial grass, under a range of environmental conditions and using different UDF products
2) conducted site visits with a diverse array of farmers to a) develop effective strategies for incorporating urine-based fertilizers into current farm operations, and b) conceptualize the tools, techniques and further research needed to support this potentially transformative practice.
Urine-derived fertilizer (UDF) has a long and established record as a safe and effective replacement for synthetic fertilizer (WHO, 2006) on a wide variety of grains, fruits, and vegetables. Numerous treatment methods are available to inactivate pathogens, including the pasteurization method permitted in Vermont and employed by the Rich Earth Institute. The question of pharmaceutical presence in urine is often raised; our research to date, (performed with urine from our program,) has shown that although pharmaceuticals are present in urine, a person would have to eat close to a million pounds of lettuce fertilized with urine to receive a single dose of any pharmaceuticals investigated (Mullen, et. al., 2015). Even so, to avoid any issues with public perception our efforts focus on fertilization of animal feed crops.
The Institute has conducted four years of field trials using urine-derived fertilizer on hay (SARE ONE13-188, ONE14-218, ONE15-244, and an earlier 2012 trial). Two trials on very moist sandy loam soils both showed strong positive yield response comparable to synthetic fertilizer controls, while trials on drier and sandier soils showed strong response one year and poorer response on the following year.
For urine-derived fertilizer to be effectively utilized in hay production, we believe that the most important factor to control is the loss of nitrogen through ammonia volatilization. This is an issue with other nitrogen-rich fertilizers; ammonia volatilization losses from surface-applied synthetic urea can exceed 60% (Horneck et al., 2011), while losses from animal manure can exceed 90% (Jokela and Meisinger, 2008). These losses can be virtually eliminated through immediate incorporation, such as by simple tillage in row crops, but subsurface incorporation on grasslands cannot be done without specialized equipment. To make urine-derived fertilizers more accessible to hay producers, guidelines are needed for effective surface application.
Manure characteristics affecting ammonia volatilization include solids content, NH4-N content, and pH, with increases in each characteristic leading to increased risk of volatilization (Jokela and Meisinger, 2008). Our own testing shows that nearly all the nitrogen in sanitized urine is in the ammonia/ammonium form, (all urea having hydrolyzed to ammonia,) and the pH is around 9, near ammonium’s pKa of 9.25. Due to these two factors, unincorporated sanitized urine is very prone to volatilization. Fortunately the solids content of urine is very low, so urine is more able to self-incorporate into soil by soaking in, compared to manure slurries which pool on the surface and remain exposed. Furthermore, our custom applicator dispenses urine in many low-velocity streams, mimicking the ammonia-conserving technique of applying liquid manure in discrete bands (Pfluke et al, 2011). How these opposing factors balance out when UDF is applied to perennial grasslands has been unknown, as have been the specific effects of environmental factors and fertilizer application methods.
Since 2015, the Rich Earth Institute has also conducted trials producing urine-enriched composts. Results indicate that co-composting urine with leaves has the benefit of binding about 65% of the nitrogen from urine into a solid form with an elemental N:P ratio above 10, and acceptable ammonia and salinity levels (SARE ONE15-244 final report). Limitations of this method include the 35% of nitrogen that is lost during composting, and the requirements for carbon feedstock, compost-making/application equipment, and labor. Because of these factors, we believe it is also important to develop best practices for surface application of liquid urine-derived fertilizers.
In collaboration with a multidisciplinary team coordinated by the University of Michigan and funded by an NSF grant, the Rich Earth Institute is developing and testing new UDF production techniques while conducting social research about public perception and attitudes. One of the new UDF formulas is an acid-stabilized urine concentrate which contains most of its nitrogen in urea form. Our hope is that this fertilizer will absorb fully into the soil before the urea can degrade into ammonia, greatly reducing volatilization. An alternate alkali stabilization method (Randall, 2016) also maintains nitrogen in urea form, but at a high pH. This product also seems likely to initially absorb into the soil with low ammonia losses, but the product’s high pH could drive later volatilization once soil urease degrades the urea into ammonia.
The potential for urine-derived fertilizer production is immense. With 65.6 million people living in the Northeast SARE region, each producing 4 kg of nitrogen in their urine annually (Vinnerås, 2002), there is a maximum potential to source 262 million kg of nitrogen fertilizer each year (not to mention P, K and trace nutrients). This could meet 84% of the region’s approximately 312 million kg demand for N fertilizer (EPA, 2017).
Cooperators
- (Educator and Researcher)
- (Researcher)
- (Educator and Researcher)
- (Educator and Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Educator)
- (Educator)
- (Educator)
- (Educator)
- (Educator and Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
- (Researcher)
Research
Ammonia Volatilization Trials
The purpose of this experiment was to measure how much of the nitrogen in urine was lost into the air through the process of ammonia volatilization, following the application of urine to perennial grass under different scenarios (including different field conditions, application methods, and urine pre-treatment methods). We conducted four trials testing four variables:
-
- Urine application method
- Ground wind speed
- Irrigation/rainfall
- Urine pre-treatment methods
We measured ammonia loss by first applying urine to small subplots in a hayfield, and then immediately placing a plastic container over each subplot so that any ammonia that volatilized would be held in the air within the container. Inside each container was an ammonia trap, which absorbed the ammonia from the air and saved it for later analysis.
There are many ways of measuring ammonia volatilization in the field, each with advantages and disadvantages. We chose a closed dynamic chamber method, which uses an acid trap placed inside a transparent chamber covering the test area. The acid trap contains dilute acid, which catches any ammonia gas that comes into contact with it. Air circulation is facilitated by a small fan suspended over the trap, blowing air down onto the trap. The acid trap can be replaced and analyzed periodically to determine the amount of ammonia captured over time. The method we used was based on the method described by Campbell-Nelson (2009).
Pre-trials in 2018 and 2019

Before starting the field trials, we tested the method in a series of pretrials to validate and optimize the design. An acid trap (containing H2SO4) and an ammonia emitter (containing a known quantity of ammonia solution) were placed on a table, covered with an enclosure, and left for 24 hours. The acid trap and ammonia emitter were both then removed and analyzed. The emitter contents were titrated with 0.1N H2SO4 to confirm that the ammonia had fully volatilized. The acid trap contents were analyzed to determine the amount of ammonia captured. This tabletop testing was repeated in a variety of different configurations, including with and without fans blowing air onto the acid traps, and with and without air exchange holes in the sides of the chambers.
Analysis of the acid traps was initially attempted colorimetrically in 2018 using the Hach High Range Ammonia Nitrogen Test ‘N Tube method. Unfortunately, the colorimetric quantification produced inconsistent results that we were not able to resolve, which delayed the rest of the experiment until the field season of 2019. In 2019 we developed a new quantification method using titration with 0.1N NaOH to measure the amount of acid that had been neutralized as the trap absorbed ammonia. This worked well and is the method used and described in the rest of this report.
In preliminary indoor table-top testing of the ammonia capture system, using a chamber without holes and a fan circulating air over the ammonia trap, we measured a 93% ammonia capture rate. However, when we tested the ammonia capture chamber outdoors on turfgrass, the capture rates declined dramatically. We again tested chambers with and without ventilation holes, with different size fans, with weights or stakes securing the chamber (to minimize gaps between the chamber and the ground), and with a hard surface placed over the grass inside the chamber to eliminate absorption into the grass our soil. Chambers without holes had slightly higher capture rates than chambers with holes, but still only captured 30-40% of volatilized ammonia regardless of any modifications made.
To account for this low capture rate, we added a set of positive controls to each field trial, in order to calculate what fraction of the volatilized ammonia was actually captured in the trap. This positive control treatment involved placing urine in a dish, rather than applying it to the ground, thus ensuring that all the ammonia would volatilize. By dividing the amount of ammonia captured by the amount of ammonia known to have been volatilized from the urine in the dish, we calculated the ammonia capture rate. We then used the ammonia capture rate from the positive control as a reference for calculating the percentage volatilized from the other treatments. For example, if the trap in the positive control captures 40% of ammonia in the urine, and a soil application captures 10%, the adjusted capture rate for the soil application is 10/40 = 25% volatilization. A weakness of this method is that it assumes that the unexplained loss is the same in all chambers. However, the values that we obtained in the field trials are within the ranges that we expected, and we feel it is reasonably accurate.
Field Trials in 2019
Experiment Location and Soil Type
All field trials were conducted on the back hay field at Fair Winds Farm in Brattleboro, VT. Different parts of the field were cut at different points during the summer, allowing us to place each trial on a different section of newly-cut hay.
The soil in this field is a sandy loam, with 72% sand, 21% silt, and 7% clay, and a pH of 6.6. The top two inches of soil contain 7% organic matter and have a CEC of 11.1me/100g.
We would have liked to repeat the irrigation trial on a second farm with a less sandy soil, but because of the extensive method development phase, there was insufficient time to do this.
Equipment and experiment setup
Chambers
The volatilization chambers were Sterilite brand 58-quart / 55-liter plastic storage tubs turned upside down. The dimensions of each chamber were:
23 ½” L x 16 ⅞” W x 12 ¼” H
59.7 cm x 42.9 cm 31.1 cm
Chambers were placed in 3 rows of 6 each. Rows were spaced approximately 1 meter apart, and chambers within each row were spaced approximately 50cm apart. Each chamber was held in place by two 5.7” plastic tent stakes, one on each end.

Experimental setup with chambers and weather station
Acid traps
Acid traps contained 250 mL of 0.1N H2SO4. The traps were clear, round, 500-mL polypropylene food containers with snap-on lids. All acid traps were filled and weighed in the lab and capped for transport to the field. These same containers were also used to transport urine from the lab to the field.
Each trap was held approximately 1.5” off the ground with a stand made of rigid plastic mesh. A 6x6cm Sunon brand fan was placed on top of each trap to circulate air to the surface. At 12V, each fan drew 0.18 amps and produced a wind speed of 9.7mph. Fans were attached to small, stiff wire fixtures that rested over the acid traps.
The traps were placed in the application area, avoiding placing them directly on top of the applied urine (except when broadcast), and the chamber placed over the entire area as quickly as possible after application.
The first two trials used a 12 volt, 75 amp hour lead-acid battery to power the small fans and weather station. The battery was placed in the center of chamber arrangement underneath a smaller plastic chamber and swapped with a fully charged battery at each sampling time. Therefore two of the experiment chambers were right next to this smaller tub, which may have slightly changed the microclimate in those two chambers.
Urine application
The urine used in trials 1-3 was collected from our portable urine collection toilets and urinals at a large public festival, and pasteurized at 80°C for 1.5 minutes using custom pasteurization equipment.
108 g of urine was applied to a 12” x 15” area within the footprint in each chamber. The urine had an N content of 6.13 g/L, resulting in an application rate of 50.8 lbs N/acre. The 12” dimension was chosen to match the spacing of the custom applicator that we currently use for farm field application of urine. The 15” length was chosen arbitrarily to fit within the chamber.
All trials included two sets of controls:
-
- Blank control: acid trap with no urine applied. In all trials, all controls yielded an ammonia capture rate of 0 or slightly below 0.
- Positive control: Urine was poured into the cut-off bottom of a gallon plastic water jug - the “emitter.” The emitter was placed on a support and adjusted until it was roughly level, with the liquid covering the bottom of the emitter. The positive control was included as a reference in order to calculate the capture rate, as described in the pretrial section.
Titrations
The amount of ammonia caught in each trap was calculated by measuring the quantity of acid neutralized in the trap. To calculate this, we recorded the volume and concentration of acid that was added to the trap before it was placed in the enclosure, to quantify the total moles of acid in the trap. After the trap was exposed to ammonia, we determined the volume of the trap contents again and titrated a subsample with 0.01 M NaOH, to determine the total moles of acid remaining. This allowed us to calculate the moles of acid (H+) consumed during the experiment, which equals the moles of ammonia captured, assuming no other sources of alkalinity entered the trap.
Sampling times
Due to the very rapid ammonia volatilization rate from liquid manure (and presumably urine), with the great majority of ammonia loss occurring within the first two days (Meisinger and Jokela, 2000), trials 1-3 were sampled after 24 hours and 48 hours. These samples accounted for most of the ammonia captured. A final sample was taken on day 3 or 5, depending on scheduling availability, but the amount collected in the final sample was always very small (<10% of total).
We used a longer sampling window in the fourth trial testing urea and urine treatments, because urea is known to volatilize ammonia over a longer time period. This trial was sampled on days 4, 8, and 12.
Weather data collection
We set up a weather station next to the chambers to record temperature and rainfall. There were two thermometers, one measuring the ambient air temperature, and one inside a control chamber, measuring the interior chamber temperature. This data was logged on an SD card and downloaded at each sampling time for the first two trials. The Arduino was damaged during setup of the third trial, so there was no weather data for that trial. For the final trial, the data was recorded directly from the Arduino.
Soil moisture sampling method
After setting up each experiment, we used a homemade drill-soil corer with a 2 centimeter diameter to take soil samples from in between the chambers and the area immediately around them (no further than 1m away from any chamber).
For each trial we collected three replicates, each consisting of six soil cores of the top 2” of soil collected from random locations in the setup area. (18 total 2” soil cores taken). Each replicate was oven-dried at 140°F until a stable weight was achieved, which was used to calculate the starting soil moisture level.
Data analysis
The charts display the average amount of ammonia captured for each treatment adjusted by the average amount captured in the positive control, with error bars indicating compound error from this calculation. To calculate the compound error when dividing the average value for each treatment (a) by the average value for the positive control (b)
Compound error formula: 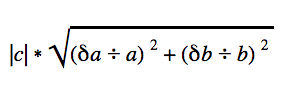
Where a is the standard deviation from average a, b is the standard deviation of average b, and c is the resulting number from the calculation involving a and b.
Significant difference between treatments was analyzed using the actual total amount of ammonia captured, not the amount adjusted by the positive control. These data were tested for homogeneity of variance using the Levene test and for normality using the Shapiro-Wilk test (both tests accepted at p>0.05). Non-normal data were transformed and reanalyzed for normality. Data were then analyzed for significant differences using a one-way analysis of variance (ANOVA) and Tukey post hoc test, with significance determined at p<0.05. Significant difference is indicated with letters in each chart. Statistical analysis was carried out with R version 3.6.2 using RStudio version 1.2.5033, and plots were created using Microsoft Excel 2016.
Details on the setup of individual trials
1. Application method trial
Description of each treatment:
-
- Broadcast: Urine was poured into a 120 mL plastic bottle with a lid perforated with holes. Bottle was squeezed evenly over 12”x15” application area.
- Band: Urine was poured into a 120 mL plastic bottle and poured onto the ground in two 15” rows 12” apart.
- Spot: Urine was poured directly onto the ground in one place within the application area.
- Injection: Two 5” holes were made in the ground using a plastic rod. Approx 54 mL of urine was injected into each hole using a syringe pressed firmly into the ground. A small fraction of the urine rose and puddled around the surface, but quickly sank back into the soil.
- Broadcast: Urine was poured into a 120 mL plastic bottle with a lid perforated with holes. Bottle was squeezed evenly over 12”x15” application area.
Sampling times:
Traps were changed and titrated after 24, 49, and 117 hours, except the control, which was only titrated after 117 hours, due to a miscommunication.

Setting up Application Method trial
|
Application method trial |
|||
|
# replicates |
# sampling times |
Fertilizer type |
Fertilizer application method |
|
3 |
3 |
urine |
band |
|
3 |
3 |
urine |
spot |
|
3 |
3 |
urine |
broadcast |
|
3 |
3 |
urine |
injection |
|
3 |
1 |
None (control) |
n/a |
|
3 |
3 |
None (positive control) |
n/a |
2. Irrigation trial
Setup:
This trial was originally conceived to test different starting soil moisture levels, but due to the challenge of creating different starting soil moisture levels within in each chamber area, we modified this trial to test simulated rainfall/irrigation before and after urine application.
We started this experiment after a period of warm, dry weather, with soil that had a much lower moisture level than in the previous trial. The urine was applied in bands by pouring from a 120 mL bottle in two 15” lines within a 12”-wide irrigation area.
Description of each treatment:
Irrigation water was applied 1 hour before urine application, immediately after urine application (within less than a minute), 1 hour after urine application, or not at all.
To apply the irrigation water, we measured approximately 500mL tap water into a 1000mL bottle with holes drilled into the lid. We squeezed the water out over the application area, marked by flags, attempting to cover it evenly.
Irrigation calculation:
We had planned to simulate typical July rainfall events by irrigating with 0.5 inches of water, which was determined to be a typical rainfall event by referencing data for July from the Advanced Hydrologic Precipitation Service for Vermont on NOAA’s webpage. This would have required 1.5L of water per application area (12 x 15 x 0.5 inches = 90 cubic inches, divided by 60 cubic inches per liter). However, we made a mistake while applying, and instead applied the equivalent of a light rain of 0.17 inches (⅓ the intended rate).
Sampling times:
Traps were changed and titrated after 24, 47, and 71 hours.
|
Irrigation Trial |
||||
|
# replicates |
# sampling times |
Fertilizer type |
Fertilizer application method |
Irrigation timing |
|
3 |
3 |
urine |
band |
1 hour before application |
|
3 |
3 |
urine |
band |
No irrigation |
|
3 |
3 |
urine |
band |
Immediately after application |
|
3 |
3 |
urine |
band |
1 hour after application |
|
3 |
3 |
None (control) |
n/a |
No irrigation |
|
3 |
3 |
None (positive control) |
n/a |
No irrigation |
3. Wind speed trial
Setup:
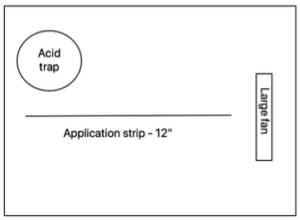
We used 120mm tube-axial fans (called “large fans” in this description to differentiate from the small fans on the acid traps) mounted on dowels driven into the soil to create high, medium, and low wind speeds at ground level inside each chamber. Fan orientation was checked using a square to ensure it was pointed parallel to the ground. We used a handheld Kestrel 2000 wind speed meter to measure the wind speeds. These measurements were taken at both ends of the application strip (where the near end is defined as the point on the application strip closest to the large fan). The wind speeds we observed are in the table below.
NOTE: The wind speeds in the table below were measured within about 2” of the ground. In field measurements taken during experimental design to assess typical field conditions, wind speed at chest height was several times higher than wind speed at 2”.
|
Wind Speed |
Wind Speed at Near End |
Wind Speed at Far End |
|
High |
4.0 mph |
2.0 mph |
|
Medium |
2.0 mph |
1.0 mph |
|
Low |
1.5 mph |
0.7 mph |
The urine was poured in a single 12” strip in front of the fan (starting ~2 inches from the fan) and marked by flags. The acid trap was set up in the same corner of every chamber. Each chamber was placed so that there was approximately 2” between the fan and the rear wall of the chamber. Each chamber had this layout:
The large fans only operated for the first 48 hours, after which they were removed from all chambers.
The previous two experiments had been logging very high temperatures (up to 54°C) inside the chambers. To help keep the internal temperatures more realistic, we draped shade cloth over the bins after they were set up and secured it in place with plastic tent stakes.
Sampling times:
Traps were changed after 24, 48, and 117.5 hours.

Setup of trials 3 and 4 with solar panels and shade cloth
|
Wind speed trial |
||||
|
# replicates |
# sampling times |
Fertilizer type |
Fertilizer application method |
Wind speed |
|
3 |
3 |
urine |
band |
high |
|
3 |
3 |
urine |
band |
medium |
|
3 |
3 |
urine |
band |
low |
|
3 |
3 |
urine |
band |
calm |
|
3 |
3 |
None (control) |
n/a |
calm |
|
3 |
3 |
None (positive control) |
n/a |
calm |
4. Urine pre-treatment trial
Background:
When urine leaves the body, nearly all of the nitrogen is in the form of urea. Once it enters the environment, the urea is rapidly hydrolyzed into ammonia (NH4) and CO2, raising the pH of the urine to around 9. Some urine collection methods seek to prevent this hydrolysis, because preventing hydrolysis helps prevent precipitation of minerals in plumbing and also minimizes odor and NH4 emissions in handling, storage, and transportation of urine. Because the urease enzyme only functions in a narrow pH range, modifying the pH either above or below this range is one way to prevent hydrolysis. Adding a small amount of vinegar is sufficient to lower the pH and is currently a common practice in our urine collection program. We were interested in testing whether this could also help reduce ammonia emissions in the field.
Setup:
This trial compared ammonia losses from untreated urine, in which urea had been fully hydrolyzed to ammonia, with losses from urine that had been treated with acid or base to prevent urea hydrolysis.
The urine used in this trial was different than that used in the other trials. It was collected in the bathroom at our research center, using a process that split each urination into equal fractions, and subjected each fraction to a different treatment. The low pH fraction was treated with 5% acetic acid (vinegar) at a rate of 50ml/L, the high pH fraction with slaked lime at a rate of 10 g/L, and the hydrolyzed fraction with no additions (to encourage urea hydrolysis).
Once this urine was collected, we took initial pH and conductivity measurements, and then stored the high and low pH samples at 4°C, leaving the untreated urine at room temperature to hydrolyze. We monitored this fraction periodically and decided it was fully hydrolyzed when the conductivity stabilized around 43 mS. Approx 1 L of each type of urine was then pasteurized in a volumetric flask on a hot plate in the lab, reaching maximum temperatures of 83-88 °C.
Because these urines had a slightly higher N content than the urine used in previous trials, (determined by measuring the ammonia content of the hydrolyzed fraction in our lab,) we used a slightly smaller amount, 103 mL. We used the same band application method as in trials 1 and 2, and the shade cloth and solar panels used in trial 3.
Sampling times:
Traps were changed after 4, 8, and 12 days.
|
Urine treatment trial |
|||
|
# replicates |
# sampling times |
Fertilizer type |
Fertilizer application method |
|
3 |
3 |
Urine (untreated and fully hydrolyzed) |
band |
|
3 |
3 |
High pH stabilized urine |
band |
|
3 |
3 |
Low pH stabilized urine |
band |
|
3 |
3 |
Urea pellet |
broadcast |
|
3 |
3 |
None (control) |
n/a |
|
3 |
3 |
None (positive control) |
n/a |
Site Visits
The purpose of this portion of the study was to engage in dialogue with farmers from a diverse array of livestock farms, in order to explore the best strategies and opportunities for using current or slightly modified equipment to use UDF effectively on their farms for forage crops, while minimizing ammonia losses. We conducted site visits and interviews on ten farms in southern and central Vermont in the late winter of 2019. Some of the interviews included more than one farmer, for a total of 14 farmer participants. Our focus was farms raising livestock and growing forage crops, because our earlier research has suggested that using urine as a fertilizer on animal feed (as opposed to vegetable crops) is currently preferable in terms of public acceptance. We also sought a diversity of farm size, livestock type and management style. Participating farms ranged in size from ten to 1700 acres, with several between 300 and 600. Crops included vegetable gardens, hay and pasture, corn silage, corn grain, as well as other enterprises such as maple sugaring, hemp and cut flowers. Livestock included cows, goats, sheep, horses, alpaca, chickens and pigs. Numbers of livestock ranged from a few to 400 goats, 500 sheep and 1600 cows on the larger farms. Of the ten, two are certified organic, four are “conventional”, and four are “something else” such as “animal-based.”
Site visits lasted approximately 90 minutes and included dialogue based on an interview guide. An extensive list of both quantitative and open-ended questions had been developed from topics and ideas raised in previous farmer surveys (SARE ONE14-218). These were used to create a guideline for dialogue at each site visit, with a focus on allowing each site’s specific characteristics and each farmer’s ideas to be documented, while creating a consistent format for each visit following standard qualitative research procedures. The guide was modified for this study in consultation with agricultural educators Drew Adams and Heather Darby. The interview guide included basic information about the nutrient value of urine, and research results with regard to hay yield, dilution levels, and levels of microconstituents (i.e. pharmaceuticals) based on Rich Earth data.
The documentation and dialogue included observation and discussion of current soil fertility management practices; equipment used; and application methods for synthetic fertilizers, organic amendments, manures, liquid manures and digestates. In addition, the visits included conversation with farmers concerning the potential for incorporating urine-based fertilizers in their management plans using existing or modified equipment, as well as farmer observations and beliefs about the best timing and weather conditions for application of nitrogen-based fertilizers; the potential obstacles farmers foresee in using UDF (including social issues such as customer perception, and best methods for addressing these); recommendations and ideas for equipment modification; and suggestions for additional research, such as recommendations for soil health parameters that could be evaluated in future trials.
At the start of each visit, farmers and interviewer orally went over an “informed consent” document, which farmers signed. The consent form included consent for some portions of the interviews to be published in various media. They received a copy of the form. Farmers received a stipend of $100 for their participation, with the exception of one farmer who refused the stipend.
All site visits were recorded and later transcribed. Given the relatively small number of interviews, this data was not subject to formal content analysis. Instead, the interviews were reviewed for key ideas relevant to the study objectives. We are cognizant of the fact that the results represent the ideas of each farmer and apply only to his or her specific situation. However, we chose a wide range of farm types and sizes to gain as much diversity in perspective as possible within the geographic region of the study.
We are providing farmer participants with copies of the current report. We will solicit comments and feedback which will then be incorporated into articles produced for wider distribution to other farmers and the general public.
Ammonia volatilization trials
Overall, the ammonia volatilization loss in all trials was low to moderate, ranging from 5-34% of applied ammoniacal nitrogen. This is generally lower than the 30-50% ammonia loss common for broadcast dairy manure in Vermont, which can reach 90% in extreme cases (Jokela and Meisinger, 2008). Band application of urine in our trials resulted in losses of 13-34%, similar to losses from band applied liquid pig (17-22%) and cattle (32-42%) manures on grassland (Hansen et al. 2008, cited in Feilberg and Sommer, 2013). Our values are similar to or lower than values of 20–86% reported for band spreading and broadcast application of cattle urine to grassland (Rodhe et al. 1997).
These overall results are encouraging for farmers interested in using UDF, as they indicate that ammonia losses will probably be comparable or lower than losses that farmers are already expecting from urea and manure fertilizers. Furthermore, as with manure, application management can help reduce NH4 losses from UDF. These management methods are outlined in the following trial results.
1. Application Method
Starting soil moisture level: 38.3%
Weather conditions during first 24 hours: sunny/clear
Inside chamber temperature after first hour: 46 °C
Temperature during first 24 hours:
|
Outside Chamber |
Inside Chamber |
|
|
Min |
13.5 |
17.1 |
|
Max |
36.1 |
46.8 |
|
Avg |
23.9 |
29.5 |
The results from this trial indicate that application method is an important driver for ammonia loss from urine. Broadcasting resulted in the largest loss of N to volatilization (though not statistically different from band spreading in this trial) while injection resulted in the lowest losses. The findings agree with what is already well known from ammonia volatilization studies on manure: greater contact with air results in greater ammonia losses. Furthermore, our measurement method did not capture ammonia lost in the air during broadcasting, so in practice broadcasting loss could be even higher than what we measured. Our measured measured broadcast loss of 31% is similar to UVM Extension reports of average losses of around 40% for thin liquid manures in Vermont (dry matter<5%). Urine has a dry matter content around 5% (all of which is dissolved).
Based on the results of this trial, and what is known about losses from other types of liquid manure, we recommend avoiding any method of broadcasting or spraying urine, and instead applying urine in a gentle stream close to (or directly into) the ground. By avoiding broadcasting, farmers have the potential to reduce ammonia losses from 31% down to 13% or below. Better equipment choices could include existing liquid manure application equipment such as injectors, drop/trailing hoses, or drag shoe (also called ‘sleighfoot’ applicators or trailing shoes). Rich Earth has also designed a custom applicator that applies urine in a gentle stream (similar to a drop hose but without the hose) from a distance of about 24 inches above the ground. We did not include a treatment simulating this custom equipment, since we did not have a way to measure ammonia loss in the air, but we believe that losses from this equipment would be similar to band spreading treatment. In the future we may consider altering our custom equipment to apply the urine closer to the ground, possibly by adding hoses. However, earlier designs which included small-diameter hoses experienced problems with clogging.
The broadcast, band, and injection methods in this trial were intended to simulate available manure application equipment. Because our trial only simulated these pieces of equipment, we suggest additional field trials testing ammonia loss from application using the actual equipment.
 Farm partner Dean Hamilton applying urine with Rich Earth’s custom designed urine applicator
Farm partner Dean Hamilton applying urine with Rich Earth’s custom designed urine applicator
2. Irrigation trial
Starting soil moisture level: 19.4%
Weather conditions during first 24 hours: sunny/clear
Inside chamber temperature after first hour: 50 °C
Temperature during first 24 hours:
|
Outside Chamber |
Inside Chamber |
|
|
Min |
13 |
13 |
|
Max |
36 |
54 |
|
Avg |
23 |
30 |
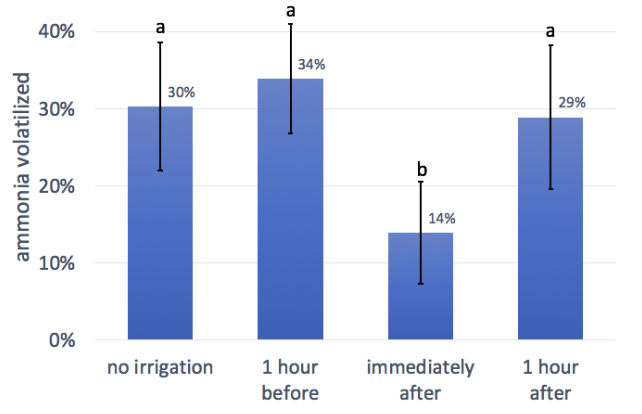
This trial showed a significant reduction in ammonia loss only for irrigation immediately after urine application. However, this trial was conducted in hot, sunny weather on dry soil, in chambers that reached internal temperatures of 50°C one hour after setup. Furthermore, the amount of irrigation/rainfall applied was low--only 0.17”. Given these factors, we suspect that the irrigation water applied 1 hour may have completely evaporated from the soil prior to urine application. Likewise, most of the ammonia may have volatilized before the irrigation in the “one hour after” treatment. We hypothesize that in less extreme circumstances and/or with greater irrigation volume, the 1-hour before and 1-hour after treatments might have more of an effect.
Still, we can conclude from this trial that applying urine immediately before rainfall could reduce ammonia losses. For the purposes of making recommendations, we are describing this as during rainfall, rather than “immediately before” rainfall. This is because the time between application and irrigation in our trial was truly immediate - only a few seconds - whereas on a farm, many people might consider 1 hour before to be “immediately” before (and application itself could take several hours). Therefore, our guideline is to apply urine during rainfall or concurrent with irrigation. This could reduce ammonia losses by about half, compared to applying to dry soil.
3. Wind speed
Starting soil moisture level: 29.6%
Weather conditions during first 24 hours: partly cloudy
Temperature during first 24 hours: not available
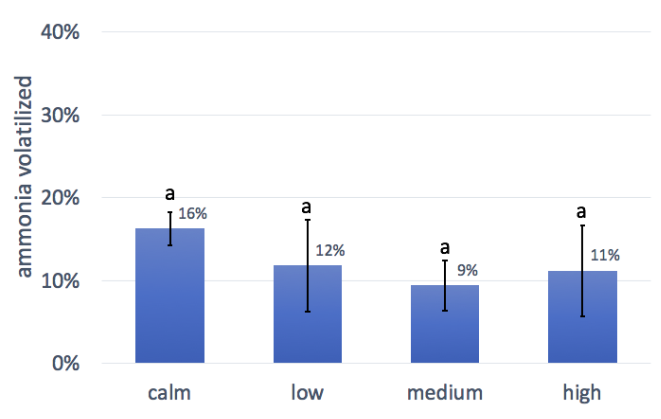
There were no significant differences in the different wind speed treatments in this trial. The non-significant trends show lower ammonia emissions with wind, which is the opposite of what we expected based on research on ammonia loss from manure. During the trial we noticed that greater wind speed resulted in less interior condensation in the chambers, possibly due to greater air exchange with the outside, which could be the source of error. Because of this, we are hesitant to draw any conclusion about the effect (or lack of effect) of wind on ammonia losses, and suggest that this trial should be redone with a different method for measuring ammonia volatilization.
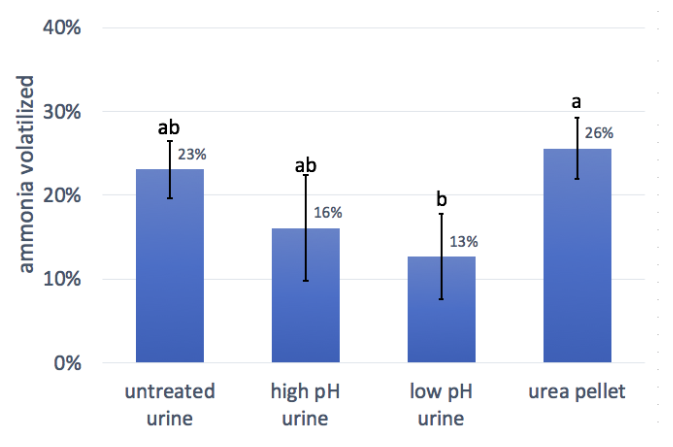
4. Urine treatment
Starting soil moisture level: 33.9%
Weather conditions during first 24 hours: partly cloudy
Inside chamber temperature after first hour: 37 °C
Temperature during first 24 hours:
|
Outside Chamber |
Inside Chamber |
|
|
Min |
14 |
18 |
|
Max |
30 |
38 |
|
Avg |
21 |
26 |
The results of this trial did not show significant difference between any of the urine treatments. However, the differences between the treated urine and untreated urine show a trend towards lower ammonia loss when urea hydrolysis is prevented. This could be because the urine is able to infiltrate while nitrogen is still in urea form, before hydrolysis begins. Since urine pH modification is a viable practice during urine collection, we suggest further exploration of these trends with additional research.
Only one of the urine applications, the low pH treatment, had significantly lower ammonia losses than the urea pellets. This was surprising, since we thought the urine would have lower losses due to better soil infiltration, especially in the absence of rain or irrigation. Still, the reduced losses in the low pH urine compared to urea could be valuable to farmers fertilizing pasture or no-till fields, where manure or urea fertilizer cannot be incorporated.
Interior chamber temperature and starting soil moisture level
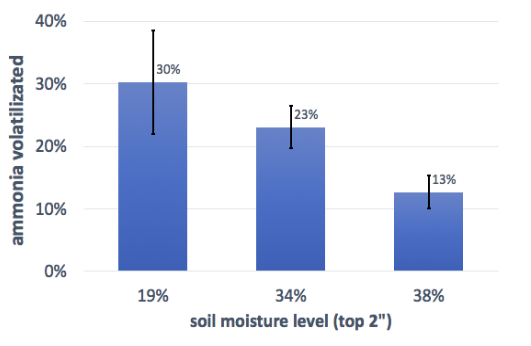
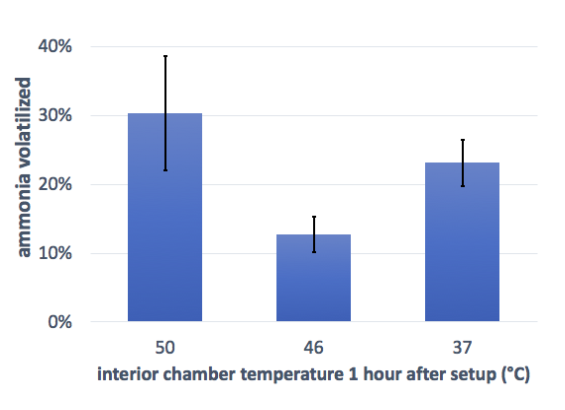
The four trials were sequential over a period of weeks, and two parameters (soil moisture and air temperature within the chamber) varied considerably between trials. Since one of the treatments (band spreading of untreated urine without irrigation) was used in three of the trials, we were able to compare the relative results (loss as a fraction of positive control) for three different pairings of temperature and soil moisture across the three trials. Analysis for significant difference was not possible since the absolute values for each replicate are not comparable across trials.
It’s important to note that the temperature, weather, and soil moisture were all variables across these trials, but each chart only reflects one variable, so the results are not truly comparable. Still, the trend suggests that a higher starting soil moisture level may result in lower ammonia volatilization. This could be because the moist soil helps the urine to infiltrate the soil, rather than staying near the surface; this is similar to the effect we saw in the irrigation trial. Temperature seems to be less important. It would be good to follow up these initial findings with a controlled experiment.
Site visits
The goal of the site visits was to engage in dialogue with area farmers to explore their interest, ideas and recommendations for effectively applying urine-based fertilizers in their farming systems, and to consider options for converting current equipment to enable the inclusion of UDF in nutrient management plans.
Motivation for interest in UDF
The dialogue with farmers suggests there is overall interest and openness to the use of urine or urine-derived fertilizers, specifically as a high-N fertilizer for hay and corn silage, and possibly hemp. Organic farmers noted that the high-N fertilizers and amendments they often use are costly. Farmers also noted interest in UDF because of its potassium content (sometimes a limiting nutrient for these farmers), and potentially for secondary and micronutrients, currently supplied by purchased amendments such as wood ashes. Environmental concerns were also a motivating factor for farmer interest (i.e. keeping human waste local and not "wasting" this potential resource.) Farmers saw urine differently, and (in some cases) more positively than biosolids. It was noted that biosolids are high in phosphorus which many Vermont farmers don't need, but relatively low in nitrogen and potassium, which many do need. On the other hand, the farmers who used biosolids currently, or in the past, noted that they were beneficial to soil organic matter, while UDFs would not have that property.
Current fertilization practices inform the role that urine could play in farm nutrient management strategies. Most of the farmers interviewed primarily relied on their own livestock manure for fertility management, usually applied with a “traditional” broadcast manure spreader in the spring and/or fall. Farmers used composted manure, liquid manure, and manure mixed with bedding. Several of the farmers relied on rotational grazing of animals on pasture, as well as crop rotation and cover crops, to supply their nutrient needs. Several of the farmers also purchased wood ashes, specifically to meet potassium and boron requirements for grasses, and several purchased lime. Some purchased amendments for trace nutrients, such as greensand and Azomite. Those who buy in fertilizers and amendments mentioned urea, potash, phosphate and sulfate. One used a 26-0-26 synthetic fertilizer on pasture, while another used a 20-5-25 mix. One farm used the organic mix “Pro-Grow” (5-3-4). Some also used alfalfa and soybean meal as high-N fertilizers. One also uses fish emulsion as a foliar feed. One farm currently used biosolids, for which the farm is paid by the municipality that provides it, and two of the farms had used biosolids in the past.
Many of the farmers reported that they do not get enough nutrients for their farms from their own livestock manure. Several had developed bartering relationships with other farmers, exchanging animal feed for manure, for example. The prospect of additional source of nutrients was attractive, especially a high N fertilizer for grass crops, corn, and hemp. The additional potassium and micronutrients present in urine were also attractive and could serve to reduce external purchased inputs where these nutrients are limiting. In addition to meeting existing farm nutrient needs, the potential for more controlled and even application of UDF (as compared to broadcasting manure) was appealing. For farmers grazing animals on pasture, there was a potential benefit of urine over manure in terms of timing of applications (because of potential pathogens in manure) as well as palatability. One said, “If you could avoid the biosecurity risk of the manure on the pasture, and the palatability problem of spreading manure on the pasture, then human urine would be a great option there.”
Application, storage, and equipment modification
With regard to application, farmers generally fertilized in the spring or fall and commented that when applying liquid manure or synthetic nitrogen-based fertilizer they choose a day when it was raining or just prior to rain and cloudy, to avoid nitrogen loss. On tilled land they incorporated their fertilizer immediately. On pasture, however, the fertilizer was surface applied, hence the importance of timing application to appropriate weather conditions. None of the farmers interviewed owned injection equipment.
Farmers were asked if they would prefer a dry product or a liquid product (although a dry product is not currently available). There were differing opinions about this. Several farmers thought a dry product could offer more application flexibility, have lower labor requirements, and could ease timing concerns if it could be mixed with compost or manure. One was concerned that a liquid product could create anaerobic conditions which could harm soil microbiota. However, some felt that the UDF could be more evenly distributed if spread in liquid form. Additionally, questions were raised about the expense and energy requirements to create the dry product which might make it cost-prohibitive. One farmer was interested in the possibility of inoculating biochar with urine as part of their fertility and soil management practice. Another suggested the idea of using liquid urine as a foliar feed for corn, in which case the farmer thought he would need diluted urine for this application. This farmer was also interested in side-dressing corn, using some type of adapted equipment that could be rolled over the field when the corn was knee-high.
In terms of storage, facilities to store urine were limited for some farmers. They felt they would need to invest in storage totes, which might be too costly. However, some farmers indicated that urine could be added to current lagoons, slurry tanks, irrigation water, stacked compost, or (in one case) an anaerobic digester. For example, one farmer noted that he had a slurry tank that holds ¾ million gallons, and suggested “We’d probably add it to the pool of our slurry tank and wouldn’t notice it. It would just be some of the drops in the big bucket. The slurry will go on some corn land and it also goes nicely on some pasture land.” It was noted that if a large resource of urine were available, it could be added to the existing lagoons or slurry tanks of larger farms and perhaps re-distributed to the smaller farms.
With regard to equipment modification, most of the farmers interviewed were uncertain as to whether their current equipment could be readily modified for urine application. Most used traditional bucket-loaders and manure spreaders with beaters. Farmers who used liquid manure spreaders thought that urine could be added to this equipment - one noted his liquid manure tank could hold 2300 gallons, and he would like to increase the nitrogen content of his tank. One farmer had dry fertilizer delivered in a tender which they then used to apply on their own fields. This farmer thought that a dry UDF product could be used with this equipment. Others purchased both the dry fertilizer and spreading services. These farmers suggested that the UDF supplying company could also apply the fertilizer to their fields. For example, one said, with regard to a liquid product:
If you had the tools to apply it, like if it came with a sprayer, and you guys had a tank and it could do 10 acres or something like that, I might pay for that service just to have a high nitrogen tank. And then the real dream, if you had something that could pour [i.e. a dry product] I would pay a fair amount for that.
Farmers suggested that rather than modifying their own equipment, some kind of cost-sharing or cooperative use strategy could facilitate the development and use of custom equipment. More discussion with several of these farmers might yield possibilities for equipment design. One farmer suggested using UDF in a system on his farm that currently pipes lagoon-stored liquid fertilizer (such as dairy whey) to pastures, and spreads it using an irrigation gun. This farmer also thought that foliar application could be accomplished with a spray rig using a tank mounted on tractor. Fertigation using existing irrigation drip lines was also a possibility some farmers mentioned.
Limitations and Concerns
Farmers suggested that several factors limited their current ability to use UDF in their nutrient management plans. For the certified organic farmers, they would require organic certification for UDF because, although UDF are not specifically prohibited, they fall into a “grey area” and the participating organic farmers felt strongly that this created too much of a risk. For the larger farmers, there was concern about the availability of sufficient quantities to meet their needs. For all farmer participants, there was concern about the availability of the material at the appropriate time, and current lack of appropriate tools/equipment for application. Farmers suggested the possibility of cooperative purchase or shared use of adapted equipment. One suggested, for example, “If a conservation district purchased a urine applicator or something in a way that drips it onto a pasture, I would consider that in a total package as something that might be worth my time and I’d probably pay for the nutrient that way.”
Some farmers did have the capability for storage, or the addition or urine to their current storage of liquid manure or digestate. However, the one farm that utilizes an anaerobic digester had concerns about altering the carefully managed recipe for input materials, and the expected additional permitting requirements for that adjustment.
A significant concern was the perceived higher time and labor requirements for UDF as compared to current amendments; however, if UDF could be integrated into current methods, such as a dry product being spread by a supplier, or liquid UDF added to existing slurry tanks, or applied via fertigation, this problem could be ameliorated. Regarding price, some farmers were willing to pay the same or slightly more than they currently pay for high-N fertilizer if UDF were available, because of a preference for a local product and for “closing loops.” One commented, in this regard: “The price we are paying for synthetic fertilizer doesn’t include all the externalized societal costs. Recycling something locally could potentially reduce some of those impacts.” Others were only interested if the price were equivalent to current purchased fertilizers. There was a wide range of money spent on purchased inputs among this small sample of farmers, and fertilization costs also include the cost of renting equipment or spreading services, so it is difficult to assess how the use of UDF might affect the cost/benefit analysis for nutrient management. That said, several did not currently buy in high-N fertilizer (relying instead on their own manure or bartering arrangements with other farms supplying manure) but were still interested in UDF to enhance current nutrient management.
Several of the farmers interviewed expressed a range of concerns about the effect of UDF on soil and environmental health. For example, some farmers expressed concerns about the effect of high liquid N application on microbial diversity/balance, as well as impact on soil organic matter over time. Several wanted to know more about the effect of potential micro-contaminants on soil microbiota, plant, animal, human health. One said, for example: "I guess, what I wonder is if those levels are going to accumulate in the soil. And then if they accumulate in the soil, it’s going to have a negative effect on the bacteria that are in the soil, you know, making everything available and just leading to a healthy soil. And if you start to take out the beneficial bacteria, then you’re not doing good for the soil." Farmers urged more research in these areas.
Several farmers raised concerns about the potential presence of microcontaminants, such as antibiotics or hormones, specifically in regard to livestock health and well-being. For example, one dairy farmer said “Breeding Jerseys are touchy. If they’re not right on, if there’s anything wrong, they will not breed back and that’s not good for a dairy cow.” This farmer would need to know there were no hormones detected in a UDF product. A few farmers suggested that some livestock may be averse to the odor of human urine. For example, one said: "[Goats] smell everything and might not touch it...That is one of the most important things, because there is no point in using it. It wouldn’t benefit us if we couldn’t use the crop.” While current farmer experience suggests that urine odor dissipates rapidly, more research on palatability was suggested.
Farmers also indicated concern about nutrient losses to waterways, reinforcing the importance of proper timing, amount, and method of application. Farmers would like guidelines, and had some recommendations. For example, they would need the product to be available in accordance with both their seasonal nutrient needs and weather conditions. Several farmers indicated they would want to apply it within 4-5 days of mowing, and timed to appropriate weather. They would like more information about the nutrient composition of urine, particularly with regard to secondary and trace nutrients to determine if UDF could replace some purchased amendments.
Farmers emphasized the importance of careful communication to the general public (i.e. their customers) about this product. They suggested the focus be on “closing loops.” One said: "It’s...it’s as simple as recycling and it’s a recycling process... And maybe that’s the whole key...[that] it’s termed as putting a natural product back into the environment.” This farmer recommended language for the product such as “ultra urea.”
While it is not possible to generalize widely from the ten interviews/site visits conducted, the diversity of farm types and sizes participating in this study enabled us to draw from several different perspectives such that we can now better determine how to meet the needs of specific types of farmers in our region, and we have identified a number of areas of research needed to further support broader use of UDF by area farmers. We are distributing the present report back to participating farmers to solicit their feedback. It is anticipated that this step will help clarify specific recommendations for equipment modification, application guidelines, and research results needed to support acceptance and use of UDF.
Ammonia loss is a known problem when surface-applying manure or urea fertilizers. The goal of the ammonia volatilization trials was to identify best practices for reducing ammonia losses from urine fertilizer, in order to increase fertilizer value and economic benefit to farmers. We addressed this goal by quantifying ammonia loss from urine applied to hayfields under various environmental conditions, using different application methods and urine treatments.
Based on our results, we have identified two guidelines for minimizing ammonia loss from urine application to hayfields:
- Avoid broadcasting urine - instead, apply in a stream close to (or into) the ground. Our trials measured the following ammonia loss rates for different application methods: broadcast 31%; band 13%, spot 8%; injection 5%.
- Apply urine to moist soil or during rainfall or irrigation. In our trial, a small amount of irrigation reduced ammonia loss by one half.
Urine pre-treatments that keep nitrogen in urea form, rather than ammonia form, may reduce ammonia losses. In our trial, low-pH-treated urine lost less ammonia than urea pellets.
Rich Earth Institute currently supplies urine to four farmers in the Brattleboro area, who apply the urine on hayfields using Rich Earth’s custom application equipment. Based on the results of this trial, we will continue using this equipment, but will consider future designs that minimize air exposure by placing urine closer to or directly into the ground. We will time future applications to coincide with rainfall and/or already-moist soil.
From the site visits, we learned that farmers from a wide range of farm types are interested and open to using urine fertilizer for pasture and feed crops (such as corn for silage) both for its environmental benefits and its nutrient value. They have a number of specific concerns regarding: availability (quantity and timing); availability of appropriate storage and application equipment; and labor requirements. Most would like additional information, such as any impacts of urine fertilization on soil biology, aquatic species, and human health. Several were concerned about palatability of urine-fertilized hay for their livestock. Certified organic farmers, while highly supportive, felt that certification by the Organic Materials Review Institute would be essential if they were to use it.
The ammonia trials have provided answers to some farmer questions about how to time applications and what equipment to use. Application method was the most important factor for reducing ammonia losses, but many farms lack liquid fertilizer application equipment, or have only broadcast spreaders. All farms would therefore need access to appropriate equipment (or modification of existing equipment) in order to minimize ammonia losses. Farmers identified that funding opportunities and cooperative ownership arrangements could help overcome this challenge. Currently, Rich Earth owns two pieces of custom application equipment that are successfully shared between four farm partners.
The recommendations for applying to moist soil or during rain come with differing logistical requirements. Our interviews indicated that farmers are already familiar with the practice of timing pasture N fertilization during or just before rainfall. In order to precisely control the timing of UDF application, farmers would need either sealed on-farm storage capability or very flexible and responsive delivery options. (Unsealed storage like lagoons or slurry tanks could result in significant nitrogen losses.) Application to moist soil has a much broader timing window, making it more compatible with on-demand delivery, but moist soil is more susceptible to compaction by application equipment. Additional research is needed to better understand whether application to moist soil is as effective in reducing losses as active rain. Creative thinking and ongoing dialogue with farmers will help us to address the various challenges we have identified regarding timing and application guidance.
Education & outreach activities and participation summary
Participation summary:
Rich Earth organized one on-farm field day on Thursday, August 22, 2019 from 11am-2pm at Whetstone Valley Farm in Brattleboro. Staff researchers Bradley Kennedy and Tatiana Schreiber presented results of the ammonia volatilization trial, and results of the site visits and farmer dialogue, respectively. Three of Rich Earth Institute’s current farm partners (Jay Bailey, Jesse Kayan, and Dean Hamilton) spoke on a panel and shared their experiences using urine for growing hay. After the presentations, farmer Dean Hamilton demonstrated urine application to his hayfields using Rich Earth’s custom urine applicator. Participants at the field day included a UVM Extension Research Associate; a UVM Agronomy Outreach Specialist, a Technical Consultant with the FAO, as well as a number of area farmers.
Two articles have been submitted to peer-reviewed journals on attitudes and perceptions towards urine diversity and re-use as a fertilizer. One is in press with the journal Elementa: Science of the Anthropocene. (Schreiber, et. al. “Integrative Research and Inclusive Innovation for Urine Derived Fertilizer.) The second, (Schreiber, et. al. “Nested Risks and Responsibilities in Urine Derived Fertilizer Potential in New England and the Upper Midwest”) is in review with the journal Sustainability. Both articles were informed by the farmer interviews conducted for this study.
The contents of this report have been distributed to farmer cooperators for their feedback and comments. Their responses will be used to inform ongoing and future research, and in articles for farmers and the general public, such as for the regional NOFA newspaper, The Natural Farmer.
Findings from the ammonia volatilization trial were incorporated into Rich Earth’s guidance document, Using Urine as a Fertilizer in Home Gardens
Not directly a result of this project
During the time period of this grant, Rich Earth has published quarterly newsletters and maintained a Facebook and Twitter presence. Staff and volunteer urine donors marched again in the agriculturally-themed 2019 Strolling of the Heifers parade.
Rich Earth convened more than 50 stakeholders in Vermont at the August 2018 Urine Diversion Summit and a new group of over 35 midwest and west coast stakeholders at the Rich Earth Summit in Michigan in November 2019.
Rich Earth Executive Director, Kim Nace attended the 2019 AgroExpo in St Johns, Michigan, demonstrating a urine derived fertilizer product (acidified, concentrated, and charcoal filtered urine) created in our Lab in Vermont, that was used to fertilize cucumbers, tomatoes and corn in a demonstration plot.
Rich Earth hosted ten tours of its facilities which included the lab, featuring our Research Associates and interns working on the SARE project, among others.
Rich Earth also presented the results of our work to:
- The New Hampshire Septic Designers & Installers Training in Concord, NH, Dec. 2018.
- Green Consortium in Northampton, MA on Dec. 2019.
- Solarfest at Earth Sky Time Farm in Manchester, VT July 2019
- Students from Yale University graduate programs visiting our research center
- Living Futures conference in Seattle, WA April 2019
- Long Island Sound Study Watersheds & Embayments Work Group webinar in Nov. 2019
Additional publications include:
- Guide to Starting a Community Scale Urine Diversion Program - published March 2019
- Village Sanitation Pilot Study Feasibility Report - to be published February 2020.
Public outreach to increase urine supply:
Rich Earth’s local outreach resulted in an increased volume urine collected and distributed to farmers. Interns and our first ECO AmeriCorps service member tabled at our local farmer’s market and food coop to increase participation in the project. Rich Earth also received a $5,000 grant to provide urine collection equipment to participants at no cost. In 2019, Rich Earth collected 10,091 gallons, the most in any year since the project’s inception. Rich Earth received grant funding in 2019 and 2020 from the Long Island Sound Futures Fund to install permanent urine diverting urinals and toilet fixtures in homes and public buildings.
Learning Outcomes
- understanding the nutrient value of urine and urine-derived fertilizers
- awareness of the current state of research on UDF application and treatment
- understanding of application possibilities
Project Outcomes
The site visits enabled us to engage with changing farmer perspectives and practices, and identified key areas for further research, which could include collaborative projects with some of the farmer participants.
The dialogue during site visits proved an important way for farmers to learn more about the potential value of UDF. While some farmers were already familiar with the idea of urine as fertilizer, others were introduced to the concept for the first time. In either case, most participants were supportive of wider UDF use in agriculture. For example, one farmer said:
We’ve understood for a long time, as being primarily grass farmers, that that was one of our number one limiting factors was nitrogen for the plants to grow. And so that’s something that we attend to, in the way we manage the cow manure, trying to not waste it, or let the liquid part of our cow manure…we don’t want that to get away. So it seems very intuitive to me … what Rich Earth is doing.
Another commented:
The whole idea, the first thing that comes to mind, besides using something that normally gets thrown away, is sort of the circular economy, of the idea that maybe the farm is growing some kind of food that makes its way into the community, and then if you could get something back from the community to take back to the farm, that would be a great thing...not just because it would be helpful, but because it would actually be resourceful. And just make all the sense in the world. Rather than just flushing it.
The interviews reinforced the importance of ongoing research on pharmaceuticals and other microconstituents, especially antibiotics and hormones, in terms of impact on livestock, soil microbiota, and aquatic species in order to address farmer concerns, and support their ability to communicate accurate information to their customers.
A significant insight from this research is the strong concern area farmers have for environmental stewardship. They see the use of UDF as a part of that, and a motivation for their interest. For example, one said:
Every ton of urea that goes to the atmosphere you can put a dollar value on—what a ton of urea costs to produce from petroleum, besides the tons of carbon that are burned to volatilize the nitrogen. It’s a carbon footprint thing, ultimately. Because the cost of urea is directly linked to carbon. Any time the price of carbon goes up, fertilizer goes up. So just helping to crack that wanton waste of carbon might help on the public policy side, because you’re dealing with it many times over.
Several of the farmer cooperators in this study felt that, with additional research to support the environmental benefits of UDFs, they could effectively communicate these values to their customers. One noted,"I think my customers [would] know that I checked it out and wouldn’t be doing it if I didn’t think it was a good thing to do.”
Our field day was valuable because many participants said that word-of-mouth and proof of concept were important for enhancing acceptance of urine or UDF among farmers. Two of the farmers who came to the field day are now using sanitized urine as a fertilizer, supplied by Rich Earth Institute. Another farmer attendee was enthusiastic about the use of UDF in organic production and has begun advocacy for organic certification of UDF on the state and national level.
Additionally, one of the farmers who was interviewed also began using sanitized urine as a fertilizer, supplied by Rich Earth. Two other interviewed farmers expressed interest in doing pilot research projects on their farms using UDF for hay, cut flowers, and hemp.
Overall, our methods for both parts of the project worked well and answered our research questions. The major challenge was the underestimation of time required for both parts of the project, particularly for the methods development in the ammonia trials. The site visits might also have been improved by including someone with more technical knowledge of farm equipment and design, to enable more specific recommendations for equipment needs.
The support for UDF among farmer participants, combined with manageable levels of ammonia loss in our field trials, indicate that UDF is a viable option for perennial grass if the additional research questions identified can be answered to farmers’ satisfaction. Some of these research questions are already being addressed by Rich Earth, while others will need to be addressed in the future.
Research needs currently underway:
- Farmers expressed strong interest in effects of UDF on soil health over time (for example, effects on soil organic matter and microbiota). Rich Earth is currently working with the University of Michigan to initiate soil health studies, and is applying for grants to expand this work.
- Rich Earth is working on urine concentration technology to reduce the volume of urine. This would help address farmer concerns about additional labor and storage needs.
- There is interest among hemp farmers in a local source of a high-N fertilizer such as urine, but concern that hemp used for CBD be free of any contamination from microconstituents, so more research was suggested. Rich Earth is currently developing a charcoal filtration technique to remove microconstituents from urine, and is part of an ongoing project investigating pharmaceutical and hormone degradation in soil and uptake in crop tissue.
Future research needs:
- Organic certification would be valuable for many farmers. Research is needed to determine what additional information is required for certification, and appropriate avenues to achieve certification.
- Farmer interest in the possibility of UDF application in different cropping systems suggests a potential need to develop new customized equipment.
- The feasibility of cooperatively owning or sharing equipment, and potential grant and funding pathways should be investigated.
- To understand differing soil and nutrient management needs, site visits and interviews should be conducted with a wider range of farmers, such as those in horticulture, floriculture and silviculture, and farmers in a broader geographic range.
- Some farmers expressed concern about the palatability of urine fertilized hay for livestock and would like to see “taste tests” of urine fertilized forage.
- Additional field trials should be conducted, demonstrating the effect of UDF for a variety of different crops, using different application systems.
- Results from our ammonia volatilization trials should be confirmed in full-scale field trials with actual application equipment, including injectors, band spreaders, custom equipment, and fertigation systems.
- Additional research could also look at ammonia loss in storage to help identify on-farm storage options - for example, lagoon, covered slurry tank, or manure/stacked compost systems
- Our research suggested applying urine to moist soil or during rainfall to minimize NH4 emissions; however, it is possible that this could increase N2O emissions, which are higher in moist soil and in fluctuating soil moisture conditions (Signor and Cerri 2013). Research is needed on N2O emissions in various soil moisture conditions to ensure that recommendations for avoiding NH4 loss do not result in pollution swapping for N2O.
- Farmers were concerned about nutrient losses due to leaching. Research is needed to address nutrient losses under varying weather conditions, and differing soil management regimes.
Future collaborations with nutrient management advisors, NRCS staff, state and national regulators, and certifying agencies are important to achieve more widespread use of UDF in agriculture. We intend to continue building our relationships within the regional farming community through sharing our ongoing research, and dialogue concerning farmer needs.